Abstract
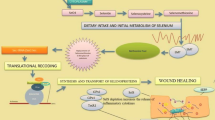
Monocytes and macrophages play a key role in the development of inflammation induced by lipopolysaccharides (LPS), absorbed from the intestine: these cells form reactive oxygen species (ROS) and cytokines, thus initiating the development of oxidative stress, inflammation and/or apoptosis in all types of tissues. In the cells LPS induce an “internal” TLR4-mediated MAP-kinase inflammatory signaling pathway and cytokines through the superfamily of tumor necrosis factor receptor (TNFR) and the “death domain” (DD) initiate an “external” caspase apoptosis cascade or necrosis activation causing necroptosis. Many of the proteins involved in intracellular signaling cascades (MYD88, ASK1, IKKα/β, NF-κB, AP-1) are redox-sensitive and their activity is regulated by antioxidants thioredoxin, glutaredoxin, nitroredoxin, and glutathione. Oxidation of these signaling proteins induced by ROS promotes development of inflammation and apoptosis, and antioxidants, have an opposite effect: they stabilize the signaling cascades, prevent the vicious circle of oxidative stress, accompanied by inflammation and apoptosis. Reduction of non-enzymatic antioxidants (thioredoxin, glutaredoxin, nitroredoxin, glutathione) requires antioxidant (AO) enzymes thioredoxin reductase (TRXR), glutaredoxin reductase (GLRXR), glutathione reductase (GR), while the AO enzymes SOD, catalase, GPX are needed for ROS degradation. The key AO enzymes (TRXR and GPX) are selenium-dependent and therefore selenium deficiency leads to a decrease in the antioxidant defense, development of oxidative stress, inflammation, and/or apoptosis in various cell types. The Nrf2-Keap1 signaling pathway activated by selenium deficiency and/or oxidative stress is necessary to restore redox homeostasis in the cell. Selenium deficiency also influences expression of some genes. Consequently, growth and proliferation of cells, their movement, development, death, and survival, as well as interaction between cells, redox regulation of intracellular signaling cascades of inflammation and apoptosis, depend on the selenium status of the body. Prophylactic administration of selenium-containing preparations (natural and synthetic (organic and inorganic)) is able to normalize activity of AO enzymes and the general status of the body. Organic selenium compounds have a high bioavailability and, depending on their concentration, can act both as selenium donors preventing selenium deficiency and as antitumor drugs due to their toxicity and involvement in regulation of signaling pathways of apoptosis. Known selenorganic compounds diphenyldiselenide and ethaselen share similarity with the Russian organoselenium compound, diacetophenonylselenide (DAPS-25), which serves as a source of bioavailable selenium, exhibits a wide range of biological activity, including antioxidant activity, that governs cell redox balance and regulates inflammation and apoptosis.





