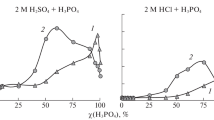
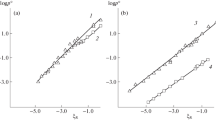
Abstract
Electrochemical classification of the inhibitors of metal corrosion in acidic and neutral environments is analyzed, and its certain modification is proposed. The role of changes in the corrosion potential (which is an electrochemical factor) in the deceleration of the process is considered. Expressions for calculating the effect of the electrochemical factor are derived.
Similar content being viewed by others
References
Rozenfeld, I.L., Ingibitory korrozii (Corrosion Inhibitors), Moscow: Khimiya, 1977.
Antropov, L.I., Makushin, E.M., and Panasenko, V.F., Ingibitory korrozii metallov (Metal Corrosion Inhibitors), Kiev: Tekhnika, 1981.
Damaskin, B.B., Petrii, O.A., and Tsirlina, G.A., Elektrokhimiya (Electrochemistry), Moscow: Khimiya, 2001.
Antropov, L.I., Zashch. Met., 1977, vol. 13, no. 4, p. 387.
Ekilik, V.V. and Chikov, O.V., Zashch. Met., 1991, vol. 27, no. 1, p. 72.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.V. Ekilik, A.G. Berezhnaya, G.N. Ekilik, E.N. Balakshina, published in Korroziya: Materialy, Zashchita, 2010, No. 5, pp. 15–18.
Rights and permissions
About this article
Cite this article
Ekilik, V.V., Berezhnaya, A.G., Ekilik, G.N. et al. On the classification of inhibitors of homogeneous metal corrosion and the role of electrochemical factor in deceleration of process. Prot Met Phys Chem Surf 46, 771–774 (2010). https://doi.org/10.1134/S2070205110070063
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205110070063