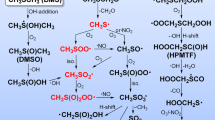
Abstract
The XCO2 (X = F, NH2, CH3) radicals are present in the Earth atmosphere, where they are produced by the degradation of Volatile Organic Compounds (VOCs), either industrial or natural. Here, we use advanced ab initio methodologies to characterize these species in their ground and electronically excited states. Computations are carried out using the Coupled Clusters, both standard and explicitly correlated versions, and multiconfigurational approaches. Several basis sets were used. Afterward, the geometrical parameters and the total energies were extrapolated to the complete basis set (CBS) limit. We also mapped their potentials along the central bond to have insights on the XCO2 → X + CO2 reactions. We thus show that the ground and the lowest electronic excited states are long-lived, for which we provide a set of accurate structural and spectroscopic parameters. The upper electronic states are subject of unimolecular decompositions producing CO2 and X fragments. Our calculations show that the FCO2 (X2B2) → F(2P) + CO2 (X1Σg+) and the CH3CO2 (X2A″) → CH3(X2A″2) + CO2 (X1Σg+) processes require at least 3.5 eV energy to occur, while less energy (of ∼ 2.5 eV) is needed for the NH2CO2 (X2A″) → NH2(X2B1) + CO2 (X1Σg+) reaction. The present findings and data are useful to characterize these radicals in the laboratory, in planetary atmospheres and in combustion and to understand their physical chemistry there.
Graphical Abstract





Similar content being viewed by others
Data availability
This manuscript has no associated data or the data will not be deposited. [Authors' comment: The data of this article consist on spectroscopic parameters, which are already provided in the tables.]
References
F. W. Taylor, Encyclopedia of the solar system. Oxford, Elsevier Science & Technology, 2014, 1311 p. (ISBN 978-0-12-415845-0).
A. Grant, A.T. Archibald, M.C. Cooke, D.E. Shallcross, Modelling the oxidation of seventeen volatile organic compounds to track yields of CO and CO2. Atmos. Environ. 44, 3797–3804 (2010). https://doi.org/10.1016/j.atmosenv.2010.06.049
D. Schröder, M. Semialjac, H. Schwarz, Potential role of methyl-radical adducts with carbon dioxide in the Martian atmosphere. Eur. J. Mass Spectrom. 9, 287–294 (2003). https://doi.org/10.1255/ejms.552
S. von Ahsen, H. Willner, G.A. Argüello, Fluorocarbon oxy and peroxy radicals. J. Fluor. Chem. 125, 1057–1070 (2004). https://doi.org/10.1016/j.jfluchem.2004.02.008
P. Glarborg, J.A. Miller, B. Ruscic, S.J. Klippenstein, Modeling nitrogen chemistry in combustion. Prog. Energy Combust. Sci. 67, 31–68 (2018). https://doi.org/10.1016/j.pecs.2018.01.002
J.S. Francisco, A.N. Goldstein, Z. Li, Y. Zhao, I.H. Williams, Theoretical investigation of chlorofluorocarbon degradation processes: structures and energetics of XC(O)Ox intermediates (X = F, Cl). J. Phys. Chem. 94, 4791 (1990). https://doi.org/10.1021/j100375a010
M.M. Maricq, J.J. Szente, T.S. Dibble, J.S. Francisco, Atmospheric chemical kinetics of FC(O)O. J. Phys. Chem. 98, 12294–12309 (1994). https://doi.org/10.1021/j100098a027
V. Mörs, G.A. Argüello, A. Hoffmann, W. Malms, E.P. Roeth, R. Zellner, Kinetics of the reactions of FC(O)O radicals with NO, NO2, O3, O(3P), CH4, and C2H6. J. Phys. Chem. 99, 15899–15910 (1995). https://doi.org/10.1021/j100043a031