Similar content being viewed by others
REFERENCES
Hayakawa, R., Hayakawa, T., Takeda, K., and Ichijo, H., Proc. Jpn. Acad. Ser. B Phys. Biol. Sci., 2012, vol. 88, pp. 434–453. https://doi.org/10.2183/pjab.88.434
Clark, A. and Mach, N., Front Physiol., 2017, vol. 8, 319. https://doi.org/10.3389/fphys.2017.00319
Fan, J., Frey, R.S., and Malik A.B., J. Clin. Invest., 2003, vol. 112, no. 8, pp. 1234–1243. https://doi.org/10.1172/JCI18696
Fedotov, I.V., Rusetskaya, N.Yu., Bobyleva, E.V., and Borodulin, V.B., Patogenez, 2018, vol. 16, no. 3, pp. 14–22. https://doi.org/10.25557/2310-0435.2018.03.14-22
Strandberg, L., Verdrengh, M., Enge, M., Andersson, N., Amu, S., Onnheim, K., Benrick, A., Brisslert, M., Bylund, J., Bokarewa, M., Nilsson, S., and Jansson, J.O., PLoS One, 2009, vol. 4, e7605. https://doi.org/10.1371/journal.pone.0007605
van der Heijden, R.A., Bijzet, J., Meijers, W.C., Yaka-la, G.K., Kleemann, R., Nguyen, T.Q., de Boer, R.A., Schalkwijk, C.G., Hazenberg, B.P., Tietge, U.J., and Heeringa, P., Sci. Rep., 2015, vol. 5, 16474. https://doi.org/10.1038/srep16474
Khoso, P.A., Yang, Z., Liu, C., and Li, S., Biol. Trace Elem. Res., 2015, vol. 167, pp. 48–55. https://doi.org/10.1007/s12011-015-0282-y
Liang, Y., Lin, S.L., Wang, C.W., Yao, H.D., Zhang, Z.W., and Xu, S.W., Biol. Trace Elem. Res., 2014, vol. 160, pp. 41–48. https://doi.org/10.1007/s12011-014-0024-6
Lescure, A., Rederstorff, M., Krol, A., Guicheney, P., and Allamand, V., Biochim. Biophys. Acta, 2009, vol. 1790, pp. 1569–1574. https://doi.org/10.1016/j.bbagen.2009.03.002
Gao, X., Zhang, Z., Li, Y., Shen, P., Hu, X., Cao, Y., and Zhang, N., Biol. Trace Elem. Res., 2016, vol. 172, pp. 449–457. https://doi.org/10.1007/s12011-015-0614-y
Yang, T., Zhao, Z., Liu, T., Zhang, Z., Wang, P., Xu, S., Lei, X.G., and Shan, A., Oncotarget, 2017, vol. 8, pp. 12 428–12 439. https://doi.org/10.18632/oncotarget.14550
Patel, U., Rajasingh, S., Samanta, S., Cao, T., Dawn, B., and Rajasingh, J., Drug Discov. Today, 2017, vol. 22, no. 1, pp. 186–193. https://doi.org/10.1016/j.drudis.2016.08.006
Mills E.L. and O’Neill, L.A., Eur. J. Immunol., 2016, vol. 46, pp. 13–21. https://doi.org/10.1002/eji.201445427
Murphy, M.P., Biochem. J., 2009, vol. 417, pp. 1–13. https://doi.org/10.1042/BJ20081386
Sazanov, L.A., Biochemistry, 2007, vol. 46, pp. 2275–2288. https://doi.org/10.1021/bi602508x
Kussmaul, L. and Hirst, J., Proc. Natl. Acad. Sci. USA, 2006, vol. 103, pp. 7607–7612. https://doi.org/10.1073/pnas.0510977103
Sangiuliano, B., Pérez, N.M., Moreira, D.F., and Belizário, J.E., Mediators Inflamm., 2014, vol. 2014, 821043. https://doi.org/10.1155/2014/821043
Stottmeier, B. and Dick, T.P., Free Radic. Biol. Med., 2016, vol. 101, pp. 93–101. https://doi.org/10.1016/j.freeradbiomed.2016.10.004
Into, T., Inomata, M., Nakashima, M., Shibata, K., Häcker, H., and Matsushita, K., Mol. Cell. Biol., 2008, vol. 28, pp. 1338–1347. https://doi.org/10.1128/MCB.01412-07
