Abstract
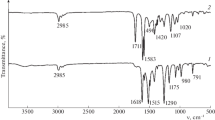
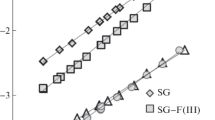
Surface properties of silica gels modified by trifunctional polyfluorosilanes n-C6F13(CH2)2SiCl3 and iso-C3F7(CH2)2SiCl3 are studied using the methods of adsorption and gas chromatography with respect to nitrogen, hydrocarbon, and water molecules. It is shown that the branched structure of the grafted group and high concentration of grafted groups (2.7 nm–2) provide better screening of both residual silanols of the support and of additional hydroxyl groups formed in the course of synthesis as a result of hydrolysis. Comparison with the properties of silica gel with grafted monofunctional silane n-C6F13(CH2)2Si(CH3)2Cl shows that, in the case of an overall decrease in the adsorption values and considerable surface lyophobization, modification by trifunctional silanes irrespective of the modifier chain length allows obtaining adsorbents containing more polar centers than those in silica gel due to the possible participation in adsorption of additional silanols and a polar fragment of the grafted chain of–CH2–CF2–. A considerable difference in the course of isotherms of nitrogen and hexane adsorption–desorption is found in the range of capillary condensation hysteresis related to liophobicity of polyfluoroalkyl layers, a significant decrease in the pore wetting and the corresponding increase in wetting angle θ by hexane. Adsorption of water on fluorinated silicas is negligible and capillary condensation is not observed due to nonwettability of the pore surface by liquid adsorbate (θ > 90°).
Similar content being viewed by others
References
Parker, A.R. and Lawrence, Ch.R., Nature, 2001, vol. 414, p. 33.
Tuteja, A., Choi, W., Mabry, J.M., et al., Proc. Natl. Acad. Sci. USA, vol. 105, p. 18200.
Kang, S.-M., Hwang, S., Jin, S.-H., et al., Langmuir, 2014, vol. 30, p. 2828.
Ishizaki, T., Saito, N., and Takai, O., Langmuir, 2010, vol. 26, p. 8147.
Grinthal, A., Kang, S.H., Epstein, A.K., et al., Nano Today, 2011, no. 7, p. 35.
Khimiya privitykh poverkhnostnykh soedinenii (Chemistry of Grafted Surface Compounds), Lisichkin, G.V., Ed., Moscow: Fizmatlit, 2003.
Fadeev, A.Y., in Encyclopedia of Surface and Colloid Science, Somasundaran, P., Ed., New York: Taylor & Francis, 2006, p. 2854.
Muidinov, M.R., Ross. Khim. Zh., 2002, vol. 46, p. 64
Muidinov, M.R., Ross. Khim. Zh., 2008, vol. 52, p. 81.
Darmanin, T., Tarrade, J., Celia, E., and Guittard, F., J. Phys. Chem., 2014, vol. 118, p. 2052.
Bernardoni, F. and Fadeev, A.Y., J. Colloid Interface Sci., 2011, vol. 356, p. 690.
Kudryavtsev, G.V. and Staroverov, S.M., Zh. Vses. Khim. O-va im. D. I. Mendeleeva, 1989, vol. 34, p. 308.
Duchet, J., Chabert, B., Chapel, J.P., et al., Langmuir, 1997, vol. 13, p. 2271.
Kiselev, A.V., Mezhmolekulyarnye vzaimodeistviya v adsorbtsii i khromatografii (Intermolecular Interactions in Adsorption and Chromatography), Moscow: Vysshaya Shkola, 1986.
Kiselev, A.V., Lisichkin, G.V., Nikitin, Yu.S., et al., Zh. Fiz. Khim., 1983, vol. 57, p. 1829.
Gurevich, K.B., Roshchina, T.M., Shonia, N.K., et al., J. Colloid Interface Sci., 2002, vol. 254, p. 39.
Tarasevich, Yu.I., Polyakov, V.E., Serdan, A.A., and Lisichkin, G.V., Colloid J., 2005, vol. 67, no. 5, pp. 638–643.
Roshchina, T. M., Shoniya, N.K., Tayakina, O. Ya., et al., Prot. Met. Phys. Chem. Surf., 2014, vol. 50, no. 3, pp. 304–311.
Roshchina, T.M., Shoniya, N.K., Tayakina, O.Ya., et al., Russ. J. Phys. Chem. A, 2012, vol. 86, no. 3, pp. 437–446.
Greg, S. and Sing, K., Adsorption, Surface Area and Porosity, New York: Academic Press, 1982.
Eksperimental'nye metody v adsorbtsii i molekulyarnoi khromatografii (Experimental Methods in Adsorption and Molecular Chromatography), Nikitin, Yu.S. and Petrova, R.S., Eds., Moscow: Moscow State Univ., 1990.
Touhara, H. and Okino, F., Carbon, 2000, vol. 38, no. 2, p. 241.
Monde, T., Nakayama, N., Yano, K., et al., J. Colloid Interface Sci., 1997, vol. 185, p. 111.
Roshchina, T.M., Gurevich, K.B., Fadeev, A.Y., et al., J. Chromatogr. A, 1999, vol. 844, p. 225.
Karnaukhov, A.P., Adsorbtsiya. Tekstura dispersnykh i poristykh materialov (Adsorption. Texture of Dispersed and Porous Materials), Novosibirsk: Nauka, 1999.
Avgul', N.N., Berezin, G.I., Kiselev, A.V., and Lygina, I.A., Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 1957, p. 1051.
Zefirov, Yu.V. and Zorkii, P.M., Usp. Khim., 1995, vol. 64, p. 446.
Roshchina, T.M., Shoniya, N.K., Bernardoni F., and Fadeev A.Y., Langmuir, 2014, vol. 30, p. 9355.
Roshchina, T.M., Shoniya, N.K., Lagutova, M.S., and Fadeev, A.Yu., Russ. J. Phys. Chem. A, 2008, vol. 82, no. 3, p. 390.
Roshchina, T.M., Shoniya, N.K., Tayakina, O.Ya., and Fadeev, A.Yu., Russ. J. Phys. Chem. A, 2011, vol. 85, no. 2, p. 298.
Lopatkin, A.A., Russ. J. Phys. Chem. A, 1997, vol. 71, no. 5, p. 811.
Handbuch der Gaschromatographie, Leibnitz, E. and Struppe, H.G., Eds., Leipzig: Akademische Verlagsgesellschaft Geest&Portig, 1966.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.M. Roshchina, N.K. Shoniya, O.Ya. Tegina, F. Bernardoni, A.Y. Fadeev, 2016, published in Fizikokhimiya Poverkhnosti i Zashchita Materialov, 2016, Vol. 52, No. 4, pp. 354–362.
Rights and permissions
About this article
Cite this article
Roshchina, T.M., Shoniya, N.K., Tegina, O.Y. et al. Surface properties of silica gel modified by trifunctional polyfluoroalkyl silanes according to data of adsorption–structural and gas chromatography studies. Prot Met Phys Chem Surf 52, 590–598 (2016). https://doi.org/10.1134/S2070205116040201
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205116040201