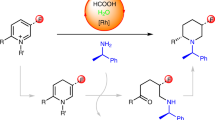
Abstract
Piperazine synthesis methods on intermolecular and intramolecular cyclization catalytic processes are compared and reviewed analytically. The advantages and disadvantages of current ways of synthesizing piperazine are described while focusing on the preferred and highly selective processes of intramolecular cyclization using aminoethylethanolamine and diethylenetriamine, and one-step intermolecular cyclization using ethylenediamine, mono-, and diethanolamine.










Similar content being viewed by others
REFERENCES
Piperazin: Sbornik statei/AN Latviiskoi SSR (Piperazine: Collection of Papers/AN Latvian SSR), Giller, S.A. and Shimanska, M.V., Eds., Riga: Zinatne, 1965.
GNITIVE Marker Research. Global Piperazine–PIP Market Report 2022. https://www.cognitivemarketresearch.com/chemical-%26-materials/piperazine-%28 pip%29-market-report. Cited April 17, 2022.
Sridhar, S. and Carter, R.G., in: Kirk-Othmer Encyclopedia of Chemical Technology, New York: Wiley, 2001. https://doi.org/10.1002/0471238961.0409011303011820.a01.pub
Roose, P., Eller, K., Henkes, E., Rossbacher, R., and Höke, H., in: Ullmann’s Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, 2015. https://doi.org/10.1002/14356007.a02_001.pub2
Shimanskaya, M.V., Oshis, Ya.F., and Anderson, A.A., Chem. Heterocycl. Compd., 1983, vol. 19, no. 12, pp. 1249–1262.
KR Patent 20170041077, 2017.
Lei, M., Li, Y., An-Hui, L., and Yunjie, D., RSC Adv., 2018, vol. 8, no. 15, pp. 8152−8163. https://doi.org/10.1039/c7ra12891f
Borisenko, V.S., Bobylev, V.A., and Tereshchenko, G.F., Zh. Obshch. Khim., 1985, vol. 55, no. 5, pp. 1141–1146.
Borisenko, V.S., Bobylev, V.A., Selivanov, S.I., Utsal’, V.A., and Tereshchenko, G.F., Zh. Obshch. Khim., 1986, vol. 56, no. 10, pp. 2339–2345.
Jpn. Patent 6301695, 2018.
Clapp, L.B., J. Am. Chem. Soc., 1948, vol. 70, no. 1, pp. 184–186. https://doi.org/10.1021/ja01181a053
Ishiguro, T., Kitamura, E., and Matsumura, M., J. Pharm. Soc. Jpn., 1957, vol. 77, no. 10, pp. 1051–1054. https://doi.org/10.1248/yakushi1947.77.10_1051
USSR Inventor’s Certificate no. 166033, Byull. Izobret., 1964.
Ishiguro, T., Kitamura, E., Matsumura, M., and Ogawa, H., J. Pharm. Soc. Jpn., 1955, vol. 75, no. 11, pp. 1318–1321. https://doi.org/10.1248/yakushi1947.75.11_1318
Sasaki, T., J. Synt. Org. Chem., Jpn. 1958, vol. 16, no. 11, pp. 614–620. https://doi.org/10.5059/yukigoseikyokaishi.16.614
Balandin, A.A., Karpeiskaya, E.I., Ferapontov, V.A., and Tolstopyatova, A.A., Dokl. Akad. Nauk SSSR, 1965, vol. 165, no. 1, pp. 99–102.
Balandin, A.A., Ferapontov, V.A., Karpeiskaya, E.I., Gorshkova, L.S., and Tolstopyatova, A.A., Dokl. Akad. Nauk SSSR, 1966, vol. 168, no. 5, pp. 1061–1064.
Sun, Y., J. Chem. Eng. Chin. Univ., 1999, vol. 13, no. 2, pp. 178–181.
US Patent 4234730, 1980.
Jiangang, L., Jiansheng, L., Xiuyun, S., and Lianjun, W., Petrochem. Technol. (China), 2004, vol. 33, no. 9, pp. 861–864.
Zhang, Y., Bai, G., Yan, X., Li, Y., Zeng, T., Wang, J., Wang, H., Xing, J., Luan, D., Tang, X., and Chen, L., Catal. Commun., 2007, vol. 8, no. 7, pp. 1102–1106. https://doi.org/10.1016/j.catcom.2006.10.018
Shang, H., Wang, L., Wang, S., Gao, P., and Zheng, X., Chem. React. Eng. Technol. (China), 2012, vol. 28, no. 2, pp. 188–192.
Jpn. Patent 5803619, 2013.