Hanschmann, E.-M., Godoy, J.R., Berndt, C., Hudemann, C., and Lillig, C.H., Antioxid. Redox Signal., 2013, vol. 19, pp. 1539–1605. https://doi.org/10.1089/ars.2012.4599
Daily, D., Vlamis-Gardikas, A., Offen, D., Mittelman, L., Melamed, E., Holmgren, A., and Barzilai, A., J. Biol. Chem., 2001, vol. 276, pp. 1335–1344. https://doi.org/10.1074/jbc.M008121200
Hirota, K., Matsui, M., Murata, M., Takashima, Y., Cheng, F.S., Itoh, T., Fukuda, K., and Yodoi, J., Biochem. Biophys. Res. Commun., 2000, vol. 274, pp. 177–182. https://doi.org/10.1006/bbrc.2000.3106
Lorenzen, I., Mullen, L., Bekeschus, S., and Hanschmann, E.-M., Oxid. Med. Cell Longev., 2017, vol. 2017, 8 459 402. https://doi.org/10.1155/2017/8459402
Abais, J.M., Xia, M., Zhang, Y., Boini, K.M., Li, P.-L., Antioxid. Redox Signal., 2015, vol. 22, pp. 1111–1129. https://doi.org/10.1089/ars.2014.5994
Nishiyama, A., Matsui, M., Iwata, S., Hirota, K., Masutani, H., Nakamura, H., Takagi, Y., Sono, H., Gon, Y., and Yodoi, J., J. Biol. Chem., 1999, vol. 274, pp. 21 645–21 650. https://doi.org/10.1074/jbc.274.31.21645
Abderrazak, A., Syrovets, T., Couchie, D., El Hadri, K., Friguet, B., Simmet, T., and Rouis, M., Redox Biol., 2015, vol. 4, pp. 296–307. https://doi.org/10.1016/j.redox.2015.01.008
Sengupta, R., Billiar, T.R., Kagan, V.E., and Stoyanovsky, D.A., Biochem. Biophys. Res. Commun., 2010, vol. 391, pp. 1127–1130. https://doi.org/10.1016/j.bbrc.2009.12.036
Sengupta, R. and Holmgren, A., Biochim. Biophys. Ac-ta, 2012, vol. 1820, no. 6, pp. 689–700. https://doi.org/10.1016/j.bbagen.2011.08.012
Sumbayev, V.V., Arch. Biochem. Biophys., 2003, vol. 415, pp. 133–136.
Kelleher, Z.T., Sha, Y., Foster, M.W., Foster, W.M., Forrester, M.T., and Marshall, H.E., J. Biol. Chem., 2014, vol. 289, pp. 3066–3072. https://doi.org/10.1074/jbc.M113.503938
Mitchell, D.A., Morton, S.U., Fernhoff, N.B., and Marletta, M.A., Proc. Natl. Acad. Sci. USA, 2007, vol. 104, pp. 11 609–11 614. https://doi.org/10.1073/pnas.0704898104
Mitchell, D.A., and Marletta, M.A., Nat. Chem. Biol., 2005, vol. 1, pp. 154–158. https://doi.org/10.1038/nchembio720
Li, H., Wan, A., Xu, G., Ye, D., Acta Biochim. Biophys. Sin., 2013, vol. 45, pp. 153–161. https://doi.org/10.1093/abbs/gms103
Nagarajan, N., Oka, S., and Sadoshima, J., Free Radic. Biol. Med., 2017, vol. 109, pp. 125–131. https://doi.org/10.1016/j.freeradbiomed.2016.12.020
Cardenas-Rodriguez, M., and Tokatlidis, K., FEBS Lett., 2017, vol. 591, pp. 2661–2670. https://doi.org/10.1002/1873-3468.12766
Tagaya, Y., Maeda, Y., Mitsui, A., Kondo, N., Mat-sui, H., Hamuro, J., Brown, N., Arai, K., Yokota, T., Wakasugi, H., EMBO J., 1989, vol. 8, pp. 757–764.
Nakamura, H., Nakamura, K., and Yodoi, J., Annu. Rev. Immunol., 1997, vol. 15, pp. 351–369.
Yamawaki, H. and Berk, B.C., Curr. Opin. Nephrol. Hypertens., 2005, vol. 14, pp. 149–153.
Holmgren, A., Annu. Rev. Biochem., 1985, vol. 54, pp. 237–271.
