Abstract
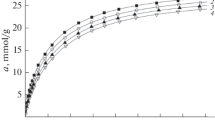
A microporous carbon adsorbent labeled as LCN was prepared by thermochemical synthesis from lignocellulose, and methane adsorption on it was studied within the temperature range from 303 to 333 K and at pressures up to 40 MPa. The total pore volume was determined to be 0.68 cm3/g. The maximum values of methane adsorption of ~14.5 mmol/g (23.2 wt %) were obtained at the temperature of 303 K and the pressure of 40 MPa. The calculated differential molar isosteric heat of methane adsorption is, on average, ~12–13 kJ/mol. Methane adsorption on LCN was calculated with the use of the Dubinin–Radushkevich and Dubinin–Astakhov equations, as well as the linearity of adsorption isosteres. It was shown that the Dubinin–Radushkevich and fundamental regularities of the theory of volume filling of micropores provide the best description of experimental data using only the standard structural and energy characteristics of the adsorbent.








Similar content being viewed by others
REFERENCES
Sychev, V.V., Vasserman, A.A., Zagoruchenko, V.A., Kozlov, A.D., Spiridonov, G.A., and Tsymarnyi, V.A., Termodinamicheskie svoistva metana (Thermodynamic Properties of Methane), Moscow: Izd. Standartov, 1979.
Vasil'ev, Yu.N., Gritsenko, A.I., and Chirikov, K.Yu., Gazozapravka transporta (Gas Filling of Transport), Moscow: Nedra, 1995.
Cleaver, Ph., Johnson, M., and Ho, B., J. Hazard. Mater., 2007, vol. 140, p. 429.
Maiorets, M. and Simonov, K., Szhizhennyi gaz - budushchee mirovoi energetiki (Liquefied Gas is Future of the World Power Engineering), Moscow: Al’pina Pabl., 2013.
Tsivadze, A.Yu., Aksyutin, O.E., Ishkov, A.G., Men’shchikov, I.E., Fomkin, A.A., Shkolin, A.V., Khozina, E.V., and Grachev, V.A., Usp. Khim., 2018, vol. 87, no. 10, p. 950.
Shkolin, A.V., Fomkin, A.A., Tsivadze, A.Yu., Anuchin, K.M., Men’shchikov, I.E., and Pulin, A.L., Prot. Met. Phys. Chem. Surf., 2016, vol. 52, p. 955.
Goetz, V., Pupier, O., and Guillot, A., Adsorption, 2006, no. 12, p. 55.
Strizhenov, E.M., Fomkin, A.A., Zherdev, A.A., and Pribylov, A.A., Prot. Met. Phys. Chem. Surf., 2012, vol. 48, no. 6, p. 614.
Rios, R.B., Silva, F.W.M., Torres, A.E.B., Azevedo, D.C.S., et al., Adsorption, 2009, vol. 15, no. 3, p. 271.
Kel’tsev, N.V., Osnovy adsorbtsionnoi tekhniki (Foundations of Adsorption Technique), Moscow: Khimiya, 1976.
Handbook of Air Pollution Technology, Calvert, S. and Englund, H.M., Eds., New York: Wiley, 1984.
Shumyatskii, Yu.I., Promyshlennye adsorbtsionnye protsessy (Industrial Adsorptive Processes), Moscow: Koloss, 2009.
Dubinin, M.M., Adsorbtsiya i poristost' (Adsorption and Porosity), Moscow: Military Academy of Chemical Defense Named after Marshal of the USSR S.K. Timoshenko, 1972.
Fenelonov, V.B., Poristyi uglerod (Porous Carbon), Novosibirsk: Boreskov Institute of Catalysis Siberian Branch Russ. Acad. Sci., 1995.
Mukhin, V.M., Tarasov, A.V., and Klushin, V.N., Aktivnye ugli Rossii (Active Carbons of Russia), Moscow: Metallurgiya, 2000.
