Abstract
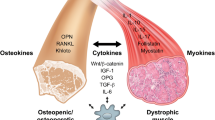
The article reviews the particular qualities of the key signal component of anabolic pathways (serine/threonine kinase Akt1) in the regulation of skeletal muscle functioning, both under normal conditions and in the dystrophic process, including Duchenne’s muscular dystrophy. The current data confirm the participation of the Akt1 signal pathway in the processes of skeletal muscle regeneration, as well as in the mechanism of angiogenesis amplification, which demonstrates the great therapeutic potential of Akt1 as a target for the treatment of a wide range of hereditary and acquired neuromuscular diseases.

Similar content being viewed by others
REFERENCES
Akasaki, Y., Ouchi, N., Izumiya, Y., et al., Glycolytic fast-twitch muscle fiber restoration counters adverse age-related changes in body composition and metabolism, Aging Cell, 2014, vol. 13, pp. 80–91.
Altomare, D.A. and Testa, J.R., Perturbations of the Akt signaling pathway in human cancer, Oncogene, 2005, vol. 24, pp. 7455–7464.
Amoasii, L., Hnia, K., Chicanne, G., et al., Myotubularin and PtdIns3P remodel the sarcoplasmic reticulum in muscle in vivo, J. Cell Sci., 2013, vol. 126, pp. 1806–1819.
Araki, S., Izumiya, Y., Hanatani, S., et al., Akt1-mediated skeletal muscle growth attenuates cardiac dysfunction and remodeling after experimental myocardial infarction, Circ. Heart Fail, 2012, vol. 5, pp. 116–125.
Atochin, D. and Huang, P., Protective role of Akt1-S1177 against stroke, in Adaptation Biology and Medicine: Current Trends, New Delhi: Narosa Press, 2017, pp. 167–172.
Bellacosa, A., Kumar, C.C., Di Cristofano, A., and Testa, J.R., Activation of Akt kinases in cancer: implications for therapeutic targeting, Adv. Cancer Res., 2005, vol. 94, pp. 29–86.
Blaauw, B., The Effects of Akt Overexpression in Normal and Dystrophic Skeletal Muscle, Padova: Univ. Degli Stud. Padova, 2008.
Bodine, S.C., Latres, E., Baumhueter, S., et al., Identification of ubiquitin ligases required for skeletal muscle atrophy, Science, 2001, vol. 294, pp. 1704–1708.
Boppart, M.D., Burkin, D.J., and Kaufman, S.J., Activation of Akt signaling promotes cell growth and survival in α7β1 integrin-mediated alleviation of muscular dystrophy, Biochim. Biophys. Acta, 2011, vol. 1812, no. 4, pp. 439–446.
Brazil, D.P. and Hemmings, B.A., Ten years of protein kinase B signaling: a hard Akt to follow, Trends Biochem. Sci., 2001, vol. 26, pp. 657–664.
Brazil, D.P., Park, J., and Hemmings, B.A., PKB binding proteins. Getting in on the Akt, Cell, 2002, vol. 111, pp. 293–303.
Brodbeck, D., Cron, P., and Hemmings, B.A., A human protein kinase B[gamma] with regulatory phosphorylation sites in the activation loop and in the C-terminal hydrophobic domain, J. Biol. Chem., 1999, vol. 274, pp. 9133–9136.
Brookes, P.S., Yoon, Y., Robotham, J.L., et al., Calcium, ATP, and ROS: a mitochondrial love-hate triangle, Am. J. Physiol. Cell Physiol., 2004, vol. 287, pp. 817–833.
Brugarolas, J., Lei, K., Hurley, R.L., et al., Regulation of mTOR function in response to hypoxia by REDD1 and the TSC1/TSC2 tumor suppressor complex, Genes Dev., 2004, vol. 18, pp. 2893–2904.
Brunet, A., Bonni, A., Zigmond, M.J., et al., Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor, Cell, 1999, vol. 96, pp. 857–868.
Burkin, D.J., Wallace, G.Q., Nichol, K.J., et al., Enhanced expression of the α7β1 integrin reduces muscular dystrophy and restores viability in dystrophic mice, J. Cell Biol., 2001, vol. 152, pp. 1207–1218.
Capetanaki, Y., Bloch, R.J., Kouloumenta, A., et al., Muscle intermediate filaments and their links to membranes and membranous organelles, Exp. Cell Res., 2007, vol. 313, pp. 2063–2076.
Cheung, M. and Testa, J.R., Diverse mechanisms of Akt pathway activation in human malignancy, Curr. Cancer Drug Targ., 2013, vol. 13, pp. 234–244.