Corma, A., Ródenas, T., and Sabater, M.J., Chem.—Eur. J., 2010, vol. 16, no. 1, pp. 254–260. https://doi.org/10.1002/chem.200901501
Jenner, G. and Bitsi, G., J. Mol. Catal., 1988, vol. 45, no. 2, pp. 165–168. https://doi.org/10.1016/0304-5102(88)80005-1
US Patent 3068232, 1962.
Yang, Z., Gong, F., and Qi, Y., Chem. React. Eng. Technol. (China), 2016, vol. 32, no. 6, pp. 570–575.
Weitkamp, J., Ernst, S., Buysch, H.-J., and Lindner, D., Stud. Surf. Sci. Catal., 1991, vol. 65, pp. 297–304. https://doi.org/10.1016/S0167-2991(08)62914-6
Sun, S.-H., Shen, W., Xu, H.-L., Zhou, Y.-M., Xiang, Y., and Liu, J.-X., Chin. J. Catal., 1998, vol. 19, no. 5, pp. 432–435.
Liu, R.-J., Bao, J.-Y., Zhang, Y., Tian, Z., and Wei, Z.-X., Chem. Eng. (China), 2006, vol. 34, no. 6, pp. 68–71.
Wei, X.-X., Tian, Z.-S., Liang, J.-G., Liu, R.-J., Hao, H., and Wei, N.-N., Chem. Eng. (China), 2012, vol. 40, no. 1, pp. 56–58.
Gen-zhi, G., Yan-xin, L., Bin, Z., and Xiao-feng, Z., J. Petrochem. Univ. (China), 2006, vol. 19, no. 3, pp. 48–50.
Ishiguro, T., Kitamura, E., and Matsumura, M., J. Pharm. Soc. Jpn., 1953, vol. 73, no. 10, pp. 1110–1114. https://doi.org/10.1248/yakushi1947.73.10_1110
Sasaki, T., J. Synt. Org. Chem., Jpn., 1959, vol. 17, no. 1, pp. 17–28. https://doi.org/10.5059/yukigoseikyokaishi.17.17
Kliger, G.A., Fidler, Kh., Lesik, O.A., Khabish, D., Khlebov, L.S., Zaikin, V.G., Khaage, K., and Loktev, S.M., Neftekhimiya, 1988, vol. 28, no. 5, pp. 684–694.
US Patent 8981093, 2015.
Ishiguro, T., Kitamura, E., Matsumura, M., and Ogawa, H., J. Pharm. Soc. Jpn., 1955, vol. 75, no. 11, pp. 1367–1369. https://doi.org/10.1248/yakushi1947.75.11_1367
Lorentz-Petersen, L.L.R., Nordstrøm, L.U., and Madsen, R., Eur. J. Org. Chem., 2012, vol. 34, pp. 6752–6759. https://doi.org/10.1002/ejoc.201201099
US Patent 3112317, 1963.
Kelly, J.W., Eskew, N.L., and Evans, S.A.Jr., J. Org. Chem., 1986, vol. 51, no. 1, pp. 95–97. https://doi.org/10.1021/jo00351a020
CN Patent 102442957, 2016.
Kitchen, L.J. and Pollard, C.B., J. Am. Chem. Soc., 1947, vol. 69, no. 4, pp. 854–855. https://doi.org/10.1021/ja01196a034
Karpeiskaya, E.I., Ferapontov, V.A., Tolstopyatova, A.A., and Balandin, A.A., Russ. Chem. Bull., 1968, vol. 17, no. 7, pp. 1419–1424. https://doi.org/10.1007/BF00907837
Hammerschmidt, W., Baiker, A., Wokaun, A., and Fluhr, W., Appl. Catal., 1986, vol. 20, nos. 1–2, pp. 305–312. https://doi.org/10.1016/0166-9834(86)80022-7
Chen, L.-G., Song, Y., Xu, Z.-S., Wang, D.-L., and Meng, Y., Chem. Ind. Eng. (China), 2001, vol. 18, no. 1, pp. 40–43.
Subrahmanyam, M., Kulkarni, S.J., and Srinivas, B., React. Kinet. Catal. Lett., 1993, vol. 49, no. 2, pp. 455–459. https://doi.org/10.1007/BF02067715
Wu, G.-J. and Yan, Z.-G., J. East China Univ. Technol., Nat. Sci., 2001, vol. 27, no. 1, pp. 30–33.
CN Patent 100334078, 2007.