Men'shchikov, I.E., Fomkin, A.A., Shkolin, A.V., Yakovlev, V.Yu., and Khozina, E.V., Izv. Akad. Nauk, Ser. Khim., 2018, no. 10, p. 1814.
Greg, S.J. and Sing, K.S.W., Adsorption, Surface Area and Porosity, London, New York: Academic Press, 1982.
Pribylov, A.A., Kalashnikov, S.M., and Serpinskii, V.V., Izv. Akad. Nauk SSSR, Ser. Khim., 1990, no. 6, p. 1233.
Bakaev, V.A., Izv. Akad. Nauk SSSR, Ser. Khim., 1971, no. 2, p. 2648.
Fomkin, A.A., Adsorption, 2005, vol. 11, p. 425.
Shkolin, A.V. and Fomkin, A.A., Colloid J., 2009, vol. 71, no. 1, p. 119.
Tolmachev, A.M., Anuchin, K.M., Kryuchenkova, N.G., and Fomkin, A.A., Prot. Met. Phys. Chem. Surf., 2011, vol. 47, no. 2, p. 150.
Reid, R., Prausnitz, J., and Sherwood, T., The Properties of Gases and Liquids, New York: McGraw-Hill, 1977.
Tolmachev, A.M., Kuznetsova, T.A., and Godovikov, I.A., Prot. Met. Phys. Chem. Surf., 2011, vol. 47, no. 3, p. 281.
Feng-Chin Wua, Pin-Hsueh Wub, Ru-Ling Tseng, and Ruey-Shin Juang, J. Taiwan Inst. Chem. Eng., 2014, vol. 45, p.1757.
Dubinin, M.M. and Kadlets, O., Izv. Akad. Nauk SSSR, Ser. Khim., 1984, p. 496.
Tsivadze, A.Yu. and Fomkin, A.A., in Fizicheskaya khimiya adsorbtsionnykh yavlenii (Physical Chemistry of Adsorption Phenomena), Moscow: Granitsa, 2011.
Fomenkov P.E., Gumerov M.R., Tolmachev A.M., Prot. Met. Phys. Chem. Surf., 2018, vol. 54, no. 4, p. 565.
Kuznetsova, T.A., Godovikov, I.A., and Tolmachev, A.M., Prot. Met. Phys. Chem. Surf., 2012, vol. 48, no. 4, p. 424.
Kuznetsova, T.A., Tolmachev, A.M., Kryuchenkova, N.G., Firsov, D.A., and Fomkin, A.A., Prot. Met. Phys. Chem. Surf., 2013, vol. 49, no. 4, p. 367.
Men’shchikov, I.E., Fomkin, A.A., Arabei, A.B., Shkolin, A.V., and Strizhenov, E.M., Prot. Met. Phys. Chem. Surf., 2016, vol. 52, no. 4, p. 575.
Chkhaidze, E.V., Fomkin, A.A., Serpinskii, V.V., Tsitsishvili, G.V., and Dubinin, M.M., Izv. Akad. Nauk SSSR, Ser. Khim., 1988, no. 11, p. 2640.
Yakovlev, V.Yu. and Fomkin, A.A., Colloid J., 2009, vol. 71, no. 6, p. 877.
Men’shchikov, I.E., Fomkin, A.A., Tsivadze, A.Yu., Shkolin, A.V., Strizhenov, E.M., and Pulin, A.L., Prot. Met. Phys. Chem. Surf., 2015, vol. 51, no. 4, p. 493.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Khozina
Rights and permissions
About this article
Cite this article
Shevchenko, A.O., Pribylov, A.A., Zhedulov, S.A. et al. Methane Adsorption in Microporous Carbon Adsorbent LCN Obtained by Thermochemical Synthesis from Lignocellulose. Prot Met Phys Chem Surf 55, 211–216 (2019). https://doi.org/10.1134/S2070205119020242
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205119020242