Clarke, B.A., Drujan, D., Willis, M.S., et al., The E3 ligase MuRF1 degrades myosin heavy chain protein in dexamethasone-treated skeletal muscle, Cell Metab., 2007, vol. 6, pp. 376–385.
Cohen, S., Brault, J.J., Gygi, S.P., et al., During muscle atrophy, thick, but not thin, filament components are degraded by MuRF1-dependent ubiquitylation, J. Cell Biol., 2009, vol. 185, pp. 1083–1095.
Csibi, A., Cornille, K., Leibovitch, M.P., et al., The translation regulatory subunit eIF3f controls the kinase-dependent mTOR signaling required for muscle differentiation and hypertrophy in mouse, PLoS One, 2010, vol. 5, no. 2, p. 8994.
Deconinck, N., Tinsley, J., De Backer, F., et al., Expression of truncated utrophin leads to major functional improvements in dystrophin-deficient muscles of mice, Nat. Med., 1997, vol. 3, pp. 1216–1221.
Dogra, C., Changotra, H., Wergedal, J.E., and Kumar, A., Regulation of phosphatidylinositol 3-kinase (PI3K)/Akt and nuclear factor-kappa B signaling pathways in dystrophin-deficient skeletal muscle in response to mechanical stretch, J. Cell Physiol., 2006, vol. 208, pp. 575–585.
Dubowitz, V., Sewry, C.A., and Oldfors, A., Muscle Biopsy: A Practical Approach, Amsterdam: Elsevier, 2013.
Durbeej, M. and Campbell, K.P., Muscular dystrophies involving the dystrophin-glycoprotein complex: an overview of current mouse models, Curr. Opin. Genet. Dev., 2002, vol. 12, pp. 349–361.
Ebihara, S., Guibinga, G.H., Gilbert, R., et al., Differential effects of dystrophin and utrophin gene transfer in immunocompetent muscular dystrophy (mdx) mice, Physiol. Genomics, 2000, vol. 3, pp. 133–144.
Egerman, M.A. and Glass, D.J., Signaling pathways controlling skeletal muscle mass, Crit. Rev. Biochem. Mol. Biol., 2014, vol. 49, pp. 59–68.
Ervasti, J.M., Structure and function of the dystrophinglycoprotein complex, in Molecular Mechanisms of Muscular Dystrophies, Winder, S.J., Ed., Boca Raton: CRC Press, 2006, pp. 1–13.
Ervasti, J.M., Ohlendieck, K., Kahl, S.D., et al., Deficiency of a glycoprotein component of the dystrophin complex in dystrophic muscle, Nature, 1990, vol. 345, pp. 315–319.
Fanzani, A., Conraads, V.M., Penna, F., and Martinet, W., Molecular and cellular mechanisms of skeletal muscle atrophy: an update, J. Cachexia Sarcopenia Muscle, 2012, vol. 3, pp. 163–179.
Fayard, E., Tintignac, L.A., Baudry, A., and Hemmings, B.A., Protein kinase B/Akt at a glance, J. Cell Sci., 2005, vol. 118, pp. 5675–5678.
Gomes, M.D., Lecker, S.H., Jagoe, R.T., et al., Atrogin-1, a musclespecific F-box protein highly expressed during muscle atrophy, Proc. Natl. Acad. Sci. U.S.A., 2001, vol. 98, pp. 14440–14445.
Gort, E.H., Groot, A.J., Derks van de Ven, T.L., et al., Hypoxia-inducible factor-1α expression requires PI 3-kinase activity and correlates with Akt1 phosphorylation in invasive breast carcinomas, Oncogene, 2006, vol. 25, pp. 6123–6127.
Gunaratnam, L., Morley, M., Franovic, A., et al., Hypoxia inducible factor activates the transforming growth factor-alpha/epidermal growth factor receptor growth stimulatory pathway in VHL(–/–) renal cell carcinoma cells, J. Biol. Chem., 2003, vol. 278, pp. 44966–44974.
Hanatani, S., Izumiya, Y., Araki, S., et al., Akt1-mediated fast/glycolytic skeletal muscle growth attenuates renal damage in experimental kidney disease, J. Am. Soc. Nephrol., 2014, vol. 25, pp. 2800–2811.
Hers, I., Vincent, E.E., and Tavare, J.M., Akt signalling in health and disease, Cell. Signaling, 2011, vol. 23, pp. 1515–1527.
Heydemann, A. and McNally, E.M., Consequences of disrupting the dystrophin-sarcoglycan complex in cardiac and skeletal myopathy, Trends Cardiovasc. Med., 2007, vol. 17, pp. 55–59.