T.J. Wallington, T. Ellermann, O.J. Nielsen, J. Sehested, Atmospheric chemistry of FCOx radicals: UV spectra and self-reaction kinetics of FCO and FC(O)O2 and kinetics of some reactions of FCOx with O2, O3, and NO at 296 K. J. Phys. Chem. 98, 2346–2356 (1994). https://doi.org/10.1021/j100060a024
G.A. Arguello, H. Grothe, M. Kronberg, H. Willner, H.-G. Mack, IR and visible absorption spectrum of the fluoroformyloxyl radical, FCO2°, isolated in inert gas matrixes. J. Phys. Chem. 99, 17525–17531 (1995). https://doi.org/10.1021/j100049a010
Z. Zelinger, P. Dréan, A. Walters, J.R. Avilès Moreno, M. Bogey, H. Pernice, S. von Ahsen, H. Willner, H. Bürgere, High resolution infrared spectroscopy assisted by ab initio calculations. J. Chem. Phys. 118, 1214 (2003). https://doi.org/10.1063/1.1528607
A. Perrin, M. Strizik, H. Beckers, H. Willner, Z. Zelinger, P. Pracna, V. Nevrly, E. Grigorova, First analysis of the high resolution FTIR spectrum of the ν2 band of the FCO2 radical at 9702 cm-1. Mol. Phys. 108, 723 (2010). https://doi.org/10.1080/00268970903514579
S. Bailleux, Z. Zelinger, H. Beckers, H. Willner, E. Grigorova, High-resolution FTIR study of the CO stretching band ν4 of the fluoroformyloxyl radical, FCO2. J. Mol. Spectrosc. 278, 11 (2012). https://doi.org/10.1016/j.jms.2012.06.015
M.M. Maricq, J.J. Szente, Z. Li, J.S. Francisco, Visible absorption spectroscopy of the B2A1–X2B2 transition of fluoroformyloxyl radical, FC(O)O. J. Chem. Phys. 98, 784 (1993). https://doi.org/10.1063/1.464241
L. Kolesnikova, J. Varga, H. Beckers, M. Simecova, Z. Zelinger, L.N. Striteska, P. Kania, H. Willner, S. Urban, Detailed study of fine and hyperfine structures in rotational spectra of the free fluoroformyloxyl radical FCO2•. J. Chem. Phys. 128, 224302 (2008). https://doi.org/10.1063/1.2933499
D.W. Arnold, S.E. Bradforth, E.H. Kim, D.M. Neumark, Study of halogen–carbon dioxide clusters and the fluoroformyloxyl radical by photodetachment of X-(CO2) (X=I, Cl, Br) and FCO2-. J. Chem. Phys. 102, 3493 (1995). https://doi.org/10.1063/1.468575
Y. Sun, M. Wang, C. Yang, Y. Guo, Ab initio study of spectroscopic constants and anharmonic force field of FCO2 radical. J. Mol. Struct. Theochem 951, 77–81 (2010). https://doi.org/10.1016/j.theochem.2010.04.011
M.M. Maricq, J.J. Szente, G.A. Khitrov, J.S. Francisco, Temperature dependent kinetics of the formation and self-reactions of FC(O)O2 and FC(O)O radicals. J. Chem. Phys. 98, 9522 (1993). https://doi.org/10.1063/1.464383
W.F. Schneider, M.M. Maricq, J.S. Francisco, The vibrational spectrum of FC(O)O radical: A challenging case for single-reference electron correlation methods. J. Chem. Phys. 103, 6601 (1995). https://doi.org/10.1063/1.470389
M.L. McKee, T.R. Webb, Fluoroformyl hypofluorite, fluoroformyl peroxyhypofluorite and fluoroformyl peroxide a density functional study. J. Phys. Chem. 100, 11292–11296 (1996). https://doi.org/10.1021/jp960676+
T.J. Wallington, M.D. Hurley, M.M. Maricq, FTIR product study of the self-reaction of FC(O)O2 radicals. Chem. Phys. Lett. 205, 62 (1993). https://doi.org/10.1016/0009-2614(93)85167-M
J.S. Francisco, A.N. Goldstein, Dissociation dynamics of FC(O)O and ClC(O)O radicals. Chem. Phys. 127, 73–79 (1988). https://doi.org/10.1016/0301-0104(88)87108-8
J. Breidung, W. Thiel, Thermochemistry of the fluoroformyloxyl radical: a computational study based on coupled cluster theory. J. Phys. Chem. A 110, 1575 (2006). https://doi.org/10.1021/jp053883v
