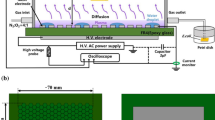
Abstract
Atmospheric pressure plasmas are becoming relevant in local microbial deactivation and other combined effects of plasmas on living organisms. For this reason, our research was focussed on optimisation of atmospheric pressure plasma jet (APPJ) parameters to complete the deactivation of different bacteria strains in a medium. Different helium APPJ treatments with different discharge parameters were used, such as input voltages and gas flows. To better understand plasma properties behind complete bacteria deactivation at optimised discharge parameters, optical and electrical plasma jet diagnostics were performed, including electrical characterisation of the plasma source, optical emission spectroscopy of the plasma plume and intensified charged coupled device imaging of the discharge behaviour for every set of plasma parameters. Then, the resulting plasma liquid chemistry was assessed to establish the connections between reactive species generated in the gaseous and liquid phases. The most efficient deactivation was found for higher discharge powers and gas flow rates, and that was linked to higher densities of reactive oxygen and nitrogen species, especially hydrogen peroxide and medium solvated charges.
Graphical abstract












Similar content being viewed by others
Data availability statement
This manuscript has no associated data or the data will not be deposited. [Authors’ comment: Data available on request from the authors - The data that support the findings of this study are available from the corresponding author, A. J., upon reasonable request.]
References
Z. Dai, J. Ronholm, Y. Tian, B. Sethi, X. Cao, Sterilization techniques for biodegradable scaffolds in tissue engineering applications. J. Tissue Eng. 7, 204173141664881 (2016). https://doi.org/10.1177/2041731416648810
J. Ehlbeck et al., Low temperature atmospheric pressure plasma sources for microbial decontamination. J. Phys. D Appl. Phys. (2011). https://doi.org/10.1088/0022-3727/44/1/013002
R. Ben Gadri et al., Sterilization and plasma processing of room temperature surfaces with a one atmosphere uniform glow discharge plasma (OAUGDP). Surf. Coat. Technol. 131(1–3), 528–541 (2000). https://doi.org/10.1016/s0257-8972(00)00803-3
P.M. Schneider, New technologies and trends in sterilization and disinfection. Am. J. Infect. Control 41(5 SUPPL.), S81–S86 (2013). https://doi.org/10.1016/j.ajic.2012.12.007
A. Sakudo, Y. Yagyu, T. Onodera, Disinfection and sterilization using plasma technology: fundamentals and future perspectives for biological applications. Int. J. Mol. Sci. (2019). https://doi.org/10.3390/ijms20205216
M. Laroussi, Nonthermal decontamination of biological media by atmospheric-pressure plasmas: review, analysis, and prospects. IEEE Trans. Plasma Sci. 30(4I), 1409–1415 (2002). https://doi.org/10.1109/TPS.2002.804220
X. Liao et al., Bacterial spore inactivation induced by cold plasma. Crit. Rev. Food Sci. Nutr. 59(16), 2562–2572 (2019). https://doi.org/10.1080/10408398.2018.1460797
H. Halfmann, B. Denis, N. Bibinov, J. Wunderlich, P. Awakowicz, Identification of the most efficient VUV/UV radiation for plasma based inactivation of Bacillus atrophaeus spores. J. Phys. D Appl. Phys. 40(19), 5907–5911 (2007). https://doi.org/10.1088/0022-3727/40/19/019
S. Hofmann, Atmospheric pressure plasma jets - characterisation and interaction with human cells and bacteria, no. december. (2013)
M. Keidar, E. Robert, Preface to special topic: plasmas for medical applications. Phys. Plasmas 22(12), 121901 (2015). https://doi.org/10.1063/1.4933406
M. Keidar, D. Yan, I.I. Beilis, B. Trink, J.H. Sherman, Plasmas for treating cancer: opportunities for adaptive and self-adaptive approaches. Trends Biotechnol. 36(6), 586–593 (2018). https://doi.org/10.1016/j.tibtech.2017.06.013
S. Schneider et al., The role of VUV radiation in the inactivation of bacteria with an atmospheric pressure plasma jet. Plasma Process. Polym. 9(6), 561–568 (2012). https://doi.org/10.1002/ppap.201100102
