Abstract
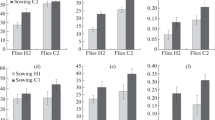
Adaptation to stress factors is frequently accompanied by negative side effects that are manifested in a decreased adaptation to an environment in which this stress factor is absent. This can cause ecological specialization of forms adapted to the stress conditions to the point of their separation into a new species (ecological speciation). On the other hand, the existence of eurybiont species with a wide spectrum of ecological tolerance indicates that adaptation to marginal conditions can presumably occur without negative side effects or even be accompanied by positive effects (which leads to expansion of the ecological niche). There are few experimental confirmations of the reality of such an evolutionary scenario. During an evolutionary experiment on the Drosophila melanogaster, experimental fly lines successfully adapted to life on stressful feed substrates with an increased NaCl content for 20 generations. This was manifested in an increase in the total amount of descendants produced by parents from adapted lines on stressful substrate for a fixed time as compared with the control (unadapted) line, as well as in a reduction of the larval development delay caused by high NaCl concentrations. Not only did the adaptation to a standard (favorable) feed medium in lines adapted to salt not decrease, but it even increased. Thus, adaptation to unfavorable medium actually resulted in trophic niche expansion. Along with similar results previously obtained during a study of the D. melanogaster adaptation to a depleted feed medium based on starch, these data indicate that an adaptation to marginal conditions that is accompanied by positive side effects and does not lead to constriction but to expansion of the trophic niche can be a common phenomenon in such eurybiont species as D. melanogaster, which probably largely explains their eurybiontity. The small number of experimentally confirmed examples of such a scenario can be partly explained be the small number of attempted searches for them. Adaptive changes of symbiotic microbiota, which are efficiently transmitted in the Drosophila from parents to descendants when the descendants eat the same substrate as the parents, can be one possible mechanism of “wide-profile” adaptation output during the development of the stressful substrate. Thus, a growth of the number of symbiotic Lactobacilli in the Drosophila intestine can promote acceleration of larvae growth, extension of imago life, and an increased efficiency of the used feed substrate. Further studies are required to detect the specific mechanisms responsible for the changes in adaptability found during the evolutionary experiment.
Similar content being viewed by others
References
Aigner, P.A., The evolution of specialized floral phenotypes in a fine-grained pollination environment, in Plant-Pollinator Interactions: From Specialization to Generalization, Waser, N.M. and Ollerton, I., Eds., Chicago: Univ. Chicago Press, 2006, pp. 23–46.
Arbuthnott, D. and Rundle, H.D., Misalignment of natural and sexual selection among divergently adapted Drosophila melanogaster populations, Anim. Behav., 2014, vol. 87, pp. 45–51.
Blum, J.E., Fischer, C.N., Miles, J., and Handelsman, J., Frequent replenishment sustains the beneficial microbiome of Drosophila melanogaster, mBio, 2013, vol. 4, p. e00860.
Brummel, T., Ching, A., Seroude, L., Simon, A.F., and Benzer, S., Drosophila lifespan enhancement by exogenous bacteria, Proc. Natl. Acad. Sci. U.S.A., 2004, vol. 101, pp. 12974–12979.
Collins, N., Population ecology of Ephydra cinerea Jones (Diptera: Ephydridae), the only benthic metazoan of the Great Salt Lake, USA, Hydrobiologia, 1980, vol. 68, pp. 99–112.
Condon, C., Acharya, A., Adrian, G., Hurliman, A., Malekooti, D., Nguyen, P., Zelic, M., and Angilletta, M.J., Jr., Indirect selection of thermal tolerance during experimental evolution of Drosophila melanogaster, Ecol. Evol., 2015, vol. 5, pp. 1873–1880.
Cook, L.M. and Saccheri, I.J., The peppered moth and industrial melanism: evolution of a natural selection case study, Heredity, 2013, vol. 110, pp. 207–212.
