Abstract—
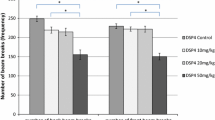
The closed enriched cross maze test was employed as a novel experimental model of the attention deficit disorder (ADD) for evaluation of the behavioral and neurochemical effects of the nootropic drug pantogam (100 mg/kg, intraperitoneally) and atomoxetine hydrochloride (3 mg/kg, intraperitoneally) administered subchronically to CD-1 outbred mice. Two subpopulations of rodents spontaneously diverging in attention to enriched compartments (ED-Low and ED-High), were estimated on the basis of time spent by the mice in the empty or enriched compartments. The ED-Low and ED-High mice did not differ in parameters associated with anxiety, exploratory efficacy, and locomotor activity. Subchronic administration of both drugs in selected doses produced a corrective effect on animal behavior manifested as selective increase in the ED-ratio values in the ED-Low subpopulation (p < 0.05). The radioligand binding assays revealed differences in the distribution of dopamine D2 and GABAB receptors (Bmax) in prefrontal cortex of control ED-Low and ED-High mice. In prefrontal cortex of ED-Low mice treatment with atomoxetine produced a decrease in the Bmax values of D2 receptors by 14%, while pantogam decreased the Bmax values of D2 receptors by 22% (p < 0.05) and increased the Bmax values for GABAB receptor binding by 44%. Thus, subchronic administration of pantogam had a selective corrective effect on the behavior parameters and the density of the studied receptor subtypes in animals having had severe attention deficit in the test.



Similar content being viewed by others
REFERENCES
Am. Psychiatric Assoc., Diagnostic and Statistical Manual of Mental Disorders., 2013, 5th ed.https://doi.org/10.1176/appi.books.9780890425596
Visser, S.N., Danielson, M.L., Bitsko, R.H., et al., J. Am. Acad. Child. Adolesc. Psychiatry, 2014, vol. 53, no. 1, pp. 34–46.https://doi.org/10.1016/j.jaac.2013.09.001
Bykov, Yu.V. and Bekker, R.A., Consilium Medicum, 2018, vol. 20, no. 2, pp. 112–123. https://doi.org/10.26442/2075-1753_2018.2.112-123
Souvorinova, N.Yu., Pediatrics. Suppl. Consilium Medicum, 2018, no. 3, pp. 99–106.
Zavadenko, N.N., Giperaktivnost’ i defitsit vnimaniya v detskom vozraste (Hyperactivity and Childhood Attention-Deficit/Hyperactivity Disorder), Moscow: Urait, 2019.
Wickens, J.R., Hyland, B.I., and Tripp, G., Br. J. Pharmacol., 2011, vol. 164, no. 4, pp. 1107–1128. https://doi.org/10.1111/j.1476-5381.2011.01412.x
Salimov, R.M. and Kovalev, G.I., J. Behav. Brain Sci., 2013, vol. 3, no. 2, pp. 210–216. https://doi.org/10.4236/jbbs.2013.32022
Kovalev, G.I., Salimov, R.M., Sukhorukova, N.A., Kondrakhin, E.A., and Vasileva, E.V., Neurochemical J., 2020, vol. 37, no. 1, pp. 1–9. https://doi.org/10.31857/S1027813320010148
Iversen, L.L. and Glowinski, J., J. Neurochemistry, 1966, vol. 13, no. 8, pp. 655–669. https://doi.org/10.1111/j.1471-4159.1966.tb09874.x
Breese, C.R., Marks, M.J., and Logel, J., J. Pharmacol. Exp. Ther., 1997, vol. 282, no. 1, pp. 7–13.
Sun, W., Ginovart, N., and Ko, F., Mol. Pharmacol., 2003, vol. 63, no. 2, pp. 456–462. https://doi.org/10.1124/mol.63.2.456
Bowery, N.G., Hill, D.R., and Hudson, A.L., Neuropharmacology, 1985, vol. 24, no. 3, pp. 207–210. https://doi.org/10.1016/0028-3908(85)90075-9
Szekely, A.M., Barbaccia, M.L., and Costa, E., J. Pharmacol. Exp. Ther., 1987, vol. 243, no. 1, pp. 155–159.
Waterborg, J.H. and Matthews, H.R., Methods Mol. Bi-ol., 1984, vol. 1, pp. 1–3. https://doi.org/10.1385/0-89603-062-8:1
Gamo, N.J., Wang, M., and Arnsten, A.F.T., J. Am. Acad. Child. Adolesc. Psychiatry, 2010, vol. 49, no. 10, pp. 1011–1023. https://doi.org/10.1016/j.jaac.2010.06.015
Moon, S.J., Kim, Ch.J., and Lee, Y.J., PLoS One, 2014, vol. 9, no. 10, e108918. https://doi.org/10.1371/journal.pone.0108918
Kovalev, G.I., Firstova, Iu.Iu., Abaimov, D.A., and Starikova, N.A., Zh. Nevrol. Psikhiatr. im. S.S. Korsakova, 2012, vol. 112, no. 3, pt 1, pp. 39–43.
Vorobyov, V., Schibaev, N., Kaptsov, V., et al., Brain Res., 2011, vol. 1383, pp. 154–168. https://doi.org/10.1016/j.brainres.2011.01.107
Barry, R., Clarke, A.R., and Johnstone, S.J., Clin. Neurophysiol., 2003, vol. 114, no. 2, pp. 171–183. https://doi.org/10.1016/S1388-2457(02)00362-0
Funding
The study was performed as a part of State Assignment no. 0521-2019-009 “Analysis of receptor mechanisms and search for pharmacological protection of the central nervous system in the case of cerebrovascular and cognitive disorders.”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
The organization of the experiments was in accordance with the ethical standards governing animal experimentation (European Convention for the Protection of Vertebrate Animals Used for Experimental or Other Scientific Purposes: EST no. 123 of March 18, 1986, Strasbourg; “Good Laboratory Practice,” approved by order of the Ministry of Health of Russian Federation no. 199n dated April 1, 2016). The keeping of animals corresponded to the rules of laboratory practice during preclinical studies in the Russian Federation (GOST 33215-2014 and 33216-2014). The experiments were approved by the Commission on Biomedical Ethics of the Zakusov Research Institute of Pharmacology.
Additional information
Translated by A. Medvedev
Rights and permissions
About this article
Cite this article
Kovalev, G.I., Sukhorukova, N.A., Vasileva, E.V. et al. The Influence of Pantogam and Atomoxetine on Attention Stability and Distribution of Dopamine D2 and GABAB Receptors in the Attention Deficit Mouse Model. Biochem. Moscow Suppl. Ser. B 16, 30–37 (2022). https://doi.org/10.1134/S1990750822010048
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990750822010048