C.E. Miller, J.I. Lynton, D.M. Keevil, J.S. Francisco, Dissociation pathways of peroxyacetyl nitrate (PAN). J. Phys. Chem. A 103, 11451–11459 (1999). https://doi.org/10.1021/jp992667h
S.M. Park, C.H. Kwon, H.L. Kim, Dynamics of H atom production from photodissociation of acetic acid-d(1). J. Phys. Chem A. 119, 9474–9480 (2015). https://doi.org/10.1021/acs.jpca.5b05241
L.R. McCunn, K.-C. Lau, M.J. Krisch, L.J. Butler, J.-W. Tsung, J.J. Lin, Unimolecular dissociation of the CH3OCO radical: an intermediate in the CH3O + CO reaction. J. Phys. Chem. A 110, 1625–1634 (2006). https://doi.org/10.1021/jp054238r
E.C. Tuazon, W.P.L. Carter, R. Atkinson, Thermal decomposition of peroxyacetyl nitrate and reactions of acetyl peroxy radicals with nitric oxide and nitrogen dioxide over the temperature range 283–313 K. J. Phys. Chem. 95, 2434–2437 (1991). https://doi.org/10.1021/j100159a059
W.R. Stockwell, J.B. Milford, D. Gao, Y.-J. Yang, The effect of acetyl peroxy-peroxy radical reactions on peroxyacetyl nitrate and ozone concentrations. Atmos. Environ. 29, 1591–1599 (1995). https://doi.org/10.1016/1352-2310(95)00093-E
F. De Smedt, X.V. Bui, T.L. Nguyen, J. Peeters, L. Vereecken, Theoretical and experimental study of the product branching in the reaction of acetic acid with OH radicals. J. Phys. Chem. A 109, 2401–2409 (2005). https://doi.org/10.1021/jp044679v
S. Crunaire, J. Tarmoul, C. Fittschen, A. Tomas, B. Lemoine, P. Coddeville, Use of cw-CRDS for studying the atmospheric oxidation of acetic acid in a simulation chamber. Appl. Phys. B 85, 467–476 (2006). https://doi.org/10.1007/s00340-006-2319-6
Z. Lu, R.E. Continetti, Dynamics of the acetyloxyl radical studied by dissociative photodetachment of the acetate anion. J. Phys. Chem. A 108, 9962–9969 (2004). https://doi.org/10.1021/jp040355v
X.-B. Wang, H.-K. Woo, L.-S. Wang, B. Minofar, P. Jungwirth, Determination of the electron affinity of the acetyloxyl radical. J. Phys. Chem. A 110, 5047 (2006). https://doi.org/10.1021/jp060138p
A. Rauk, D. Yu, D.A. Armstrong, Carboxyl free radicals: formyloxyl (HCOO) and acetyloxyl (CH3COO) revisited. J. Am. Chem. Soc. 116, 8222–8228 (1994). https://doi.org/10.1021/ja00097a031
S.D. Peyerimhoff, P.S. Skell, D.D. May, R.J. Buenker, Configuration interaction study of the three lowest electronic states in the formyl and acetyl radicals. J. Am. Chem. Soc. 104, 4515–4520 (1982). https://doi.org/10.1021/ja00381a002
J.S. Francisco, The mechanism of the CH3O + CO reaction and the stability of the CH3OCO radical. Chem. Phys. 237, 1–9 (1998). https://doi.org/10.1016/S0301-0104(98)00222-5
Y.Z. Zhou, S. Li, Q.S. Li, S.W. Zhang, Theoretical investigation of the decarboxylation reaction of CH3CO2 radical. J. Mol. Struct. Theochem 854, 40–45 (2008). https://doi.org/10.1016/j.theochem.2007.12.033
Y. Li, S. Mani Sarathy, Probing hydrogen–nitrogen chemistry: a theoretical study of important reactions in NxHy, HCN and HNCO oxidation. Int. J. Hydrog. Energy 45, 23624–23637 (2020). https://doi.org/10.1016/j.ijhydene.2020.06.083
S. Rosanka, G.H.T. Vu, H.M.T. Nguyen, T.V. Pham, U. Javed, D. Taraborrelli, L. Vereecken, Atmospheric chemical loss processes of isocyanic acid (HNCO): a combined theoretical kinetic and global modelling study. Atmos. Chem. Phys. 20, 6671–6686 (2020). https://doi.org/10.5194/acp-20-6671-2020