Erkosar, B., Storelli, G., Defaye, A., and Leulier, F., Hostintestinal microbiota mutualism: “learning on the fly,” Cell Host Microbe., 2013, vol. 13, pp. 8–14.
Falconer, D.S., Introduction to Quantitative Genetics, London: Oliver and Boyd, 1960.
Faria, V.G., Martins, N.E., Paulo, T., Teixeira, L., Sucena, E., and Magalhaes, S., Evolution of Drosophila resistance against different pathogens and infection routes entails no detectable maintenance costs, Evolution, 2015, vol. 69, pp. 2799–2809.
Fellowes, M.D.E., Kraaijeveld, A.R., and Godfray, H.C.J., Trade-off associated with selection for increased ability to resist parasitoid attack in Drosophila melanogaster, Proc. R. Soc. London, Ser. B, 1998, vol. 265, pp. 1553–1558.
Herbst, D.B., Gradients of salinity stress, environmental stability and water chemistry as a templet for defining habitat types and physiological strategies in inland salt waters, Hydrobiologia, 2001, vol. 466, pp. 209–219.
Hoffmann, A.A., Hallas, R., Anderson, A.R., and Telonis-Scott, M., Evidence for a robust sex-specific trade-off between cold resistance and starvation resistance in Drosophila melanogaster, J. Evol. Biol., 2005, vol. 18, pp. 804–810.
Kalajdzic, P., Kenig, B., and Andjelkovic, M., Drosophila subobscura flies adapted to low lead concentration carry no fitness cost, Environ. Pollut., 2015, vol. 204, pp. 90–98.
Kliot, A. and Ghanim, M., Fitness costs associated with insecticide resistance, Pest Manage. Sci., 2012, vol. 68, pp. 1431–1437.
Kneitel, J.M. and Chase, J.M., Trade-offs in community ecology: linking spatial scales and species coexistence, Ecol. Lett., 2004, vol. 7, pp. 69–80.
Korol, A., Rashkovetsky, E., Iliadi, K., Michalak, P., Ronin, Y., and Nevo, E., Nonrandom mating in Drosophila melanogaster laboratory populations derived from closely adjacent ecologically contrasting slopes at ‘Evolution canyon’, Proc. Natl. Acad. Sci. U.S.A., 2004, vol. 97, pp. 12637–12642.
Kreslavskii, A.G., Sympatric speciation in animals: disruptive selection or ecological segregation, Zh. Obshch. Biol., 1994, vol. 55, nos. 4–5, pp. 404–419.
Linnen, C.R., Kingsley, E.P., Jensen, J.D., and Hoekstra, H.E., On the origin and spread of an adaptive allele in deer mice, Science, 2009, vol. 325, pp. 1095–1098.
Long, T.A.F., Rowe, L., and Agrawal, A.F., The effects of selective history and environmental heterogeneity on inbreeding depression in experimental populations of Drosophila melanogaster, Am. Nat., 2013, vol. 181, pp. 532–544.
Markov, A.V., Ivnitsky, S.B., Kornilova, M.B., Naimark, E.B., Shirokova, N.G., and Perfilieva, K.S., Maternal effect obscures adaptation to adverse environments and hinders divergence in Drosophila melanogaster, Biol. Bull. Rev., 2016, vol. 6, no. 5, pp. 429–435.
Nagai, J., Eisen, E., Emsly, J., and McAllister, A., Selection for nursing ability and adult weight in mice, Genetics, 1978, vol. 88, pp. 761–780.
Pekkala, N., Kotiaho, J.S., and Puurtinen, M., Laboratory relationships between adult lifetime reproductive success and fitness surrogates in a Drosophila littoralis population, PLoS One, 2011, vol. 6, p. e24560.
Rice, W.R. and Salt, G.W., The evolution of reproductive isolation as a correlated character under sympatric conditions: experimental evidence, Evolution, 1990, vol. 44, pp. 1140–1152.
Robinson, B.W. and Wilson, D.S., Optimal foraging, specialization, and a solution to Liem’s paradox, Am. Nat., 1998, vol. 151, pp. 223–235.
