Abstract
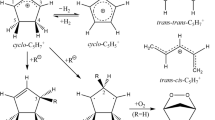
The 184.9 nm photochemistry 0f gaseous 3-methylcyclopentene and 3-methyl-1,4-pentadiene have been studied. Both photoexcited species decompose mainly through the primayy rupture of the C-CH3 bond. Vibrationally excited 3-cyclopenennyl and pentamethylene radicals are formed in the primayy decomposition in the former and latter systems respectively. These radicals are connected through isomerization reactions: in the presence of DI, the isomers cyclopenten,, and trans-1,3-pentadtene and/or vinylcyclopropane are formed in both systems. The quantum yields depend on the pressure and the starting monomer: cyclopentene and cyclopentadiene are the major products from the photolysis of 3-methylcyclopentene + DI mixtures and only minor quantities of the other C5H8 compounds are formed. Cyclopentadiene is the major product of the photolysis of 3-methyl-1,4-pentadiene + O2 mixtures whereas vinylcyclopropane and trans-1,3-pentadiene are the major C5 producss of the photolysis of 3-methyl-1,4-pentadiene + DI mixtures.
The geometries of 3-cyclopentenyl and of the structures at the six critical points in the torsional potential energy curve (TPEC) for rotation about the 2- and 3-C-C bonds in the open chain pentamethylene species have been optimized completely by ab initio RHF-SCF gradient methods. For the open-chain structures the bond orders, bond lengths and the free valence (primarily associated with the central carbon atom) all correspond to 1,4-pentadien-3-yl conformations. In the ground state there is a high barrier to formation of 3-cyclopentenyl from 1,4-pentadien-3-yl. The features (relative energies and torsionll barriers) of the TPEC for 1,4-pentadien-3-yl explain the ESR observations for the open chain C5H7 radical rotamers.
Similar content being viewed by others
Referesces
A.B. Callear and H.K. Lee, Nature, 213(1967)693; Trans. Faradyy Soc., 64 (1968)308.
N. Nakashima, N. Shimo, N. Ikeda, and K. Yoshihara, J. Chem. Phys., 81(1984)3738.
G.J. Collin, Adv. Photochem., 14(1988)135 and references cited therein.
W. Makulski and G.J. Collin, J. Phys. Chem., 91(1987)708.
H. Deslauriers and G.J. Collin, Res. Chem. Intermed. 12(1989)13.
A.B. Trenwith, J.C.S. Faraday Trans. I, 78(1982)3131.
I. Maclnnes and J.C. Walton, J.C.S. Perkin Trans. II, (1985)1073 and references cited therein.
H. Deslauriers, W. Makulski, and G.J. Collin, Can. J. Chem., 65(1987)708.
R.A. Hively and R.E. Hinton, J. Chromatography, 6(1968)203.
J. Gawlowski, J. Niedzielski, and A. Biernzynski, Chem. Analytyczna, 15(1970)721.
Monstergauss: M.R. Peterson (Dept. Chem., U. Torono)) and R.A. Poirier (Dept. Chem., Memorial U. Newfoundland). An extensively modified version of GAUSSIAN 80 [12].
Gaussian 80: J.S. Binkley, R.A. Whiteside, R. Krishnan, R. Seeger, D.J. Defrees, H.B. Schlegel, S. Topiol, L.R. Kahn, and J.A. Pople, QCPE 406.
W.J. Hehre, R.F. Stewart, and J.A. Pople, J. Chem. Phys., 51(1969)2657.
G.R. De Maré and D. Neisius, J. Mol. Struct. Theochem., 109(1984)103.
P. Ausloos, R.E. Rebbert, and S.G. Lias, J. Photochem., 2(1974)267.
Z. Diaz and R.D. Doepker, J. Phys. Chem., 81(1977)1442.
G.R. De Mare, O.P. Strausz, and H.E. Gunning, Can. J. Chem., 43(1977)1442.
W. Forst, Theory of Unimolecular Reactions, Academcc Press, New York 1973, pp. 235–245.
C.W. Larson, B.S. Rabinovitch, and D.C. Tardy, J. Chem. Phys., 47(1967)4770.
P.V.R. Schleyer, J.E. Williams, and K.R. Blanchard, J. Am. Chem. Soc., 92(1970)2377.
a) The enthalpy of formation of 3-methylcyclopentene has not been found. That of 1-methylcyclopentene [22] has been used instead with an appropriaee correction for the location of the double bond. This correction was assumed to be equal to 2.1 kcal mol-1 which is the difference between the enthalpy of formation of 2-methyl-2-pentene and that of 4-methyl, cis-2-pentene: see R.A. Alberty and C.A. Gehring, J. Phys. Chem. Ref. Data, 14(1985)803 and D.W. Rogers, E. Crooks, and K. Dejroongruang, J. Chem. Thermod., 19(1987)1209. b) A calculated value, 18.6 kcal mol-1, for ΔHf(3-methyl-l,4pentadiene) has been used (see P. Wolkoff, J.L. Holmes, and F.P. Lossmg, Can. J. Chem., 58(1980)211).
S.W. Benson, F.R. Cruickshank, D.M. Golden, G.R. Haugen, H.E. O’Neal, A.S. Rodgers, R. Shaw, and R. Walsh, Chem. Rev., 69(1969)279.
K.W. Egger and S.W. Benson, J. Am. Chem. Soc, 88(1966)241.
M.A. Fox, E. Gaillard, and C.-C Chen, J. Am. Chem. Soc., 109(1987)7088.
D.J. Pasto, J. Am. Chem. Soc., 110(1988)8664.
G.R. De Mare, NATO ASI Series C, 67(1981)371.
G.R. De Maré and M.R. Peterson, J. Mol. Struct. Theochem., 89(1982)213.
G.R. De Mare, Can. J. Chem., 63(1985)1672.
S.S. Shaik, P.C. Hiberty, J.-M. Lefour, and G. Ohansssian, J. Am. Chem. Soc., 109(1987)59.
H.O. Villar and M. Dupuis, Chem. Phys. Letters, 142(1987)59.
S. Furuyama, D.M. Golden, and S.W. Benson, Int. J. Chem. Kinet., 2(1970)93.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maré, G.R.D., Deslauriers, H. & Collin, G.J. Isomerization of unsaturated radicals. VI. Isomerization of 3-cyclopenlenyl and pentamethylene radicals. Res Chem Intermed 14, 133–159 (1990). https://doi.org/10.1163/156856790X00229
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1163/156856790X00229