H.M.T. Nguyen, T.N. Nguyen, Calculations on the complex mechanism of the HCNO+OH reaction. Chem. Phys. Lett. 599, 15–22 (2014). https://doi.org/10.1016/j.cplett.2014.03.001
D. Sengupta, M.T. Nguyen, Mechanism of NH2 + CO2 formation in OH + HNCO reaction: Rate constant evaluation via ab initio calculations and statistical theory. J. Chem. Phys. 106, 9703 (1997). https://doi.org/10.1063/1.474090
P.J. Knowles, C. Hampel, H.-J. Werner, Coupled cluster theory for high spin, open shell reference wave functions. J. Chem. Phys. 99, 5219 (1993). https://doi.org/10.1063/1.465990
P. J. Knowles, C. Hampel, H.-J. Werner. Erratum: “Coupled cluster theory for high spin, open shell reference wave functions” [ J. Chem. Phys. 99, 5219 (1993)]. J. Chem. Phys. 112, 3106 (2000). https://doi.org/10.1063/1.480886
K. Raghavachari, G.W. Trucks, J.A. Pople, M. Head-Gordon, A fifth-order perturbation comparison of electron correlation theories. Chem. Phys. Lett. 157, 479 (1989). https://doi.org/10.1016/S0009-2614(89)87395-6
T.B. Adler, G. Knizia, H.-J. Werner, A simple and efficient CCSD(T)-F12 approximation. J. Chem. Phys. 127, 221106 (2007). https://doi.org/10.1063/1.2817618
H.-J. Werner, G. Knizia, F.R. Manby, Explicitly correlated coupled cluster methods with pair-specific geminals. Mol. Phys. 109, 407–417 (2011). https://doi.org/10.1080/00268976.2010.526641
G. Knizia, T.B. Adler, H.-J. Werner, Simplified CCSD(T)-F12 methods: Theory and benchmarks. J. Chem. Phys. 130, 054104 (2009). https://doi.org/10.1063/1.3054300
P.J. Knowles, H.-J. Werner, An efficient second-order MC SCF method for long configuration expansions. Chem. Phys. Lett. 115, 259–267 (1985). https://doi.org/10.1016/0009-2614(85)80025-7
H.-J. Werner, P.J. Knowles, A second order multiconfiguration SCF procedure with optimum convergence. J. Chem. Phys. 82, 5053–5063 (1985). https://doi.org/10.1063/1.448627
H.J. Werner, P.J. Knowles, An efficient internally contracted multiconfiguration–reference configuration interaction method. J. Chem. Phys. 89, 5803–5814 (1988). https://doi.org/10.1063/1.455556
P.J. Knowles, H.-J. Werner, An efficient method for the evaluation of coupling coefficients in configuration interaction calculations. Chem. Phys. Lett. 145, 514–522 (1988). https://doi.org/10.1016/0009-2614(88)87412-8
K.R. Shamasundar, G. Knizia, H.-J. Werner, A new internally contracted multi-reference configuration interaction method. J. Chem. Phys. 135, 054101 (2011). https://doi.org/10.1063/1.3609809
T.H. Dunning, Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J. Chem. Phys. 90, 1007–1023 (1989). https://doi.org/10.1063/1.456153
R.A. Kendall, T.H. Dunning, R.J. Harrison, Electron affinities of the first-row atoms revisited. Systematic basis sets and wave functions. J. Chem. Phys. 96, 6796–6806 (1992). https://doi.org/10.1063/1.462569
F.A. Weigend, A fully direct RI-HF algorithm: Implementation, optimised auxiliary basis sets, demonstration of accuracy and efficiency. Phys. Chem. Chem. Phys. 4, 4285–4291 (2002). https://doi.org/10.1039/B204199P
C. Hättig, Optimization of auxiliary basis sets for RI-MP2 and RI-CC2 calculations: core–valence and quintuple-ζ basis sets for H to Ar and QZVPP basis sets for Li to Kr. Phys. Chem. Chem. Phys. 7, 59–66 (2005). https://doi.org/10.1039/B415208E
W. Klopper, Highly accurate coupled-cluster singlet and triplet pair energies from explicitly correlated calculations in comparison with extrapolation techniques. Mol. Phys. 99, 481–507 (2001). https://doi.org/10.1080/00268970010017315
