Abstract
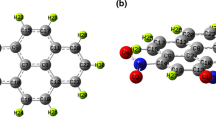
Tyrosine dipeptide (YY) nanotubes have recently been applied as functional elements of photoelectric sensors. Based on the first-principles, density functional theory (DFT) is used to calculate the electronic properties of YY molecules. It is found that YY molecules, similar to gallium nitride, silicon carbide and zinc oxide, present the electronic properties with a wide and direct band gap. Compared with the phenylalanine dipeptide (FF), the YY molecule has a lower band gap. YY has a variety of stable conformations, and the electronic properties of different conformations are different. According to the density of states and energy band of YY, the internal factors affecting its electronic properties are the functions of amino, carboxyl, hydroxyl and other functional groups of molecular structure. Adsorption of water molecules have a significant effect on the electronic properties of YY. When a few water molecules are adsorbed, the band gap of YY decrease. With the ratio of water molecules adsorption increasing, the band gap of different conformations of YY show different change. Through comparative and analysis, the band gap of linear folding boat-shaped water molecules adsorption conformational is the lowest, 2.946 eV, reduced by 0.517eV, and the adsorption ratio of YY to water molecules is 1 : 4, which is an external factor that affect the electronic properties of YY. Combined internal and external factors to explore the electronic properties of YY, the results have laid a good theoretical basis for the application of tyrosine nanomaterials.







Similar content being viewed by others
REFERENCES
B. L. Grigorenko, M. G. Khrenova, A. M. Kulakova, and A. V. Nemukhin, Russ. J. Phys. Chem. B 14, 457 (2020). https://doi.org/10.1134/S1990793120030161
S. Shahab and M. Sheikhi, Russ. J. Phys. Chem. B 14, 15 (2020). https://doi.org/10.1134/S1990793120010145
K. Tao, W. Hu, and B. Hue, Adv. Mater. 31, 1807481 (2019). https://doi.org/10.1002/adma.201807481
V. L. Sedman, L. A. Abramovich, S. Allen, et al., J. Am. Chem. Soc. 128, 6903 (2006). https://doi.org/10.1021/ja060358g
L. A. Abramovich, Z. A. Arnon, X. M. Sui, et al., Adv. Mater. 30, 1704551 (2018). https://doi.org/10.1002/adma.201704551
S. Kim and J. H. Kim, Small 11, 3623 (2015). https://doi.org/10.1002/smll.201500169
M. Wang, S. Xiong, X. Wu, et al., Small 7, 2801 (2011). https://doi.org/10.1002/smll.201100353
X. Wu, Z. Gan, and X. Zhu, Angew. Chem. 52, 2055 (2013). https://doi.org/10.1002/anie.201207992
M. S. Ekiz, G. Cinar, M. A. Khalily, et al., Nanotechnology 27, 402002 (2016). https://doi.org/10.1088/0957-4484/27/40/402002
L. Zhao, Q. Zou, and X. Yan, Bull. Chem. Soc. Jpn. 92, 70 (2019). https://doi.org/10.1246/bcsj.20180248
N. Amdursky, G. Shalev, A. Handelman, et al., APL Mater. 1, 625 (2013). https://doi.org/10.1063/1.4838815
T. V. Belysheva, E. Y. Spiridonova, M. I. Ikim, G. N. Gerasimov, V. F. Gromov, and L. I. Trakhtenberg, Russ. J. Phys. Chem. B 14, 298 (2020). https://doi.org/10.1134/S1990793120020190
I. V. Klimenko, M. A. Gradova, O. V. Gradov, S. B. Bibikov, and A. V. Lobanov, Russ. J. Phys. Chem. B 14, 436 (2020). https://doi.org/10.1134/S1990793120030070
