Abstract
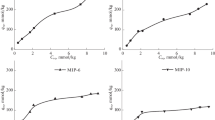
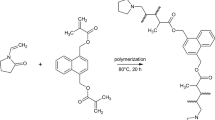
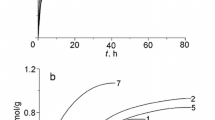
A synthesis method of granulated polymeric sorbents based on 2-hydroxyethylmethacrylate and ethylene glycol dimethacrylate that possess affinity to glucose sorption sites formed by boric acid molecules has been developed. The participation of boric acid as an additional crosslinking agent and the effect of the amount of the introduced ligand on the surface morphology of the granules and physicochemical properties of the polymeric sorbents have been determined. It has been found that this modification of the polymer matrix promotes an increase in the affinity of the sorption surface and its adsorption capacity to bind glucose in a dynamic mode, as well as high selectivity in comparison with the sorption of its structural analogue, fructose. This can further be used in the development of a method of isolation of glucose from a multicomponent solution and a sorption method of separation of sugars.







Similar content being viewed by others
REFERENCES
Ning, H., et al., J. Chromatogr. A, 2022, vol. 1671, p. 462994.
Hasanah, A.N., et al., J. Chem., 2020, vol. 2020, p. 7282415.
Tu, X., et al., Talanta, 2021, vol. 226, p. 122142.
Alipour, S., et al., J. Mater. Process. Technol., 2021, vol. 12, p. 2298.
Eivazzadeh-Keihan, R., et al., TrAC, Trends Anal. Chem., 2021, vol. 141, p. 116291.
Xu, R., et al., J. Chromatogr. A, 2021, vol. 1635, p. 461707.
Hu, Z., Wang, X., and Chen, X., Anal. Chim. Acta, 2020, vol. 1112, p. 16.
Gheybalizadeh, H. and Hejazi, P., React. Funct. Polym., 2022, vol. 171, p. 105152.
Amorim, M.S., Sales, M.G.F., and Frasco, M.F., Biosens. Bioelectron., 2022, vol. 10, p. 100131.
Li, M., et al., Kexue Tongbao/Chin. Sci. Bull., 2019, vol. 64, no. 13, p. 1321.
Askin, S., Kizil, S., and Bulbul Sonmez, H., React. Funct. Polym., 2021, vol. 167, p. 105002.
Bayraktaroglu, S., Kizil, S., and Bulbul Sonmez, H., J. Environ. Chem. Eng., 2021, vol. 9, no. 5, p. 106002.
Sulejmanović, J., et al., Chemosphere, 2022, vol. 296, p. 133971.
Wang, H.H., Shyr, T.W., and Hu, M.S., J. Appl. Polym. Sci., 1999, vol. 74, no. 13, p. 3046.
Prosanov, I.Y., et al., Mater. Today Commun., 2018, vol. 14, p. 77.
Hong, X., et al., J. Appl. Polym. Sci., 2021, vol. 138, no. 47.
Vedelago, J., et al., Sci. Rep., 2021, vol. 11, no. 1, p. 1.
Belcher, R., Tully, G.W., and Svehla, G., Anal. Chim. Acta, 1970, vol. 50, no. 2, p. 261.
Dawber, J.G. and Matusin, D.H., J. Chem. Soc., Faraday Trans., 1982, vol. 78, no. 8, p. 2521.
Crabb, W.D. and Mitchinson, C., Trends Biotechnol., 1997, vol. 15, no. 9, p. 349.
Kuznetsov, B.N., et al., Catal. Ind., 2018, vol. 10, no. 2, p. 142.
Dupont, H., et al., Macromolecules, 2021, vol. 54, no. 11, p. 4945.
Yang, Y., et al., Front. Pharmacol., 2017, vol. 8, p. 287.
Willaman, J.J. and Davison, F.R., J. Agric. Res., 1924, vol. 28, no. 5, p. 479.
Umpleby, R.J., et al., J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2004, vol. 804, no. 1, p. 141.
Hinz, C., Geoderma, 2001, vol. 99, nos. 3–4, p. 225.
Freundlich, H., Z. Phys. Chem., 1907, vol. 57U, no. 1, p. 385.
Ebadi, A., Soltan Mohammadzadeh, J.S., and Khudiev, A., Adsorption, 2009, vol. 15, no. 1, p. 65.
Ebadi, A., Soltan Mohammadzadeh, J.S., and Khudiev, A., Chem. Eng. Technol., 2007, vol. 30, no. 12, p. 1666.
Krishna Kumar, A.S., Jiang, S.J., and Tseng, W.L., J. Mater. Chem. A, 2015, vol. 3, no. 13, p. 7044.
Malarvizhi, R. and Sulochana, N., J. Environ. Prot. Sci., 2008, vol. 2, p. 40.
Liu, Y., Colloids Surf., A, 2006, vol. 274, nos. 1–3, p. 34.
Giles, C.H., Smith, D., and Huitson, A., J. Colloid Interface Sci., 1974, vol. 47, no. 3, p. 755.
Boyd, G.E., Adamson, A.W., and Myers, L.S., J. Am. Chem. Soc., 1947, vol. 69, no. 11, p. 2836.
Lagergreen, S., Z. Chem. Ind. Kolloide, 1907, vol. 2, no. 1, p. 15.
Funding
This work was performed in accordance with a state task, topic no. AAAA-A20-120022090044-2.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interest.
Additional information
Translated by E. Boltukhina
Rights and permissions
About this article
Cite this article
Garkushina, I.S., Panyuta, A.S., Morozova, P.Y. et al. Selective Sorbents Based on 2-Hydroxyethylmethacrylate and Ethylene Glycol Dimethacrylate Modified with Boric Acid as High-Affinity Ligands for Glucose. Prot Met Phys Chem Surf 59, 843–853 (2023). https://doi.org/10.1134/S2070205123701022
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205123701022