K.E. Yousaf, K.A. Peterson, Auxiliary RI basis sets (OptRI) matched to cc-pVnZ-F12: H, B-Ne. Al-Ar. J. Chem. Phys. 129, 184108 (2008). https://doi.org/10.1063/1.3009271
T. Helgaker, W. Klopper, H. Koch, J. Noga, Basis-set convergence of correlated calculations on water. J. Chem. Phys. 106, 9639–9646 (1997). https://doi.org/10.1063/1.473863
M.C. Lin, S.H. Bauer, Bimolecular reaction of N2O with CO and the recombination of O and CO as studied in a single-pulse shock tube. J. Chem. Phys. 50, 3377 (1969). https://doi.org/10.1063/1.1671561
Y. Ma, L. Peng, H. Zhang, J.-G. Yu, The potential energy surfaces of the ground and excited states of carbon dioxide molecule. Russ. J. Phys. Chem. 88, 2339–2347 (2014). https://doi.org/10.1134/S0036024414130287
M. Hochlaf, C. Puzzarini, M.L. Senent, Towards the computations of accurate spectroscopic parameters and vibrational spectra for organic compounds. Mol. Phys. 113, 1661–1673 (2015). https://doi.org/10.1080/00268976.2014.1003986
M. Hochlaf, Advances in spectroscopy and dynamics of small and medium sized molecules and clusters. Phys. Chem. Chem. Phys. 19, 21236–21261 (2017). https://doi.org/10.1039/C7CP01980G
M. Hochlaf, In-silico astrochemistry of life’s building blocks: Comment on “A never-ending story in the sky: the secrets of chemical evolution” by Cristina Puzzarini and Vincenzo Barone. Phys. Life Rev. 32, 101–103 (2020). https://doi.org/10.1016/j.plrev.2019.08.008
K. Mahjoubi, D.M. Benoit, N.-E. Jaidane, M. Mogren Al-Mogren, M. Hochlaf, Understanding of matrix embedding: a theoretical spectroscopic study of CO interacting with Ar clusters, surfaces and matrices. Phys. Chem. Chem. Phys. 17, 17159–17168 (2015). https://doi.org/10.1039/C5CP01672J
Y. Makina, K. Mahjoubi, D.M. Benoit, N.-E. Jaidane, M. Mogren Al-Mogren, M. Hochlaf, Periodic dispersion-corrected approach for isolation spectroscopy of N2 in an argon environment: clusters, surfaces, and matrices. J. Phys. Chem. 121, 4093–4102 (2017). https://doi.org/10.1021/acs.jpca.7b00093
Acknowledgements
The authors thank the Programme National “Physique et Chimie du Milieu Interstellaire” (PCMI) of Centre National de la Recherche Scientifique (CNRS)/Institut National des Sciences de l’Univers (INSU) with Institut de Chimie (INC)/Institut de Physique (INP) co-funded by Commissariat à l’Energie Atomique (CEA) and Centre National d’Etudes Spatiales (CNES). S.K. thanks a fellowship from the Tunisian ministry for higher education and research. This article is based upon work from COST Action CA18212—Molecular Dynamics in the GAS phase (MD-GAS), supported by COST (European Cooperation in Science and Technology, www.cost.eu). This work was supported by a STSM Grant to S.K. from COST Action CA18212.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection and analysis were performed by all authors. The first draft of the manuscript was written by MH and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors did not receive support from any organization for the submitted work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kechoindi, S., Ben Yaghlane, S., Terzi, N. et al. Characterization and photochemistry of XCO2 (X = F, NH2, CH3) radicals. Eur. Phys. J. Spec. Top. 232, 1905–1916 (2023). https://doi.org/10.1140/epjs/s11734-023-00918-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1140/epjs/s11734-023-00918-1