Holmgren, A. and Lu, J., Biochem. Biophys. Res. Commun., 2010, vol. 396, pp. 120–124. https://doi.org/10.1016/j.bbrc.2010.03.083
Barglow, K.T., Knutson, C.G., Wishnok, J.S., Tannenbaum, S.R., and Marletta, M.A., Proc. Natl. Acad. Sci. USA, 2011, vol. 108, pp. E600–E606. https://doi.org/10.1073/pnas.1110736108
Wu, C., Liu, T., Chen, W., Oka, S., Fu, C., Jain, M.R., Parrott, A.M., Baykal, A.T., Sadoshima, J., and Li, H., Mol. Cell Proteom., 2010, vol. 9, pp. 2262–2275. https://doi.org/10.1074/mcp.M110.000034
Weichsel, A., Brailey, J.L., and Montfort, W.R., Biochemistry, 2007, vol. 46, pp. 1219–1227. https://doi.org/10.1021/bi061878r
Weichsel, A., Kem, M., Montfort, W.R., Protein Sci., 2010, vol. 19, pp. 1801–1806. https://doi.org/10.1002/pro.455
Sengupta, R. and Holmgren, A., Antioxid. Redox Signal., 2013, vol. 18, pp. 259–269. https://doi.org/10.1089/ars.2012.4716
Benhar, M., Forrester, M.T., Hess, D.T., and Stamler, J.S., Science, 2008, vol. 320, pp. 1050–1054. https://doi.org/10.1126/science.1158265
Diamond, A.M., Nutrients, 2015, vol. 7, no. 5, pp. 3938–3948. https://doi.org/10.3390/nu7053938
Lu, J. and Holmgren, A., J. Biol. Chem., 2009, vol. 284, no. 2, pp. 723–727. https://doi.org/10.1074/jbc.R800045200
Hangauer, M.J., Viswanathan, V.S., Ryan, M.J., Bole, D., Eaton, J.K., Matov, A., Galeas, J., Dhruv, H.D., Berens, M.E., Schreiber, S.L., McCormick, F., and McManus, M.T., Nature, 2017, vol. 551, no. 7679, pp. 247–250. https://doi.org/10.1038/nature24297
Lu, J. and Holmgren, A., Free Radic. Biol. Med., 2014, vol. 66, pp. 75–87. https://doi.org/10.1016/j.freeradbiomed.2013.07.036
Pitts, M.W. and Hoffmann, P.R., Cell Calcium, 2018, vol. 70, pp. 76–86. https://doi.org/10.1016/j.ceca.2017.05.001
Zhang, X., Zhang, L., Zhu, J.H., and Cheng, W.H., IUBMB Life, 2016, vol. 68, no. 1, pp. 5–12. https://doi.org/10.1002/iub.1455
Qazi, I.H., Angel, C., Yang, H., Pan, B., Zoidis, E., Zeng, C.J., Han, H., Zhou, G.B., Molecules, 2018, vol. 23, no. 12, pii: E3053. https://doi.org/10.3390/molecules23123053
Rose, A.H. and Hoffmann, P.R., Thromb. Haemost., 2015, vol. 113, no. 3, pp. 494–504. https://doi.org/10.1160/TH14-07-0603
Hawkes, W.C. and Tappel, A.L., Biochim. Biophys. Acta, 1983, vol. 739, pp. 225–234. https://doi.org/10.1016/0167-4781(83)90033-7
Lammi, M.J. and Qu, C., Int. J. Mol. Sci., 2018, vol. 19, no. 9. pii: E2665. https://doi.org/10.3390/ijms19092665
Dixon, S.J., Lemberg, K.M., Lamprecht, M.R., Skouta, R., Zaitsev, E.M., Gleason, C.E., Patel, D.N., Bauer, A.J., Cantley, A.M., Yang, W.S., Morrison, B., 3rd, and Stockwell, B.R., Cell, 2012, vol. 149, pp. 1060–1072. https://doi.org/10.1016/j.cell.2012.03.042
Lu, B., Chen, X.B., Ying, M.D., He, Q.J., Cao, J., and Yang, B., Front. Pharmacol., 2017, vol. 8, 992. https://doi.org/10.3389/fphar.2017.00992
Seibt, T.M., Proneth, B., Conrad M., Free Radic. Biol. Med., 2018, vols. S0891–5849, no. 18, pp. 31 593–31 594. https://doi.org/10.1016/j.freeradbiomed.2018.09.014
Praticò, D., Tangirala, R.K., Rader, D.J., Rokach, J., and FitzGerald, G.A., Nat. Med., 1998, vol. 4, pp. 1189–1192. https://doi.org/10.1038/2685
Monnier, L., Mas, E., Ginet, C., Michel, F., Villon, L., Cristol, J.P., and Colette, C., JAMA, 2006, vol. 295, pp. 1681–1687. https://doi.org/10.1001/jama.295.14.1681
Ranjan, R., Swarup, D., Naresh, R., and Patra, R.C., Vet. Res. Comm., 2005, vol. 29, pp. 27–34.
