Abstract
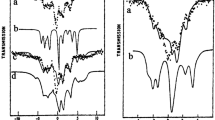
The effect of the 16O ↔ 18O substitution in the coordination sphere of permanganate anion MnO −4 on the chemical shift of 55Mn nuclei have been studied by 17O and 55Mn NMR. Time constants τ n,k of oxygen exchange in the water–permanganate anion system have been estimated. In nearly neutral solutions (pH ≈ 6.8–7.2), the oxygen exchange time is on the order of tens of hours. Bubbling gaseous HCl through this solution for a few seconds leads to the equilibrium distribution of oxygen isotopes in the manganese coordination sphere. The observed temperature dependences of isotope-induced 55Mn NMR shifts in Mn16 O 1844-n O – n (n = 0–4) have been treated as a result of rovibrational averaging of Mn–O bond lengths. The change in the Mn—O bond length in caused by the 16O → 18O isotope substitution is on the order of 10–4 Å.
Similar content being viewed by others
References
K. U. Buckler, A. R. Haase, O. Lutz, M. Muller, and A. Nolle, Z. Naturforsch. A 32, 126 (1977).
A. R. Haase, O. Lutz, M. Muller, and A. Nolle, Z. Naturforsch. A 31, 1427 (1976).
O. Lutz, W. Nepple, and A. Nolle, Z. Naturforsch. A 31, 1046 (1976).
M. Broze and Z. Luz, J. Phys. Chem. 73, 1600 (1969).
S. Dash, S. Patel, and B. K. Mishra, Tetrahedron 65, 707 (2009).
V. P. Tarasov, G. A. Kirakosyan, and K. E. German, Russ. J. Phys. Chem. B 9, 185 (2015).
C. J. Jameson and H. J. Osten, J. Chem. Phys. 81, 4300 (1984).
C. J. Jameson and H. J. Osten, Ann. Rep. NMR Spectrosc. 17, 3 (1986).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.P. Tarasov, G.A. Kirakosyan, 2016, published in Khimicheskaya Fizika, 2016, Vol. 35, No. 8, pp. 15–20.
Rights and permissions
About this article
Cite this article
Tarasov, V.P., Kirakosyan, G.A. 16O/18O oxygen isotope exchange kinetics in MnO −4 as probed by 55Mn NMR. Russ. J. Phys. Chem. B 10, 582–586 (2016). https://doi.org/10.1134/S1990793116040278
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793116040278