N. Gopal, C. Hill, P.R. Ross, T.P. Beresford, M.A. Fenelon, P.D. Cotter, The prevalence and control of bacillus and related spore-forming bacteria in the dairy industry. Front. Microbiol. (2015). https://doi.org/10.3389/fmicb.2015.01418
S. André, T. Vallaeys, S. Planchon, Spore-forming bacteria responsible for food spoilage. Res. Microbiol. 168(4), 379–387 (2017). https://doi.org/10.1016/j.resmic.2016.10.003
S. Caulier, C. Nannan, A. Gillis, F. Licciardi, C. Bragard, J. Mahillon, Overview of the antimicrobial compounds produced by members of the bacillus subtilis group. Front. Microbiol. (2019). https://doi.org/10.3389/fmicb.2019.00302
J. Vila et al., Escherichia coli: an old friend with new tidings. FEMS Microbiol. Rev. 40(4), 437–463 (2016). https://doi.org/10.1093/femsre/fuw005
J. Jang, H.-G. Hur, M.J. Sadowsky, M.N. Byappanahalli, T. Yan, S. Ishii, Environmental Escherichia coli: ecology and public health implications-a review. J. Appl. Microbiol. 123(3), 570–581 (2017). https://doi.org/10.1111/jam.13468
T.J. Foster, J.A. Geoghegan, Staphylococcus aureus, in Molecular Medical Microbiology. (Elsevier, Amsterdam, 2015), pp. 655–674
S.Y.C. Tong, J.S. Davis, E. Eichenberger, T.L. Holland, V.G. Fowler, Staphylococcus aureus Infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 28(3), 603–661 (2015). https://doi.org/10.1128/CMR.00134-14
S. Basak, P. Singh, M. Rajurkar, Multidrug resistant and extensively drug resistant bacteria: a study. J. Pathog. 2016, 1–5 (2016). https://doi.org/10.1155/2016/4065603
L. Poirel et al., Antimicrobial resistance in Escherichia coli. Microbiol. Spectr. (2018). https://doi.org/10.1128/microbiolspec.ARBA-0026-2017
M. Miletić et al., Inhibition of methicillin resistant Staphylococcus aureus by a plasma needle. Open Phys. (2014). https://doi.org/10.2478/s11534-014-0437-z
A.H. Asghar, O.B. Ahmed, A.R. Galaly, Inactivation of E. Coli using atmospheric pressure plasma jet with dry and wet argon discharges. Membranes (Basel) 11(1), 1–20 (2021). https://doi.org/10.3390/membranes11010046
A. Moldgy, G. Nayak, H.A. Aboubakr, S.M. Goyal, P.J. Bruggeman, Inactivation of virus and bacteria using cold atmospheric pressure air plasmas and the role of reactive nitrogen species. J. Phys. D Appl. Phys. 53(43), 434004 (2020). https://doi.org/10.1088/1361-6463/aba066
J.-W. Lackmann, J.E. Bandow, Inactivation of microbes and macromolecules by atmospheric-pressure plasma jets. Appl. Microbiol. Biotechnol. 98(14), 6205–6213 (2014). https://doi.org/10.1007/s00253-014-5781-9
C.A.J. van Gils, S. Hofmann, B.K.H.L. Boekema, R. Brandenburg, P.J. Bruggeman, Mechanisms of bacterial inactivation in the liquid phase induced by a remote RF cold atmospheric pressure plasma jet. J. Phys. D Appl. Phys. 46(17), 175203 (2013). https://doi.org/10.1088/0022-3727/46/17/175203
F. Saadati, H. Mahdikia, H.A. Abbaszadeh, M.A. Abdollahifar, M.S. Khoramgah, B. Shokri, Comparison of Direct and Indirect cold atmospheric-pressure plasma methods in the B16F10 melanoma cancer cells treatment. Sci. Rep. 8(1), 1–15 (2018). https://doi.org/10.1038/s41598-018-25990-9
S. Ikawa, K. Kitano, S. Hamaguchi, Effects of pH on bacterial inactivation in aqueous solutions due to low-temperature atmospheric pressure plasma application. Plasma Process. Polym. 7(1), 33–42 (2010). https://doi.org/10.1002/ppap.200900090
S.M. Hosseini, B. Hosseinzadeh Samani, S. Rostami, Z. Lorigooini, Design and characterisation of jet cold atmospheric pressure plasma and its effect on Escherichia coli colour, pH, and bioactive compounds of sour cherry juice. Int. J. Food Sci. Technol. 56(10), 4883–4892 (2021). https://doi.org/10.1111/ijfs.15220
V.S.S.K. Kondeti et al., Long-lived and short-lived reactive species produced by a cold atmospheric pressure plasma jet for the inactivation of Pseudomonas aeruginosa and Staphylococcus aureus. Free Radic. Biol. Med. 124, 275–287 (2018). https://doi.org/10.1016/j.freeradbiomed.2018.05.083
A.L.V. Cubas et al., Effect of chemical species generated by different geometries of air and argon non-thermal plasma reactors on bacteria inactivation in water. Sep. Purif. Technol. 222, 68–74 (2019). https://doi.org/10.1016/j.seppur.2019.03.057
