Abstract
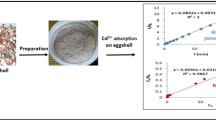
In this research article, environmental waste country eggshells were effectively utilized to prepare eggshell powder—magnetic nano-adsorbent (EM) by ultrasonic-assisted co-precipitation method to delete Cr(VI) from its aqueous solutions. The surface, physiochemical, functional bond, and thermogravimetric weight loss, magnetic characteristics of prepared nano-adsorbent were investigated by using different analytical techniques: X-Ray diffractometer, field emission scanning electron microscope, thermogravimetric analyzer, Fourier-transform infrared spectrophotometer, and vibrating sample magnetometer. The effect of cluster trial working parameters, such as time, initial Cr(VI) concentration, pH, and co-existing anions was examined. The Langmuir, Freundlich, and Temkin adsorption isotherms were analyzed with the batch adsorption equilibrium data ensuring the multilayer adsorption of Cr(VI) onto the EM sites. Thermodynamic parameters were calculated for the batch adsorption process of Cr(VI) removal by EM adsorbent. The novel eggshell coated with magnetic nano-adsorbent (EM) has the more prominent adsorbing potential for Cr(VI) removal from its aquatic environment and the best suited for treating surface, underground and tannery wastewater with 48.5% reusability strength.


















Similar content being viewed by others
REFERENCES
L. C. Hsu, S. L. Wang, and Y. C. Lin, Environ. Sci. Technol. 44, 6202 (2010).
J. Wen-Jun and C. Quan, Environ. Sci. Technol. 48, 8078 (2014).
K. Mulani, S. Daniels, and K. Rajdeo, J. Polym. 2013, 1 (2013).
J. F. Campana-Perez, P/; Partero Barahona, and P. Martin-Ramos, Environ. Sci. Pollut. Res. 26, 28162 (2019).
C. Girish and D. Lamb, Water, Air, Soil Pollut. 224, 1699 (2013).
J. Scancar and R. Milacic, J. Anal. At. Spectrom. 29, 427 (2014).
I. V. Kumpanenko, N. A. Ivanova, and M. V. Dyubanov, Russ. J. Phys. Chem. B 13, 502 (2019). https://doi.org/10.1134/S1990793119030199
L. Zhou, G. Zhang, and J. Tian, ACS Sustain. Chem. Eng. 51, 1042 (2017).
Z. Hu, L. Lei, Y. Li, and Y. Ni, Sep. Purif. Technol. 31, 13 (2003).
D. Balarak, A. R. Yari, and F. K. Mostafapour, Arch. Hyg. Sci. 5, 310 (2016).
K. Vijayaraghavan, J. Jegan, and K. Palanivelu, Miner. Eng. 18, 545 (2005).
B. T. Thanh, N. van Sau, and H. Ju, J. Nanomater. 2019, 1 (2019).
Zh. Chen, J. Song, and Q. Zhu, RSC Adv. 9, 40546 (2019).
D. Chen, Z. Zeng, and Yu. Zeng, Water Resour. Ind. 15, 1 (2016).
H. J. Park, S. W. Jeong, and J. K. Yang, J. Environ. Sci. (China) 19, 1436 (2007).
M. Naghizadeh, H. Sheibani, and M. Taher, Int. J. Energy Environ. Econ. 8, 136 (2017).
A. Faghihi, M. H. Vakili, and G. Hosseinzadeh, Desalin. Water Treatm. 57, 22655 (2016).
Gui-yin Li and Yu-ren Jiang, J. Alloys Compd. 466, 451 (2008).
A. Albadarin and Z. Yang, Chem. Eng. Res. Des. 92, 1354 (2014).
Y. Qi, Y. Wang, and Z. Li, J. Taiwan Inst. Chem. Eng. 95, 1 (2018).
M. Manshouri and H. Daraei, Desalin. Water Treatm. 41, 179 (2012).
S. O. Travin, O. B. Gromov, and D. V. Utrobin, Russ. J. Phys. Chem. B 13, 975 (2019). https://doi.org/10.1134/S1990793119060113
E. Taghdir, M. Aghaie, and V. Hadadi, Russ. J. Phys. Chem. B 9, 399 (2015). https://doi.org/10.1134/S1990793115030100
N. Samadani Langeroodi, Russ. J. Phys. Chem. B 11, 318 (2017). https://doi.org/10.1134/S1990793117020221
M. Kapur and M. K. Mondal, Chem. Eng. J. 218, 138 (2013).
H. Thangappan and A. V. Parambathu, Desalination Water Treat. 57, 21118 (2016).
E. M. El Afifi and M. F. Attallah, J. Environ. Radioact. 151, 156 (2016).
R. Rifaqat, I. Shaista, and U. Mohammad, J. Environ. Chem. Eng. 2, 1155 (2014).
L. Wang, J. Zhang, and R. Zhao, Bioresour. Technol. 101, 5808 (2010).
Z. Y. Ma, Y. P. Guan, and Y. P. Liu, Langmuir 21, 6987 (2005).
M. Moazzen, A. M. Khaneghah, and N. Shariatifar, Arab. J. Chem. 12, 476 (2019).
M. P. Naghizadeh and H. Mohammadi, Iran. J. Energy Environ. 8, 136 (2017).
H. N. Pham and X. P. Nguyen, Adv. Nat. Sci. Nanosci. Nanotechnol. 7, 1 (2016).
S. Asgari, Z. Fakhari, and S. Berijani, J. Nanostruct. 4, 55 (2016).
R. Ahmad, R. Kumar, and S. Haseeb, Arab. J. Chem. 5, 353 (2012).
J. Ren, M. Bopape, K. Setshedi, et al., Chem. Ind. Chem. Eng. Q. 18, 221 (2012).
H. I. Adegoke, F. A. Adekola, and O. S. Fatoki, Korean J. Chem. Eng. 31, 142 (2013).
P. Yuan, D. Liu, M. Fan, and D. Yang, J. Hazard. Mater. 173, 614 (2010).
H. N. Bhatti, A. W. Nasir, and M. A. Hanif, Desalination 253, 78 (2010).
O. Moradi, M. Aghaie, and K. Zare, J. Hazard. Mater. 170, 673 (2009).
A. Bhowal and S. Datta, J. Membr. Sci. 188, 1 (2001).
R. Zeynolabedin and K. Mahanpoor, J. Nanostruct. Chem. 7, 67 (2017).
H. Gao, Y. Liu, and G. Zeng, J. Hazard. Mater. 150, 446 (2008).
K. Komori, P. Wang, and K. Toda, Appl. Microbiol. Biotechnol. 31 (567), 1 (1989).
H. Daraei, A. Mittal, and N. Mohammad, Desalin. Water Treat. 53, 214 (2015).
S. Parlayici and E. Pehlivan, J. Anal. Sci. Technol. 10 (15), 1 (2019).
K. Madhu, P. Charles, and M. Dinesh, J. Colloid Interface Sci. C 442, 120 (2014).
Z. A. Zakaria, M. Suratman, and N. Mohammed, Desalination 244, 109 (2009).
M. K. Rai, G. Shahi, and V. Meena, Resource-Effic. Technol. 2, S63 (2016).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ravi, T., Sundararaman, S. Adsorptive Separation of Hexavalent Chromium From its Aqueous and Real Water Mixtures Using Thermally Treated Country Eggshell Coated With Magnetite Nanoparticles. Russ. J. Phys. Chem. B 15, 462–475 (2021). https://doi.org/10.1134/S1990793121030295
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793121030295