Abstract
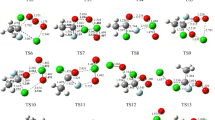
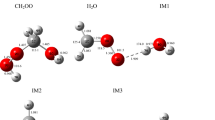
Carbonyl oxides, or Criegee intermediates, play an important role in many physicochemical processes occurring in the Earth’s atmosphere. Criegee intermediates are chemically active compounds that easily react with other atmospheric components, promoting the formation of OH and CH3 radicals, toxic compounds of nitrogen, and various acids. Traditionally, the literature considers reactions involving only those carbonyl oxides that are the most stable in the troposphere under the standard atmospheric conditions. In this study, it is shown that in the mesosphere and ionosphere, where the total concentration of molecules is low and the intensity of UV radiation and the number of free electrons are high, reactions involving electronically excited states of the Criegee intermediates CH3CHOO play a significant role. In this case, we should take into account the features of the decomposition of all isomers of the CH3CHOO molecule.



Similar content being viewed by others
REFERENCES
R. Criegee and G. Wenner, Just. Lieb. Ann. Chem. 564, 9 (1949).
M. Khan, C. Percival, R. Caravan, et al., Environ. Sci.: Proces. Impacts 20, 437 (2018).
C. A. Taatjes, M. A. H. Khan, A. J. Eskola, et al., Environ. Sci. Technol. 53, 1245 (2019).
O. Welz, A. J. Eskola, L. Sheps, et al., Angew. Chem. Int. Ed. 53, 4547 (2014).
R. Chhantyal-Pun, M. R. McGillen, J. M. Beames, et al., Angew. Chem. Int. Ed. 56, 9044 (2017).
C. A. Taatjes, O. Welz, A. J. Eskola, et al., Science (Washington, DC, U. S.) 340, 177 (2013).
Yu. A. Dyakov, S. O. Adamson, P. K. Wang, A. S. Vetchinkin, G. V. Golubkov, I. I. Morozov, S. Y. Umanskii, Y. A. Chaikina, and M. G. Golubkov, Russ. J. Phys. Chem. B 15, 782 (2021).
C. A. Taatjes, D. E. Shallcross, and C. J. Percival, Phys. Chem. Chem. Phys. 16, 1704 (2014).
J. T. Herron, R. I. Martinez, and R. E. Huie, Int. J. Chem. Kinet. 14, 201 (1982).
W. Chao, J. T. Hsieh, C. H. Chang, et al., Science (Washington, DC, U. S.) 347, 751 (2015).
B. Long, J. L. Bao, and D. G. Truhlar, J. Am. Chem. Soc. 138, 14409 (2016).
L. Sheps, A. M. Scully, and K. Au, Phys. Chem. Chem. Phys. 16, 26701 (2014).
H. Levy, Science (Washington, DC, U. S.) 173, 141 (1971).
X. H. Wang and J. M. Bowman, J. Phys. Chem. Lett. 7, 3359 (2016).
N. M. Kidwell, H. W. Li, X. H. Wang, et al., Nat. Chem. 8, 509 (2016).
Y. Fang, F. Liu, V. P. Barber, et al., J. Chem. Phys. 144, 061102 (2016).
J. Lelieveld, F. Dentener, W. Peters, et al., Atmos. Chem. Phys. 4, 2337 (2004).
M. Kanakidou, J. H. Seinfeld, S. N. Pandis, et al., Atmos. Chem. Phys. 5, 1053 (2005).
M. Hallquist, J. C. Wenger, U. Baltensperger, et al., Atmos. Chem. Phys. 9, 5155 (2009).
E. S. Foreman, K. M. Kapnas, and C. Murray, Angew. Chem. Int. Ed. 55, 10419 (2016).
L. Vereecken, H. Harder, and A. Novelli, Phys. Chem. Chem. Phys. 14, 14682 (2012).
M. Sharifi, F. Kong, S. L. Chin, et al., J. Phys. Chem. A 111, 9405 (2007).
Z. Wang, Y. A. Dyakov, and Y. Bu, J. Phys. Chem. A 123, 1085 (2019).
Yu. A. Dyakov, A. A. Puzankov, S. O. Adamson, G. V. Golubkov, I. I. Morozov, O. A. Olkhov, V. L. Shapovalov, D. V. Shestakov, and M. G. Golubkov, Russ. J. Phys. Chem. B 14, 728 (2020).
