Abstract
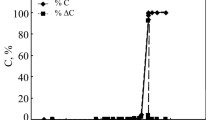
The kinetics of oxidation of a material containing single-layered carbon nanotubes was studied; the material was synthesized by the electric arc method in a water vapor flow at temperatures of 600–1315 K. The dependences of the oxidation rate on the reaction time and conversion were determined.
Similar content being viewed by others
References
A. V. Krestinin, A. V. Raevskii, N. A. Kiselev, G. I. Zvereva, O. M. Zhigalina, and O. I. Kolesova, Chem. Phys. Lett. 381, 529 (2003).
G. Blyholder and H. Eyring, J. Phys. Chem. 63, 693 (1959).
A. V. Krestinin, Ros. Khim. Zh. 48(5), 21 (2004).
A. V. Krestinin, M. B. Kislov, and A. G. Ryabenko, Nanosci. Nanotech. 4, 390 (2004).
E. I. Knerelman, G. I. Zvereva, M. B. Kislov, G. I. Davydova, and A. V. Krestinin, Nanotechnol. Russ. 5, 786 (2010).
E. I. Gusachenko and L. N. Stesik, Russ. J. Phys. Chem. B 1, 292 (2008).
L. N. Gal’perin, Yu. R. Kolesov, and V. A. Zelenov, Izmerit. Tekh., No. 4, 23 (1971).
Ya. B. Zel’dovich, Zh. Fiz. Khim. 13, 163 (1939).
D. A. Frank-Kamenetskii, Diffusion and Heat Transfer in Chemical Kinetics (Nauka, Moscow, 1987) [in Russian].
E. I. Gusachenko and L. N. Stesik, Russ. J. Phys. Chem. B 5, 220 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.I. Gusachenko, M.B. Kislov, L.N. Stesik, A.V. Krestinin, 2015, published in Khimicheskaya Fizika, 2015, Vol. 34, No. 4, pp. 92–98.
Rights and permissions
About this article
Cite this article
Gusachenko, E.I., Kislov, M.B., Stesik, L.N. et al. Kinetics of oxidation of single-walled carbon nanotubes with water vapor. Russ. J. Phys. Chem. B 9, 321–326 (2015). https://doi.org/10.1134/S1990793115020219
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793115020219