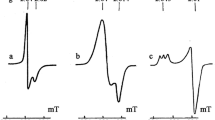
One-electron reduction of several transition metal bis-dithiolenes at the surface of TiO2 particles and in aqueous solutions were studied. Electron Paramagnetic Resonance (EPR) was used to study the reduction at the particle surface and the pulse radiolysis technique was employed for aqueous solutions. Variable temperature EPR measurements following band-gap excitation of the particles showed the disappearance of signals due to the paramagnetic Ni(III) and Cu(II) complexes of 4,5-dimercaptophthalic acid (DMPA), 3,4-dimercaptotoluene (TDT) and maleonitrile dithiolate (MNT), with the surface electron signals vanishing concomitantly. Similarly, the diamagnetic Cu(III) complex of TDT was reduced to the paramagnetic Cu(II) complex. The bimolecular rate constants for the reaction of Ni(III)DMPA and Ni(II)MNT with ea-qwere determined to be 4.3 × 109 dm3 mol-1 s-1 and 1.3× 1010 dm3 mol-1 s-1, respectively. Pulse radiolysis results were used to distinguish between reduction with electron and · CH2OH radical. It is inferred that ea-q · reduces the metal centre while · CH2OH adds to the ligand.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kapoor, S., Kartha, S. & Meisel, D. EPR and pulse radiolysis studies on electron transfer in transition metal dithiolenes at the surface of colloidal TiO2. Res Chem Intermediat 27, 317–332 (2001). https://doi.org/10.1163/156856701104202183
Published:
Issue Date:
DOI: https://doi.org/10.1163/156856701104202183