Bai, G.Y., Li, Y., Yan, X.L., He, F., and Chen, L.G., React. Kinet. Catal. Lett., 2004, vol. 82, no. 1, pp. 33–39. https://doi.org/10.1023/b:reac.0000028802.66602.0f
Wu, Z., Yang, F., Wang, H., Ma, J., Chen, L., and Li, Y., React. Kinet. Mech. Catal., 2012, vol. 106, no. 2, pp. 485–493. https://doi.org/10.1007/s11144-012-0447-z
Wu, Z., Wang, H., Sun, M., Du, X., Chen, L., and Li, Y., Res. Chem. Intermed., 2012, vol. 38, nos. 3–5, pp. 1149–1157. https://doi.org/10.1007/s11164-011-0450-4
Ford, M.E., Johnson, T.A., Premecz, J.E., and Cooper, C.A., J. Mol. Catal., 1988, vol. 44, no. 2, pp. 207–211. https://doi.org/10.1016/0304-5102(88)80032-4
Braz, G.I. and Skorodumov, V.A., Dokl. Akad. Nauk SSSR, 1948, vol. 59, no. 3, pp. 489–492.
Trejbal, J. and Petrisko, M., React. Kinet. Catal. Lett., 2004, vol. 82, no. 2, pp. 339–346. https://doi.org/10.1023/b:reac.0000034846.59643.e6
WO Patent 2013095810, 2013.
Martin, W.B. and Martell, A.E., J. Am. Chem. Soc., 1948, vol. 70, no. 5, pp. 1817–1818. https://doi.org/10.1021/ja01185a049
USSR Inventor’s Certificate no. 467073, Byull. Izobret., 1975.
USSR Inventor’s Certificate no. 539881, Byull. Izobret., 1976.
Bhat, Y.S., Kulkarnl, B.D., and Doralswamy, L.K., Ind. Eng. Chem. Process Des. Dev., 1985, vol. 24, no. 3, pp. 525–530. https://doi.org/10.1021/i200030a00
Jpn. Patent 6638364, 2020.
Mosher, H.S., Cornell, J.Jr., Stafford, O.L., and Roe, T., J. Am. Chem. Soc., 1953, vol. 75, no. 20, pp. 4949–4951. https://doi.org/10.1021/ja01116a020
US Patent 2809196, 1957.
SU Patent 697501, Byull. Izobret., 1979, no. 42.
Voutchkova, A.M., Gnanamgari, D., Jakobsche, C.E., Butler, C., Miller, S.J., Parr, J., and Crabtree, R.H., J. Organomet. Chem., 2008, vol. 693, no. 10, pp. 1815–1821. https://doi.org/10.1016/j.jorganchem.2008.02.004
Nasiruzzaman, M.S., Abdul, M.A., Kalanthoden, A.N., Helal, A., Hakeem, A.S., and Bououdina, M., Catal. Sci. Technol., 2018, vol. 8, no. 18, pp. 4709–4717. https://doi.org/10.1039/c8cy00936h
Kim, S., Loose, F., Bezdek, M.J., Wang, X., and Chirik, P.J., J. Am. Chem. Soc., 2019, vol. 141, no. 44, pp. 17900–17908. https://doi.org/10.1021/jacs.9b09540
Chatterjee, B., Kalsi, D., Kaithal, A., Bordet, A., Leitner, W., and Gunanathan, C., Catal. Sci. Technol., 2020, vol. 10, pp. 5163–5170. https://doi.org/10.1039/D0CY00928H
Ciotonea, C., Hammi, N., Dhainaut, J., Marinova, M., Ungureanu, A., El Kadib, A., Michon, C., and Royer, S., ChemCatChem, 2020, vol. 12, no. 18, pp. 4652–4663. https://doi.org/10.1002/cctc.202000704
Lei, M., Li, Y., An-Hui, L., and Yunjie, D., Chin. J. Catal., 2019, vol. 40, no. 4, pp. 567–579.
Funding
This work was supported by the RF Ministry of Science and Higher Education as part of a State Task for the Boreskov Institute of Catalysis, project no. AAAA-A21-121011390007-7.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by A. Tulyabaev
APPENDIX A
APPENDIX A
AEEA—aminoethylethanolamine
DCE—1,2-dichloroethane
DEA—diethanolamine
DETA—diethylenetriamine
IDAN—iminodiacetonitrile
MOR—morpholine
MEA—monoethanolamine
PP—piperazine
PR—pyrazine
PEA—polyethyleneamines
TEDA—triethylenediamine
TEPA—tetraethylenepentamine
EG—ethylene glycol
EDA—ethylenediamine
Rights and permissions
About this article
Cite this article
Sergeev, E.E., Gogin, L.L., Khlebnikova, T.B. et al. Methods for the Catalytic Synthesis of Piperazine. Catal. Ind. 14, 218–230 (2022). https://doi.org/10.1134/S2070050422020076
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070050422020076