Abstract
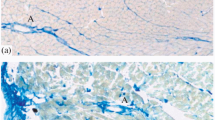
Cellular changes in different parts of the heart of Wistar rats at the age of 18–23 months are studied using histological methods of staining with toluidine blue, hematoxylin-eosin, and immunohistochemical reactions for PGP 9.5 protein, tyrosine hydroxylase (TH), and Iba-1 protein. Focal inflammatory infiltrates are found in the connective tissue of the heart base, near which PGP 9.5+ and TH+ plexuses consisting of parasympathetic and sympathetic nerve fibers are detected. In the area of the valvular heart apparatus and at the border between the fibrous ring and myocardium of the right atrium, the following pathological changes in nerve structures are observed: degeneration of nerve fibers and granular destruction of varicose axons of the terminal plexus. A close relationship is established between the axons of the terminal neural network and cells of inflammatory infiltrates and blood capillaries. The features of the localization of neurocellular inflammatory complexes consisting of nerve fibers, blood capillaries, and cells participating in the local inflammatory process (mast cells, macrophages, fibroblasts, and plasma cells) are described in various parts of the myocardium in old animals. The chronic nature of neurogenic inflammation is determined for the heart during aging.







Similar content being viewed by others
REFERENCES
Korneva, E.A., Vzaimodeistvie nervnoi i immunnoi sistem. Molekulyarno-kletochnye aspekty (Interaction between Nervous and Immune Systems. Molecular and Cellular Aspects). St. Petersburg: Nauka, 2012.
Kucher, A.N., Neurogenic inflammation: biochemical markers, genetic control of the disease, Byull. Sib. Med., 2020, vol. 19, no. 2, pp. 171–181.
Petrishchev, N.N., Disfunktsiya endoteliya. Patogeneticheskoe znachenie i metody korrektsii (Endothelial Dysfunction. Pathogenetic Significance and Methods of Correction), St. Petersburg: SPb IITs VMA, 2007.
Khavinson, V.Kh., Tarnovskaya, S.I., Linkova, N.S., et al., Epigenetic aspects of peptidergic regulation of vascular endothelial cell proliferation in aging, Adv. Gerontol., 2015, vol. 5, no. 4, pp. 219–224.
Chereshnev, V.A. and Gusev, E.Yu., Immunology of inflammation: role of cytokines, Med. Immunol., 2001, vol. 3, no. 3, pp. 361–368.
Chumasov, E.I., Maistrenko, N.A., Korzhevskii, D.E., et al., Peculiarities of neuroimmune intercellular relationships in the pancreas in chronic pancreatitis, Vestn. Ross. Voen.-Med. Akad., 2013, vol. 3, no. 43, pp. 1–5.
Shabrov, A.V., Apresyan, A.G., Dobkes, A.L., et al., Role and methods of assessment of endothelial dysfunction in practical medicine, Med. Akad. Zh., 2017, vol. 17, no. 1, pp. 7–23.
Arizono, N., Matsuda, S., Hattori, T., et al., Anatomical variation in mast cell nerve associations in the rat small intestine, heart, lung, and skin. Similarities of distances between neural processes and mast cells, eosinophils, or plasma cells in the jejunal Lamina propria, Lab. Invest., 1990, vol. 62, no. 5, pp. 626–634.
Chumasov, E.I., Petrova, E.S., Kolos, E.A., and Korzhevskii, D.E., Investigation of nerve apparatus and mast cells in the heart of old rats, Adv. Gerontol., 2021, vol. 11, no. 1, pp. 29–36.
Freeman, J.G., Ryan, J.J., Shelburne, C.P., et al., Catecholamines in murine bone marrow derived mast cells, J. Neuroimmunol., 2001, vol. 119, no. 2, pp. 231–238.
Järvikallio, A., Harvima, I.T., and Naukkarinen, A., Mast cells, nerves and neuropeptides in atopic dermatitis and nummular eczema, Arch. Dermatol. Res., 2003, vol. 295, no. 1, pp. 2–7.
Kong, P., Christia, P., and Frangogiannis, N.G., The pathogenesis of cardiac fibrosis, Cell Mol. Life Sci., 2014, vol. 71, no. 4, pp. 549–574. https://doi.org/10.1007/s00018-013-1349-6
Levick, S.P., Murray, D.B., Janicki, J.S., and Brower, G.L., Sympathetic nervous system modulation of inflammation and remodeling in the hypertensive heart, Hypertension, 2010, vol. 55, no. 2, pp. 270–276.
Luger, T.A. and Lotti, T., Neuropeptides: role in inflammatory skin diseases, J. Eur. Acad. Dermatol. Venerol., 1998, vol. 10, no. 3, pp. 207–211.
Matt, S.M. and Gaskill, P.J., Where is dopamine and how do immune cells see it?: dopamine-mediated immune cell function in health and disease, J. Neuroimmune Pharmacol., 2020, vol. 15, no. 1, pp. 114–164.
Okamura, Y., Mishima, S., Kashiwakura, J.I., et al., The dual regulation of substance P-mediated inflammation via human synovial mast cells in rheumatoid arthritis, Allergol. Int., 2017, vol. 66S. https://doi.org/10.1016/j.alit.2017.03.002
Scholtzer, T., Armstrong, C.A., Bunnett, N.W., et al., Neuropeptides in the skin: interactions between the neuroendocrine and the skin immune systems, Exp. Dermatol., 1998, vol. 7, nos. 2–3, pp. 81–96.
Sousa-Valente, J. and Brain, S.D., A historical perspective on the role of sensory nerves in neurogenic inflammation, Semin. Immunopathol., 2018, vol. 40, pp. 229–236.
Funding
The study was performed under the state assignment of the Institute of Experimental Medicine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interests.
Statement on the welfare of animals. Animals were treated in accordance with the international guidelines of the European Society on the Welfare of Laboratory Animals and Rules of Research with Experimental Animals (annex to the order of the Ministry of Health of USSR no. 755, August 12, 1977). The research was approved by Local Ethics Committee of the Institute of Experimental Medicine, St. Petersburg (protocol no. 3, November 30, 2017).
Additional information
Translated by E. Sherstyuk
Rights and permissions
About this article
Cite this article
Chumasov, E.I., Petrova, E.S. & Korzhevskii, D.E. Morphological Signs of Neurogenic Inflammation in the Heart of Rats during Aging. Adv Gerontol 12, 113–121 (2022). https://doi.org/10.1134/S2079057022020059
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2079057022020059