K. Tao, A. Levin, L. A. Abramovich, et al., Chem. Soc. Rev. 45, 3935 (2016). https://doi.org/10.1039/c5cs00889a
N. Amdursky, M. Molotskii, D. Aronov, et al., Nano Lett. 9, 3111 (2009). https://doi.org/10.1021/nl9008265
X. Yan, Y. Su, J. Li, et al., Angew. Chem., Int. Ed. 50, 11186 (2011). https://doi.org/10.1002/ange.201103941
J. S. Lee, I. Yoon, J. Kim, et al., Angew. Chem., Int. Ed. 50, 1164 (2011). https://doi.org/10.1002/ange.201003446
T. Nikitin, S. Kopyl, V. Y. Shur, et al., Phys. Lett. A 380, 1658 (2016). https://doi.org/10.1016/j.physleta.2016.02.043
N. Amdursky, I. Koren, E. Gazit, et al., J. Nanosci. Nanotechnol. 11, 9282 (2011). https://doi.org/10.1166/jnn.2011.4278
M. Telford, Mater. Today 8, 10 (2005). https://doi.org/10.1016/S1369-7021(05)71063-4
R. A. Sakovich, A. Y. Shaulov, E. M. Nechvolodova, and L. A. Tkachenko, Russ. J. Phys. Chem. B 14, 516 (2020). https://doi.org/10.1134/S1990793120030094
X. Zhao and S. Zhang, Macromol. Biosci. 7, 13 (2007). https://doi.org/10.1002/masy.200900171
O. A. Golovanova and K. K. Golovchenko, Russ. J. Phys. Chem. A 93, 2275 (2019). https://doi.org/10.1134/S0036024419110104
A. J. Hassan, Russ. J. Phys. Chem. B 13, 1064 (2019). https://doi.org/10.1134/S1990793119060186
N. V. Dokhlikova, M. V. Grishin, S. Y. Sarvadii, and B. R. Shub, Russ. J. Phys. Chem. B 13, 525 (2019). https://doi.org/10.1134/S1990793119030035
B. E. Krisyuk, Russ. J. Phys. Chem. B 14, 1 (2020). https://doi.org/10.1134/S1990793120010054
T. A. Filho, F. F. Ferreira, W. A. Alves, et al., Phys. Chem. Chem. Phys. 15, 7555 (2013). https://doi.org/10.1039/c3cp43952f
M. Wang, S. Xiong, X. Wu, et al., Small 7, 2801 (2011). https://doi.org/10.1002/smll.201100353
M. Rahimi, H. Chermette, S. Jamehbozorgi, R. Ghiasi, and M. Poor Kalhor, Russ. J. Phys. Chem. A 93, 1747 (2019). https://doi.org/10.1134/S0036024419090139
E. Y. Arkhangelskaya, N. Y. Vorobyeva, S. V. Leonov, A. N. Osipov, and A. L. Buchachenko, Russ. J. Phys. Chem. B 14, 314 (2020). https://doi.org/10.1134/S1990793120020177
S. Akyuz, S. Celik, and A. E. Ozel, Vibr. Spectrosc. 83, 57 (2016). https://doi.org/10.1016/j.vibspec.2016.01.007
S. J. Clark, M. D. Segall, C. J. Pickard, et al., Z. Kristallogr. 220, 5 (2005). https://doi.org/10.1524/zkri.220.5.567.65075
P. E. Blöchl, Phys. Rev. B 50, 17953 (1994). https://doi.org/10.1103/PhysRevB.50.17953
G. Kresse and D. Joubert, Phys. Rev. B 59, 1758 (1999). https://doi.org/10.1103/PhysRevB.59.1758
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett. 77, 3865 (1996). https://doi.org/10.1103/PhysRevLett.78.1396
H. J. Monkhorst and J. D. Pack, Phys. Rev. B 13, 5188 (1976). https://doi.org/10.1103/PhysRevB.13.5188
ACKNOWLEDGMENTS
The work was supported by Joint fund of Science & Technology Department of Liaoning Province and State Key Laboratory of Robotics, China (Grant no.: 2020-KF-22-11). The models were built and calculated using the resources of Fujian Supercomputing Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Wang, S., Li, M. Research on the Electronic Properties of Tyrosine Dipeptide Molecule: Evaluation of the First-principles Theory. Russ. J. Phys. Chem. B 15 (Suppl 2), S181–S188 (2021). https://doi.org/10.1134/S1990793121100079
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793121100079