Abstract
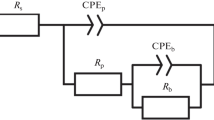
Capacitances of oxide films obtained on a EZ-1 alloy during the corrosion tests in water-vapor environment at 300, 350, and 400°C are measured. In order to estimate the thickness of a barrier layer, the barrier potential at a constant anodic current (dielectric puncture potential) is measured. It is shown that the barrier layer cannot be treated as a homogeneous environment. At the sites of intermetallide inclusions in the oxide film, the thickness of the dielectrics is locally decreased. In the first approximation, the heterogeneity of the oxide film can be taken into account by inserting two parallel RC subcircuits in the equivalent scheme. One subcircuit (C 1, R 1) describes the electrophysical properties of the capacitance whose insulator thickness corresponds to the total thickness of the oxide film. The other subcircuit (C 2, R 2) describes the electrophysical properties of the nonporous part of the oxide film between the intermetallide particles and the outer surface. Then, the results of measurements can be written as follows: C exp = θC 2 + (1 − θ)C 1, where θ is the surface part of the oxide film whose dielectric properties are changed due to the inclusion of intermetallide particles. Assuming that the mean spatial size of intermetallide particles falls in the range of 200–400 nm, one can estimate the mean concentration of the particles on the metal surface in agreement with the metallographically determined concentration of the second-phase particles (approximately 106–107 cm−2). The obtained results indicate the substantial heterogeneity of the barrier layer structure, which may cause local corrosion and premature failure of zirconium items.
Similar content being viewed by others
References
Waterside Corrosion of Zirconium Alloys in Nuclear Power Plants, IAEA-TECDOC-996, Vienna, 1998, p. 152.
Misch, R.D., US AEC Report-ANL-5229, 1953.
Polling, J.J. and Charlesby, A., Acta Met, 1954, vol. 2, no. 5, p. 667.
Nikitin, K.N., Voprosy Atom. Nauki Tekhn., Ser. Materialoved. Novye Mater., 2003. vol. 1(61), p. 129.
Cox, B., J. Nucl. Mat., 1969, vol. 29, no. 1, p. 50.
Lastman, B. and Keres, F., The Metallurgy of Zirconium, New York: McGraw-Hill, 1955.
Wanklyn, J.N. and Silvester, D.R., J. Electrochem. Soc., 1958, vol. 105, no. 11, p. 647.
Barberis, P. and Frichet, A., J. Nucl. Mat., 1999, vol. 273, p. 182.
Hunter M.S. and Fowle, P., J. Electrochem. Soc., 1956, vol. 103, no. 9, p. 482.
Flint, O. and Varley, J.H.O., J. Phys. Chem. Solids, 1958, vol. 6, nos. 2–3, p. 213.
Anada, H., Herb, B.J., Nomomoto, K., et al., 11th Int. Symp. ASTM STP 1295, 1996, p. 74.
Hutchinson, B., Lehtinen, B., Limbäck, M., et al., 15th Int. Symp. ASTM STP, 2007.
Bojinov, M., Hansson-Lyyra, L., Kinnunen, P., et al., J. ASTM Int., 2005, vol. 2, no. 4, p. 367.
Barberis, P., Ahlberg, E., Simic, N., et al., 13th Int. Symp. ASTM STP 1423, 2002, p. 33.
Bossis, P., Leievre, G., Barberis, P., et al., 12th Int. Symp. ASTM STP 1354, 2000, p. 918.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © K.N. Nikitin, V.N. Shishov, 2010, published in Fizikokhimiya Poverkhnosti i Zashchita Materialov, 2010, Vol. 46, No. 2, pp. 214–219.
Rights and permissions
About this article
Cite this article
Nikitin, K.N., Shishov, V.N. Behavior of a barrier layer of corrosion films on zirconium alloys. Prot Met Phys Chem Surf 46, 261–266 (2010). https://doi.org/10.1134/S2070205110020140
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205110020140