Abstract
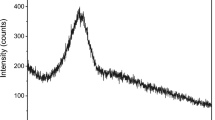
Superplasticizers are one of the most widespread chemical additives in the concrete industry. They peptize cement floccules and increase the mobility of concrete mixes at a decreased water-to-cement ratio due to the release of immobilized water, a decrease in the internal friction coefficient, and an increase in electrostatic repulsion of particles due to a significant change of their electrokinetic potential. A new generation of superplasticizers based on polycarboxylate ester have been developed in recent decades, which possesses a different dispersion ability of cement particles than do old-generation superplasticizers. However, the molecules of polycarboxylate superplasticizer are more sensitive to clay minerals in concrete mixes. The effect of the strength characteristics of the ground, such as angle of internal friction and specific cohesion, has been studied. Specific interactions between kaolinite mineral and various superplasticizers have been studied using IR spectroscopy. Intrinsic bands in IR spectra of superplasticizers corresponding to their functional groups have been interpreted.





Similar content being viewed by others
REFERENCES
J. Plank, E. Sakai, C. W. Miao, C. Yu, and J. X. Hong, “Chemical admixtures—chemistry, applications and their impact on concrete microstructure and durability,” Cem. Concr. Res. 78, 81–99 (2015).
K. Koizumi, Y. Umemura, and N. Tsuyuki, “Effects of chemical admixtures on the silicate structure of hydrated Portland cement,” in Proceedings of the 12th International Congress on the Chemistry of Cement (Montreal, 2007), pp. 64–71.
O. V. Tarakanov and V. I. Kalashnikov, “Prospects for the use of complex additives in new-generation concrete,” Izv. Kazan. Gos. Arkhit.-Stroit. Univ., No. 1, 223–229 (2017).
L. Ya. Kramar, A. I. Kudyakov, B. Ya. Trofimov, and K. V. Shuldyakov, “Cemented heavy concretes for construction of high-speed roads,” Vestn. Tomsk. Gos. Arkhit.-Stroit. Univ., No. 4, 147–157 (2017).
N. I. Strelkov, “The role of super- and hyperplasticizers in the formation of high concrete strength,” Intell. Sist. Proizvod. 16 (1), 83–91 (2018).
W. Guo, N. Sun, J. Qin, J. Zhang, M. Pei, Y. Wang, and Sh. Wang, “Synthesis and properties of an amphoteric polycarboxylic acid-based superplasticizer used in sulfoaluminate cement,” J. Appl. Polym. Sci. 125 (1), 283–290 (2012).
H. Zhao, M. Deng, and M. Tang, “Synthesis, characterization and dispersing mechanism of aminosulfonatephenol-formaldehyde superplasticizer for the cement particles,” Polym. Compos. 39 (7), 2250–2258 (2018).
H. El-Didamony, M. Heikal, and S. Abd El Aleem, “Influence of delayed addition time of sodium sulfanilate phenol formaldehyde condensate on the hydration characteristics of sulfate resisting cement pastes containing silica fume,” Constr. Build. Mater. 37, 269–276 (2012).
H. Lou, K. Ji, H. Lin, Yu. Pang, Yo. Deng, X. Qiu, H. Zhang, and Zh. Xie, “Effect of molecular weight of sulphonated acetone-formaldehyde condensate on its adsorption and dispersion properties in cementitious system,” Cem. Concr. Res. 42 (8), 1043–1048 (2012).
Z. Lu, X. Kong, H. Liu, Z. Wang, Ya. Zhang, B. Dong, and F. Xing, “Interaction of silylated superplasticizers with cementitious materials,” J. Appl. Polym. Sci. 133 (43), 44 161 (2016).
M. M. Alonso, M. Palacios, and F. Puertas, “Compatibility between polycarboxylate-based admixtures and blended-cement paste,” Cem. Concr. Compos. 35 (1), 151–162 (2013).
L. Jiang, X. Kong, Z. Lu, and Sh. Hou, “Preparation of amphoteric polycarboxylate superplasticizers and their performances in cementitious system,” J. Appl. Polym. Sci. 132 (4), 41 348 (2015).
H. Tan, B. Gu, B. Ma, X. Li, Ch. Lin, and X. Li, “Mechanism of intercalation of polycarboxylate superplasticizer into montmorillonite,” Appl. Clay Sci. 129, 40–46 (2016).
L. Lei and J. Plank, “A concept for a polycarboxylate superplasticizer possessing enhanced clay tolerance,” Cem. Concr. Res. 42, 1299–1306 (2012).
L. Zhang, Q. Lu, Zh. Xu, Q. Liu, and H. Zeng, “Effect of polycarboxylate ether comb-type polymer on viscosity and interfacial properties of kaolinite clay suspensions,” J. Colloid Interface Sci. 378, 222–231 (2012).
S. Ng and J. Plank, “Interaction mechanisms between Na montmorillonite clay and MPEG-based polycarboxylate superplasticizers,” Cem. Concr. Res. 42, 847–854 (2012).
R. Ait-Akbour, P. Boustingorry, F. Leroux, F. Leising, and Ch. Taviot-Gueho, “Adsorption of PolyCarboxylatePoly(ethylene glycol) (PCP) esters on Montmorillonite (Mmt): Effect of exchangeable cations (Na+, Mg2+ and Ca2+) and PCP molecular structure,” J. Colloid Interface Sci. 437, 227–234 (2015).
D. Zhang, C.-H. Zhou, C.-X. Lin, D.-S. Tong, and W.-H. Yu, “Synthesis of clay minerals,” Appl. Clay Sci. 50, 1–10 (2010).
P. M. Amarasinghe, K. S. Katti, and D. R. Katti, “Nature of organic fluid-montmorilloniteó interactions: An FTIR spectroscopic,” J. Colloid Interface Sci. 337, 97–105 (2009).
P. E. Bulanov, L. F. Mavliev, E. A. Vdovin, and E. M. Yagund, “The interaction between the kaolinite or bentonite clay and plasticizing surface-active agents,” Mag. Civil Eng., No. 7, 171–179 (2017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Translated by A. Muravev
Rights and permissions
About this article
Cite this article
Bulanov, P.E., Ermilova, E.Y., Yagund, E.M. et al. Interaction of Various Superplasticizers with Kaolinite Clay. Polym. Sci. Ser. D 12, 454–458 (2019). https://doi.org/10.1134/S199542121904004X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S199542121904004X