P. Lukes, M. Clupek, V. Babicky, B. Pongrac, M. Simek, Bulk-phase chemistry induced by nanosecond discharge plasma in water. In: 2017 IEEE 19th International Conference on Dielectric Liquids (ICDL), (2017), pp. 1–3, https://doi.org/10.1109/ICDL.2017.8124702
Y.S. Seo, A.A.H. Mohamed, K.C. Woo, H.W. Lee, J.K. Lee, K.T. Kim, Comparative studies of atmospheric pressure plasma characteristics between He and Ar working gases for sterilization. IEEE Trans. Plasma Sci. 38(10 PART 2), 2954–2962 (2010). https://doi.org/10.1109/TPS.2010.2058870
E. Ilik, T. Akan, Optical properties of the atmospheric pressure helium plasma jet generated by alternative current (a.c.) power supply. Phys. Plasmas (2016). https://doi.org/10.1063/1.4948718
A.N. Korbut, V.A. Kelman, Y.V. Zhmenyak, M.S. Klenovskii, Emission properties of an atmospheric-pressure helium plasma jet generated by a barrier discharge. Opt. Spectrosc. (English Transl. Opt. i Spektrosk.) 116(6), 919–925 (2014). https://doi.org/10.1134/S0030400X14040146
J.J. Camacho, J.M.L. Poyato, L. Díaz, M. Santos, Optical emission studies of nitrogen plasma generated by IR CO2 laser pulses. J. Phys. B At. Mol. Opt. Phys. 40(24), 4573–4590 (2007). https://doi.org/10.1088/0953-4075/40/24/003
S.J. Strickler, The identification of molecular spectra (Pearse, R. W. B.; Gaydon, A. G.). J. Chem. Educ. 41(5), A398 (1964). https://doi.org/10.1021/ed041pA398
T. Darny, J.M. Pouvesle, J. Fontane, L. Joly, S. Dozias, E. Robert, Plasma action on helium flow in cold atmospheric pressure plasma jet experiments. Plasma Sources Sci. Technol. (2017). https://doi.org/10.1088/1361-6595/aa8877
M. Foletto, V. Puech, J. Fontane, L. Joly, L.C. Pitchford, Evidence of the influence of plasma jets on a helium flow into open air. IEEE Trans. Plasma Sci. 42(10), 2436–2437 (2014). https://doi.org/10.1109/TPS.2014.2331393
R. Zaplotnik, M. Bišćan, Z. Kregar, U. Cvelbar, M. Mozetič, S. Milošević, Influence of a sample surface on single electrode atmospheric plasma jet parameters. Spectrochim. Acta Part B At. Spectrosc. 103–104, 124–130 (2015). https://doi.org/10.1016/j.sab.2014.12.004
P.J. Bruggeman et al., Plasma-liquid interactions: a review and roadmap. Plasma Sources Sci. Technol. (2016). https://doi.org/10.1088/0963-0252/25/5/053002
Q. Xiong et al., Temporal and spatial resolved optical emission behaviors of a cold atmospheric pressure plasma jet. J. Appl. Phys. (2009). https://doi.org/10.1063/1.3239512
G. Nersisyan, T. Morrow, W.G. Graham, Measurements of helium metastable density in an atmospheric pressure glow discharge. Appl. Phys. Lett. 85(9), 1487–1489 (2004). https://doi.org/10.1063/1.1784514
G.V. Naidis, Production of active species in cold helium-air plasma jets. Plasma Sources Sci. Technol. (2014). https://doi.org/10.1088/0963-0252/23/6/065014
R. Ono, Optical diagnostics of reactive species in atmospheric-pressure nonthermal plasma. J. Phys. D Appl. Phys. (2016). https://doi.org/10.1088/0022-3727/49/8/083001
Acknowledgements
This work was carried out within projects NATO SPS, and Slovenian Research Agency grant J4-1770. This article is also based upon work from COST Action PLAGRI—CA19110, supported by COST (European Cooperation in Science and Technology), www.cost.eu. K. S. acknowledges also partial funding from bilateral project Serbia-Slovenia from MESTD of Republic of Serbia. We thank Dr. Nevena Puac for useful advices related to electrical characterization.
Author information
Authors and Affiliations
Contributions
UC and ZLP conceived and planned the experiments. NH, MM, DV and MĐ performed plasma treatment of bacteria and liquid chemistry analyses along with the interpretation of those results. NŠ, KS and AJ performed plasma diagnostics and electrical characterization along with interpretation of those results. AJ wrote the original draft, and all co-authors helped with manuscript revision.
Corresponding author
Rights and permissions
About this article
Cite this article
Jurov, A., Škoro, N., Spasić, K. et al. Helium atmospheric pressure plasma jet parameters and their influence on bacteria deactivation in a medium. Eur. Phys. J. D 76, 29 (2022). https://doi.org/10.1140/epjd/s10053-022-00357-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjd/s10053-022-00357-y