Savinov, A.B., Autocenosis and democenosis as symbiotic systems and biological notions, Zh. Obshch. Biol., 2012, vol. 73, no. 4, pp. 284–301.
Sharon, G., Segal, D., Ringo, J.M., Hefetz, A., Zilber-Rosenberg, I., and Rosenberg, E., Commensal bacteria play a role in mating preference of Drosophila melanogaster, Proc. Nat. Acad. Sci. U.S.A., 2010, vol. 107, pp. 20051–20056.
Shin, S.C., Kim, S.H., You, H., Kim, B., Kim, A.C., Lee, K.A., Yoon, J.H., Ryu, J.H., and Lee, W.J. Drosophila microbiome modulates host developmental and metabolic homeostasis via insulin signaling, Science, 2011, vol. 334, pp. 670–674.
Shirley, M.D.F. and Sibly, R.M., Genetic basis of a between-environment trade-off involving resistance to cadmium in Drosophila melanogaster, Evolution, 1999, vol. 53, pp. 826–836.
Stergiopoulos, K., Cabrero, P., Davies, S.A., and Dow, J.A., Salty dog, an SLC5 symporter, modulates Drosophila response to salt stress, Physiol. Genomics, 2009, vol. 37, pp. 1–11.
Storelli, G., Defaye, A., Erkosar, B., Hols, P., Royet, J., and Leulier, F., Lactobacillus plantarum promotes Drosophila systemic growth by modulating hormonal signals through TOR-dependent nutrient sensing, Cell Metab., 2011, vol. 14, pp. 403–414.
Te Velde, J.H., Molthoff, C.F.M., and Scharloo, W., The function of anal papillae in salt adaptation of Drosophila melanogaster larvae, J. Evol. Biol., 1988, vol. 2, pp. 139–153.
Tobler, R., Hermisson, J., and Schlotterer, C., Parallel trait adaptation across opposing thermal environments in experimental Drosophila melanogaster populations, Evolution, 2015, vol. 69, pp. 1745–1759.
Ungar, P.S., Grine, F.E., and Teaford, M.F., Dental microwear and diet of the Plio-Pleistocene hominin Paranthropus boisei, PLoS One, 2008, vol. 3, pp. e2044.
Vijendravarma, R.K., Kraaijeveld, A.R., and Godfray, H.C., Experimental evolution shows Drosophila melanogaster resistance to a microsporidian pathogen has fitness costs, Evolution, 2009, vol. 63, pp. 104–114.
Waddington, C.H., Canalization of development and genetic assimilation of acquired characters, Nature, 1959, vol. 183, pp. 1654–1655.
Wong, C.N., Ng, P., and Douglas, A.E., Low-diversity bacterial community in the gut of the fruitfly Drosophila melanogaster, Environ. Microbiol., 2011, vol. 13, pp. 1889–1900.
Wu, C.I., Hollocher, H., Begun, D.J., Aquadro, C.F., Xu, Y., and Wu, M.L., Sexual isolation in Drosophila melanogaster: a possible case of incipient speciation, Proc. Natl. Acad. Sci. U.S.A., 1995, vol. 92, pp. 2519–2523.
Zilber-Rosenberg, I. and Rosenberg, E., Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution, FEMS Microbiol. Rev., 2008, vol. 32, pp. 723–735.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.S. Dmitrieva, S.B. Ivnitsky, A.V. Markov, 2016, published in Zhurnal Obshchei Biologii, 2016, Vol. 77, No. 4, pp. 249–261.
Rights and permissions
About this article
Cite this article
Dmitrieva, A.S., Ivnitsky, S.B. & Markov, A.V. Adaptation of Drosophila melanogaster to unfavorable feed substrate is accompanied by expansion of trophic niche. Biol Bull Rev 7, 369–379 (2017). https://doi.org/10.1134/S2079086417050024
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2079086417050024