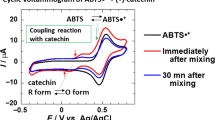
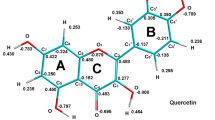
The oxidation of flavonoids is of great interest because of their action as antioxidants with the ability to scavenge radicals by means of electron-transfer processes. The redox reactions of the flavonoid derivative troxerutin, (2-[3,4-bis-(2-hydroxyethoxy) phenyl]-3[[6-deoxy-α-L-manno-pyranosyl)-β-(D-glucopyranosyl]-oxy]-5-hydroxy-7-(2-hydroxyethoxy)-4H-1-benzo-pyran-4-one), were investigated over a wide range of conditions, using pulse radiolysis and cyclic voltammetry. The oxidation mechanism proceeds in sequential steps. One-electron redox potentials for troxerutin were found to be +1.196, +0.846 and −0.634 V vs. NHE.
Similar content being viewed by others
Rights and permissions
About this article
Cite this article
Sarkar, A., Biswas, N., Kapoor, S. et al. One-electron redox reactions of troxerutin in aqueous solutions. Res Chem Intermediat 31, 857–866 (2005). https://doi.org/10.1163/156856705774576137
Published:
Issue Date:
DOI: https://doi.org/10.1163/156856705774576137