Bellinger, F.P., Raman, A.V., Reeves, M.A., and Berry, M.J., Biochem. J., 2009, vol. 422, pp. 11–22. https://doi.org/10.1042/BJ20090219
Sordillo, L.M., Vet. Med. Int., 2013, vol. 2013, 154045. https://doi.org/10.1155/2013/154045
Mattmiller, S.A., Carlson, B.A., and Sordillo, L.M., J. Nutr. Sci., 2013, vol. 2, 13. https://doi.org/10.1017/jns.2013.17
Wang, Q., Huang, J., Zhang, H., Lei, X., Du, Z., Xiao, C., Chen, S., and Ren, F., Biol. Trace Elem. Res., 2017, vol. 176, pp. 407–415. https://doi.org/10.1007/s12011-016-0823-z
Tonelli, C., Chio, I.I.C., and Tuveson, D.A., Antioxid. Redox Signal., 2018, vol. 29, no. 17, pp. 1727–1745. https://doi.org/10.1089/ars.2017.7342
Chen, Q.M. and Maltagliati, A.J., Physiol. Genomics, 2018, vol. 50, no. 2, pp. 77–97. https://doi.org/10.1152/physiolgenomics.00041.2017
Jaramillo, M.C. and Zhang, D.D., Genes Dev., 2013, vol. 27, no. 20, pp. 2179–2191. https://doi.org/10.1101/gad.225680.113
Tao, S., Liu, P., Luo, G., Rojo de la Vega, M., Chen, H., Wu, T., Tillotson, J., Chapman, E., and Zhang, D.D., Mol. Cell Biol., 2017, vol. 37, no. 8, pii: e00660-16. https://doi.org/10.1128/MCB.00660-16
Kansanen, E., Kuosmanen, S.M., Leininen, H., and Levonen, A.L., Redox. Biol., 2013, vol. 1, pp. 45–49. https://doi.org/10.1016/j.redox.2012.10.001
Kuhn, A.M., Tzieply, N., Schmid, M.V., von Knethen, A., Namgaladze, D., Yamamoto, M., and Brüne, B., Free Radic. Biol. Med., 2011, vol. 50, pp. 1382–1391.
Zenkov, N.K., Kozhin P.M., Chechushkov, A.V., Martinovich, G.G., Kandalintseva, N.V., and Menshchikova, E.B., Biochemistry (Moscow), 2017, vol. 82, no. 5, pp. 749–759.
Stewart, J.D., Hengstler, J.G., and Bolt, H.M., Arch. Toxicol., 2011, vol. 85, 239. https://doi.org/10.1007/s00204-011-0694-1
Reszka, E., Wieczorek, E., Jablonska, E., Janasik, B., Fendler, W., and Wasowicz, W., J. Trace Elem. Med. Bi-ol., 2015, vol. 30, pp. 102–106. https://doi.org/10.1016/j.jtemb.2014.11.008
Cao, T., Jin, S., Fei, D., Kang, K., Jiang, L., Lian, Z., Pan, S., Zhao, M., and Zhao, M., Inflammation, 2016, vol. 39, pp. 651–662. https://doi.org/10.1007/s10753-015-0290-2
Lv, H., Yu, Z., Zheng, Y., Wang, L., Qin, X., Cheng, G., and Ci, X., Int. J. Biol. Sci., 2016, vol. 12, pp. 72–86. https://doi.org/10.7150/ijbs.13188
Tang, X.I.D., Liu, B., Wang, X., Yu, Q., and Fang, R., Int. J. Mol. Sci., 2018, vol. 19, 848. https://doi.org/10.3390/ijms19030848
Ryoo, I.G., Shin, D.H., and Kang, K.S., Arch. Pharmacal. Res., 2015, vol. 38, pp. 272–281. https://doi.org/10.1007/s12272-014-0380-y
Park, J.S., Jung, J.S., Jeong, Y.H., Hyun, J.W., Le, T.K., and Kim, D.H., J. Neurochem., 2011, vol. 119, pp. 909–919. https://doi.org/10.1111/j.1471-4159.2011.07395.x
Malassagne, B., Ferret, P.J., Hammond, R., Tulliez, M., Bedda, S., Trébéden, H., Jaffray, P., Calmus, Y., Weill, B., and Batteux, F., Gastroenterology, 2001, vol. 121, pp. 1451–1459.