Hnia, K., Tronchere, H., Tomczak, K.K., et al., Myotubularin controls desmin intermediate filament architecture and mitochondrial dynamics in human and mouse skeletal muscle, J. Clin. Invest., 2011, vol. 121, pp. 70–85.
Hodges, B.L., Hayashi, Y.K., Nonaka, I., et al., Altered expression of the α7β1 integrin in human and murine muscular dystrophies, J. Cell Sci., 1997, vol. 110, pp. 2873–2881.
Hoffman, E.P., Brown, R.H., and Kunkel, L.M., Dystrophin: the protein product of the Duchenne muscular dystrophy locus, Cell, 1987, vol. 51, pp. 919–928.
Hollander, M.C., Maier, C.R., Hobbs, E.A., et al., Akt1 deletion prevents lung tumorigenesis by mutant K-ras, Oncogene, 2011, vol. 30, pp. 1812–1821.
Izumiya, Y., Hopkins, T., Morris, C., et al., Fast/glycolytic muscle fiber growth reduces fat mass and improves metabolic parameters in obese mice, Cell Metab., 2008, vol. 7, pp. 159–172.
Krag, T.O., Bogdanovich, S., Jensen, C.J., et al., Heregulin ameliorates the dystrophic phenotype in mdx mice, Proc. Natl. Acad. Sci. U.S.A., 2004, vol. 101, pp. 13856–13860.
Lai, K.M., Gonzalez, M., Poueymirou, W.T., et al., Conditional activation of Akt in adult skeletal muscle induces rapid hypertrophy, Mol. Cell Biol., 2004, vol. 24, pp. 9295–9304.
Langenbach, K.J. and Rando, T.A., Inhibition of dystroglycan binding to laminin disrupts the PI3K/Akt pathway and survival signaling in muscle cells, Muscle Nerve, 2002, vol. 26, pp. 644–653.
Le Cras, T.D., Korfhagen, T.R., Davidson, C., et al., Inhibition of PI3K by PX-866 prevents transforming growth factoralpha-induced pulmonary fibrosis, Am. J. Pathol., 2010, vol. 176, pp. 679–686.
Mammucari, C., Milan, G., Romanello, V., et al., FoxO3 controls autophagy in skeletal muscle in vivo, Cell Metab., 2007, vol. 6, pp. 458–471.
Michael, A., Haq, S., Chen, X., et al., Glycogen synthase kinase-3β regulates growth, calcium homeostasis, and diastolic function in the heart, J. Biol. Chem., 2004, vol. 279, pp. 21383–21393.
Nguyen, H.H., Jayasinha, V., Xia, B., et al., Overexpression of the cytotoxic T cell GalNAc transferase in skeletal muscle inhibits muscular dystrophy in mdx mice, Proc. Natl. Acad. Sci. U.S.A., 2002, vol. 99, pp. 5616–5621.
Ohlendieck, K. and Campbell, K.P., Dystrophin: the protein product of the Duchenne muscular dystrophy locus, J. Cell Biol., 1991, vol. 115, pp. 1685–1694.
Pallafacchina, G., Calabria, E., Serrano, A.L., et al., A protein kinase B-dependent and rapamycin-sensitive pathway controls skeletal muscle growth but not fiber type specification, Proc. Natl. Acad. Sci. U.S.A., 2002, vol. 99, pp. 9213–9218.
Persad, S., Attwell, S., Gray, V., et al., Regulation of protein kinase B/Akt-serine 473 phosphorylation by integrin-linked kinase, J. Biol. Chem., 2001, vol. 276, pp. 27462–27469.
Peter, A.K. and Crosbie, R.H., Hypertrophic response of Duchenne and limb-girdle muscular dystrophies is associated with activation of Akt pathway, Exp. Cell Res., 2006, vol. 312, pp. 2580–2591.
Pistollato, F., Rampazzo, E., Abbadi, S., et al., Molecular mechanisms of HIF-1α modulation induced by oxygen tension and BMP2 in glioblastoma derived cells, PLoS One, 2009, vol. 4, p. e6206.
Pong, T., Scherrer-Crosbie, M., Atochin, D.N., et al., Phosphomimetic modulation of eNOS improves myocardial reperfusion and mimics cardiac postconditioning in mice, PLoS One, 2014, vol. 9, no. 1, p. e85946.
Rodgers, J.T., King, K.Y., Brett, J.O., et al., mTORC1 controls the adaptive transition of quiescent stem cells from G0 to G (Alert), Nature, 2014, vol. 510, pp. 393–396.