Q. Q. Wang, Y. A. Dyakov, D. Wu, et al., Chem. Phys. Lett. 586, 21 (2013).
H. Li, Y. Fang, N. M. Kidwell, et al., J. Phys. Chem. A 119, 8328 (2015).
R. L. Mauldin, T. Berndt, and M. Sipila, Nature (London, U.K.) 488, 193 (2012).
Yu. A. Dyakov, S. O. Adamson, P. K. Wang, G. V. Golub-kov, O. A. Olkhov, V. D. Peskov, I. D. Rodionov, I. P. Rodionova, A. I. Rodionov, V. L. Shapovalov, D. V. Shestakov, and M. G. Golubkov, Russ. J. Phys. Chem. B 15, 559 (2021).
T. L. Nguyen, L. McCaslin, M. C. McCarthy, et al., J. Chem. Phys. 145, 131102 (2016).
A. M. Mebel, T. S. Zyubina, Y. A. Dyakov, et al., Int. J. Quant. Chem. 102, 506 (2005).
T. S. Zyubina, Y. A. Dyakov, S. H. Lin, et al., J. Chem. Phys. 123, 134320 (2005).
X. H. Zhou, Y. Q. Liu, W. R. Dong, et al., J. Phys. Chem. Lett. 10, 4817 (2019).
G. V. Golubkov, T. A. Maslov, V. L. Bychkov, O. P. Borchevkina, S. O. Adamson, Yu. A. Dyakov, A. A. Lushnikov, and M. G. Golubkov, Russ. J. Phys. Chem. B 14, 853 (2020).
V. V. Kuverova, S. O. Adamson, A. A. Berlin, et al., Adv. Space Res. 64, 1876 (2019).
G. V. Golubkov, M. G. Golubkov, and M. I. Manzhelii, Dokl. Phys. 57, 461 (2012).
G. V. Golubkov, M. I. Manzhelii, A. A. Berlin, et al., Atmosphere 11, 650 (2020).
G. V. Golubkov, M. I. Manzhelii, A. A. Berlin, N. N. Bezuglov, A. N. Klyucharev, O. P. Borchevkina, S. O. Adamson, Yu. A. Dyakov, I. V. Karpov, I. I. Morozov, L. V. Eppelbaum, and M. G. Golubkov, Russ. J. Phys. Chem. B 15, 362 (2021).
V. Gerard, C. Galopin, E. Ay, et al., Food Chem. 359, 129949 (2021).
C. K. Ni and Y. T. Lee, Int. Rev. Phys. Chem. 23, 187 (2004).
C. M. Tseng, Y. T. Lee, and C. K. Ni, J. Chem. Phys. 121, 2459 (2004).
C. K. Ni, C. M. Tseng, M. F. Lin, et al., J. Phys. Chem. B 111, 12631 (2007).
M. F. Lin, C. M. Tseng, Y. A. Dyakov, et al., J. Chem. Phys. 126, 241104 (2007).
Y. A. Dyakov, Y. C. Ho, W. H. Hsu, et al., Chem. Phys. 515, 543 (2018).
Y. A. Dyakov, S. Toliautas, L. I. Trakhtenberg, et al., Chem. Phys. 515, 672 (2018).
A. L. Sobolewski and W. Domcke, Phys. Chem. Chem. Phys. 8, 3410 (2006).
Y. L. Yang, Y. C. Ho, Y. A. Dyakov, et al., Phys. Chem. Chem. Phys. 15, 7182 (2013).
C. M. Tseng, Y. A. Dyakov, H. C. Huang, et al., J. Chem. Phys. 133, 074307 (2010).
H. C. Hsu, M. T. Tsai, Y. A. Dyakov, et al., Int. Rev. Phys. Chem. 31, 201 (2012).
H. J. Werner, P. J. Knowles, G. Knizia, et al., Wiley Int. Rev. Comp. Mol. Sci. 2, 242 (2012).
Funding
This study was carried out in the framework of State Assignment of the Ministry of Science and Higher Education of the Russian Federation (registration number 122040500060–4), with the financial support of the Ministry of Science and Technology of Taiwan (MOST grants 109-2111-M-001-001 and 110-2111-M-001-005) and by Higher Education Sprout Project, Ministry of Education to the Headquarters of University Advancement at National Cheng Kung University (NCKU), Taiwan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dyakov, Y.A., Adamson, S.O., Wang, P.K. et al. Excited State Dynamics of CH3CHOO Criegee Intermediates in the Upper Atmosphere of the Earth. Russ. J. Phys. Chem. B 16, 543–548 (2022). https://doi.org/10.1134/S1990793122030149
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793122030149