Chiba, T., Takahashi, S., Sato, N., Ishii, S., and Kikuchi, K., Eur. J. Immunol., 1996, vol. 26, pp. 1164–1169. https://doi.org/10.1002/eji.1830260530
Al-Gayyar, M.M., Abdelsaid, M.A., Matragoon, S., Pillai, B.A., and El-Remessy, A.B., Br. J. Pharmacol., 2011, vol. 164, pp. 170–180. https://doi.org/10.1111/j.1476-5381.2011.01336.x
Kataoka, K., Tokutomi, Y., Yamamoto, E., Nakamura, T., Fukuda, M., Dong, Y.F., Ichijo, H., Ogawa, H., and Kim-Mitsuyama, S., J. Hypertens., 2011, vol. 29, pp. 76–84. https://doi.org/10.1097/HJH.0b013e32833fc8b0
Saitoh, M., Nishitoh, H., Fujii, M., Takeda, K., Tobiume, K., Sawada, Y., Kawabata, M., Miyazono, K., and Ichijo, H., EMBO J., 1998, vol. 17, pp. 2596–2606. https://doi.org/10.1093/emboj/17.9.2596
Zheng, L., Jiang, W.D., Feng, L., Wu, P., Tang, L., Kuang, S.Y., Zeng, Y.Y., Zhou, X.Q., and Liu, Y., Fish Shellfish Immunol., 2018, vol. 82, pp. 408–420. https://doi.org/10.1016/j.fsi.2018.08.038
Liu, C., Sun, Z., Xu, Z., Liu, T., Pan, T., and Li, S., Oncotarget, 2017, vol. 8, pp. 58 513–58 525. https://doi.org/10.18632/oncotarget.17222
Li, M.D., Cheng, W.P., Shi, M.X., Ge, T.D., Zheng, X.L., Wu, D.Y., Hu, X.Y., Luo, J.C., Li, F.L., and Li, H., Mol. Med. Rep., 2017, vol. 15, pp. 988–994. https://doi.org/10.3892/mmr.2016.6099
Domracheva, I., Kanepe-Lapsa, I., Jackevica, L., Vasiljeva, J., and Arsenyan, P., Life Sci., 2017, vol. 186, pp. 92–101. https://doi.org/10.1016/j.lfs.2017.08.011
Vunta, H., Davis, F., Palempalli, U.D., Bhat, D., Arner, R.J., Thompson, J.T., Peterson, D.G., Reddy, C.C., and Prabhu, K.S., J. Biol. Chem., 2007, vol. 282, pp. 17 964–17 973. https://doi.org/10.1074/jbc.M703075200
Vunta, H., Belda, B.J., Arner, R.J., Channa Reddy, C., Vanden Heuvel, J.P., and Sandeep Prabhu, K., Mol. Nutr. Food Res., 2008, vol. 52, pp. 1316–1323. https://doi.org/10.1002/mnfr.200700346
Meplan, C., Johnson, I.T., Polley, A.C., Cockell, S., Bradburn, D.M., Commane, D.M., Arasaradnam, R.P., Mulholland, F., Zupanic, A., Mathers, J.C., and Hesketh, J., FASEB J., 2016, vol. 30, no. 8, pp. 2812–2825. https://doi.org/10.1096/fj.201600251R
Radchenko, E.N., Nizov, A.A., Ivanova, A.Yu., Sidorova, Yu.S., and Abramova, L.S., Voprosy Pitaniya, 2016, vol. 85, no. 3, pp. 96–103.
