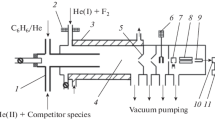
Abstract
Using a low-pressure flow reactor, the kinetic regularities of the reaction of atomic fluorine with monochloroacetic acid at temperature T = 293 K are studied. The concentrations of the reagents are monitored by molecular beam mass spectrometry, detecting the molecular peaks of the substances. Using the published data, the value of the reaction rate constant \(k = (9.6 \pm 3.8) \times {{10}^{{ - 11}}}\) cm3 molecules–1 s–1, which is the average value determined by comparison with the rate constants of competing reactions (kref), is calculated. The ratios of the rate constants k/kref, equal to 0.99 ± 0.12 and 0.75 ± 0.04, respectively, are determined for two competing reactions: a fluorine atom with 2-fluoroethanol and a fluorine atom with cyclohexane. The rate constants of the reaction of atomic fluorine with monochloroacetic acid are compared with the rate constants of reactions of atomic fluorine with chlorine-substituted acetic acids: dichloroacetic and trichloroacetic acids.





Similar content being viewed by others
REFERENCES
A. O. McDougall and F. A. Long, J. Phys. Chem. 66, 429 (1962).
D. J. Bowden, S. L. Clegg, and P. Brimblecombe, J. Atmos. Chem. 29, 85 (1998).
K. Laniewski, H. Boren, and A. Grimvall, Chemosphere 38, 393 (1999).
S. Reimann, K. Grob, and H. Frank, Environ. Sci. Technol. 30, 2340 (1996).
E. S. Vasiliev, I. I. Morozov, and G. V. Karpov, Int. J. Chem. Kinet. 51, 909 (2019).
E. S. Vasil’ev, A. G. Syromyatnikov, D. K. Shartava, et al., Khim. Bezopasn. 2 (1), 206 (2018).
NIST Standard Reference Database. Number 69, Ed. by P. J. Linstrom and W. G. Mallard (Natl. Inst. Stand. Technol., Gaithersburg, USA, 2018).
E. S. Vasil’ev, I. I. Morozov, W. Hack, K. H. Hoyermann, and M. Hold, Kinet. Catal. 47, 834 (2006).
R. Atkinson, D. L. Baulch, R. A. Cox, et al., Atmos. Chem. Phys. 6, 3625 (2006).
R. K. Pearson, J. O. Cowles, G. L. Hermann, et al., IEEE J. Quantum Electron. 9, 879 (1973).
D. J. Smith, D. W. Setser, K. C. Kim, et al., J. Phys. Chem. 81, 898 (1977).
P. Heinemann-Fiedler, K. Hoyermann, and G. Rohde, Ber. Bunsen-Ges. Phys. Chem. 94, 1400 (1990).
E. V. Shtamm, Yu. I. Skurlatov, A. V. Roshchin, V. O. Shvydkii, and L. V. Semenyak, Russ. J. Phys. Chem. B 13, 986 (2019).
Yu. I. Skurlatov, E. V. Shtamm, A. V. Roshchin, V. O. Shvydkii, L. V. Semenyak, and I. V. Semenova, Russ. J. Phys. Chem. B 13, 1033 (2019).
E. S. Vasiliev, G. V. Karpov, N. D. Volkov, I. I. Morozov, and S. V. Savilov, Russ. J. Phys. Chem. B 15, 228 (2021).
E. S. Vasiliev, N. D. Volkov, G. V. Karpov, I. I. Morozov, D. R. Nigmatullin, E. A. Saigina, S. V. Savilov, S. Ya. Umanskii, and N. I. Butkovskaya, Russ. J. Phys. Chem. B 15, 789 (2021).
Funding
This study was carried out in the framework of State Assignment of the Ministry of Science and Higher Education of the Russian Federation (registration number 122040500060–4) and was supported by the Russian Foundation for Basic Research grant no. 19-05-50076 (Microworld).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vasiliev, E.S., Karpov, G.V., Shartava, D.K. et al. Mass Spectrometric Study of the Reaction of a Fluorine Atom and Monocloroacetic Acid. Russ. J. Phys. Chem. B 16, 388–394 (2022). https://doi.org/10.1134/S1990793122030113
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793122030113