Abstract
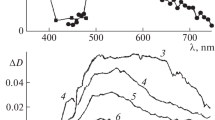
Cis-trans photoisomerization was observed in solution-phase photolysis of some photochromic aziridines, indicating a common planar intermddiate. However, in the solid state, each isomer produced a different color, and no photoisomerization occurred, indicating separate and noninterconvertible structures.
Similar content being viewed by others
References
a) A. Padwa and L. Hamilton, J. Heterocycl. Chem., 4(1967)18; b) T. DoMinh and A.M. Trozzolo, J. Am. Chem. Soc, 94(1972)4046; c) H.W. Heine, R.H. Weese, R.A. Cooper, and A.J. Durbetaki, J. Org. Chem., 32(1967)2708; d) N.H. Cromwell and H. Hoeksema, J. Am. Chem. Soc, 71(1949)740..
a) For a review of photochromies see R. Dessanrr and J.P. Paris, in W.A. Noyes Jr., G. Hammond, and J.N. Pitss Jr. (Eds.), Advancss in Photochemistry, Interscience Publishers, New York, 1963, p. 1; b) G.H. Brown and W.G. Shaw, Rev. Pure Appl. Chem. 11(1961)2; c) T. Bercovici, R. Heiligman-Rim, and E. Fischer, Mol. Photochema, 1(1969)23.
T. DoMnh and A.M. Trozzolo, J. Am. Chem. Soc, 92(1970)6997.
A. Padwa, S. Clough, and E. Glazer, J. Am. Chem. Soc, 92(1970)1778.
For monocyclic aziridines, Cromwell and Hoeksema [1d] reported da-trans photoisomenzation in 1-benzyl-2phenyl-3-tolylazin.me. Unfortunately, we have not been able to verify their result. Huisgen and co-workess reported a thermll cis-trans isomerization of dimethy1 l-(p-methoxyphenyl)-aziridine- 2,3-dicarboxylate, but not as a photoinduced reaction. R. Huisgen, W. Scheer, and H. Hubert, J. Am. Chem. Soc, 89(1967)1753.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DoMinh, T. Photochromic aziridines. A novel photoisomerization and the geometry of colored intermediates in solution phase as compared to solid state. Res Chem Intermed 12, 125–129 (1989). https://doi.org/10.1163/156856789X00087
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1163/156856789X00087