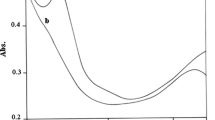
Abstract
Bilirubin (BR) was oxidized by tris-(4-bromophenyl)-ammonium hexachloraantimonate (1), 2,2,6,6-tetramethyl-4-acetyloxypiperidine oxoammonium hexachloroantimonate (2) and 2,2,6,6-tetramethyl-4-methoxypiperidine oxoamonium tetrafluoroborate (3) to the corresponding radical cation. The rate and activation parameters for the reaction were determined. An electron transfer mechanism is proposed based on the kinetic results.
Similar content being viewed by others
References
D.A. Lightner and A.F. McDonagh, Acc. Chem, Res, 17 417 (1984).
R. Schmidt and A.F. McDonagh, in J.B. Stanbury, J.B. Wyngaarden and D.S. Fredrickson (Eds.), The Metabolic Basis of Inherited Disease, McGraw-Hill, New York, 1987, p. 1221.
R. Stocker, Y. Yamamoto, A.F. McDonagh, A.N. Glazer and B.N. Ames, Science, 235, 1043 (1987).
B. Stevens and R.D. Small Jr. Photochem. Photobiol., 23, 33 (1967).
R. Kaul, H.K. Kaul, P.C. Bajpai and C.R.K. Murti, Biosci, 1, 377 (1979).
E. Knobloch, F. Mandys and R. Hodr, J. Chromotogr. Biomedical Applications, 428, 255 (1988).
E. Knobloch, R. Hodr, J. Herzmann and V. Houdkova, J. Chromotogr., 375, 245 (1986).
Q.-X. Guo, L. Yang, B. Liu and Y.-C. Liu, Chem. Res. Chin. Univ., 8, 301 (1992). [Analysis for (BR** BF4) caled for C33H36BF4N4O4: C, 58.97; H, 5.40; N, 8.34. Found C, 59.21; H, 5.94; N, 7.93. λmax(CH3CN): 650 nm.]
F.A. Bell, A. Ledwith and D.C. Sherrington, J. Chem. Soc., Commun., 2717 (1969).
R.I. Zhdanov, V.A. Golubev, V.M. Gida and E.G. Rozantsev, Dokl. Akad. Nauk. SSSR, 196, 856 (1971).
R. Brodersen, J. Biol. Chem., 254, 2364 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guo, Q.X., Wang, J., Guo, X. et al. Kinetics and mechanism of one-electron oxidation of bilirubin in dichloromethane solution. Res. Chem. Intermed. 22, 23–29 (1996). https://doi.org/10.1163/156856796X00340
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1163/156856796X00340