Ruegg, M.A. and Glass, D.J., Molecular mechanisms and treatment options for muscle wasting diseases, Ann. Rev. Pharmacol. Toxicol., 2011, vol. 51, pp. 373–395.
Sandri, M., Sandri, C., Gilbert, A., et al., FoxO transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy, Cell, 2004, vol. 117, pp. 399–412.
Schiaffino, S., Dyar, K.A., Ciciliot, S., et al., Mechanisms regulating skeletal muscle growth and atrophy, FEBS J., 2013, vol. 280, pp. 4294–4314.
Squire, S., Raymackers, J.M., Vandebrouck, C., et al., Prevention of pathology in mdx mice by expression of utrophin: analysis using an inducible transgenic expression system, Hum. Mol. Genet., 2002, vol. 11, pp. 3333–3344.
Stitt, T.N., Drujan, D., Clarke, B.A., et al., The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FoxO transcription factors, Mol. Cell, 2004, vol. 14, pp. 395–403.
Stupka, N., Plant, D.R., Schertzer, J.D., et al., Activated calcineurin ameliorates contraction-induced injury to skeletal muscles of mdx dystrophic mice, J. Physiol., 2006, vol. 575, pp. 645–656.
Takahashi, A., Kureishi, Y., Yang, J., et al., Myogenic Akt signaling regulates blood vessel recruitment during myofiber growth, Mol. Cell Biol., 2002, vol. 22, pp. 4803–4814.
Takano, K., Watanabe-Takano, H., Suetsugu, S., et al., Nebulin and N-WASP cooperate to cause IGF-1-induced sarcomeric actin filament formation, Science, 2010, vol. 330, pp. 1536–1540.
Tinsley, J.M., Potter, A.C., Phelps, S.R., et al., Amelioration of the dystrophic phenotype of mdx mice using a truncated utrophin transgene, Nature, 1996, vol. 384, pp. 349–353.
Tinsley, J., Deconinck, N., Risher, R., et al., Expression of full-length utrophin prevents muscular dystrophy in mdx mice, Nat. Med., 1998, vol. 4, pp. 1441–1444.
Tintignac, L.A., Lagirand, J., Batonnet, S., et al., Degradation of MyoD mediated by the SCF (MAFbx) ubiquitin ligase, J. Biol. Chem., 2005, vol. 280, no. 4, pp. 2847–2856.
Troussard, A.A., Mawji, N.M., Ong, C., et al., Conditional knock-out of integrin-linked kinase (ILK) demonstrates an essential role in PKB/Akt activation, J. Biol. Chem., 2003, vol. 278, pp. 22374–22378.
Whitmore, C. and Morgan, J., What do mouse models of muscular dystrophy tell us about the DAPC and its components? Int. J. Exp. Pathol., 2014, vol. 95, no. 6, pp. 365–377.
Wilson, E.M. and Rotwein, P., Selective control of skeletal muscle differentiation by Akt1, J. Biol. Chem., 2007, vol. 282, pp. 5106–5110.
Woolstenhulme, M.T., Conlee, R.K., Drummond, M.J., et al., Temporal response of desmin and dystrophin proteins to progressive resistance exercise in human skeletal muscle, J. Appl. Physiol., 2006, vol. 100, pp. 1876–1882.
Wu, C.L., Satomi, Y., and Walsh, K., RNA-seq and metabolomic analyses of Akt1-mediated muscle growth reveals regulation of regenerative pathways and changes in the muscle secretome, BMC Genomics, 2017, vol. 18, no. 1, p. 181.
Yoshida, M. and Ozawa, E., Glycoprotein complex anchoring dystrophin to sarcolemma, J. Biochem., 1990, vol. 108, pp. 748–752.
Zhang, P., Liang, X., Shan, T., et al., mTOR is necessary for proper satellite cell activity and skeletal muscle regeneration, Biochem. Biophys. Res. Commun., 2015, vol. 463, pp. 102–108.
ACKNOWLEDGMENTS
This study was supported by the Russian Science Foundation (grant no. 17-15-01111).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Сonflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. This article does not contain any studies involving animals performed by any of the authors.
Additional information
Translated by M. Novikova
Rights and permissions
About this article
Cite this article
Sukhorukov, V.S., Baranich, T.I., Atochin, D.N. et al. The Adaptation Role of Serine/Threonine Kinase Akt1 in Anabolism of Muscular Tissue. Biol Bull Rev 8, 489–496 (2018). https://doi.org/10.1134/S2079086418060105
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2079086418060105