Shadrina, V.D., Potolitzyna, N.N., Parshukova, O.I., Yeseva, T.V., and Boyko, E.R., Ekologiya Cheloveka, 2018, no. 3, pp. 33–38.
Monsen, E.R., J. Am. Diet. Assoc., 2000, vol. 100, pp. 637–640. https://doi.org/10.1016/S0002-8223(00)00189-9
Stoffaneller, R. and Morse, N.L., Nutrients, 2015, vol. 7, pp. 1494–1537. https://doi.org/10.3390/nu7031494
Lippman, S.M., Klein, E.A., Goodman, P.J., Lucia, M.S., Thompson, I.M., Ford, L.G., Parnes, H.L., Minasian, L.M., Gaziano, J.M., Hartline, J.A., Parsons, J.K., Bearden, J.D., 3rd, Crawford, E.D., Goodman, G.E., Claudio, J., Winquist, E., Cook, E.D., Karp, D.D., Walther, P., Lieber, M.M., Kristal, A.R., Darke, A.K., Arnold, K.B., Ganz, P.A., Santella, R.M., Albanes, D., Taylor, P.R., Probstfield, J.L., Jagpal, T.J., Crowley, J.J., Meyskens, F.L., Jr., Baker, L.H., and Coltman, C.A., Jr., JAMA, 2009, vol. 301, pp. 39–51. https://doi.org/10.1001/jama.2008.864
Ip, C. and Ganther, H.E., Cancer Res., 1990, vol. 50, pp. 1206–1211.
Weekley, C.M. and Harris, H.H., Chem. Soc. Rev., 2013, vol. 42, pp. 8870–8894. https://doi.org/10.1039/c3cs60272a
Medina, D., Thompson, H., Ganther, H., and Ip, C., Nutr. Cancer, 2001, vol. 40, pp. 12–17. https://doi.org/10.1207/S15327914NC401_5
Kumar, S., Bjornstedt, M., and Holmgren, A., Eur. J. Biochem., 1992, vol. 207, pp. 435–439.
Bjornstedt, M., Kumar, S., and Holmgren, A., J. Biol. Chem., 1992, vol. 267, pp. 8030–8034.
Gopalakrishna, R., Gundimeda, U., Zhou, S., Zung, K., Forell, K., and Holmgren, A., React. Oxyg. Species (Apex), 2016, vol. 2, pp. 272–289. https://doi.org/10.20455/ros.2016.851
Burk, R.F. and Hill, K.E., Annu. Rev. Nutr., 1993, vol. 3, pp. 65–81. https://doi.org/10.1146/annurev.nu.13.070193.000433
Ganther, H.E., Carcinogenesis, 1999, vol. 20, pp. 1657–1666.
Diwakar, B.T., Korwar, A.M., Paulson, R.F., and Prabhu, K.S., Adv. Cancer Res., 2017, vol. 136, pp. 153–172. https://doi.org/10.1016/bs.acr.2017.07.003
Bi, C.L., Wang, H., Wang, Y.J., Sun, J., Dong, J.S., Meng, X., and Li, J.J., Eur. J. Pharmacol., 2016, vol. 780, pp. 159–165.
Liao, W., Yu, Z., Lin, Z., Lei, Z., Ning, Z., Regenstein, J.M., Yang, J., and Ren, J., Sci. Rep., 2015, vol. 5, 18629. https://doi.org/10.1038/srep18629
Ma, Y.M., Ibeanu, G., Wang, L.Y., Zhang, J.Z., Chang, Y., Dong, J.D., Li, P.A., and Jing, L., BMC Neurosci., 2017, vol. 18, 15. https://doi.org/10.1186/s12868-017-0337-4
Takahashi, A., Masuda, A., Sun, M., Centonze, V.E., and Herman, B., Brain Res. Bull., 2004, vol. 62, pp. 497–504. https://doi.org/10.1016/j.brainresbull.2003.07.009
Spyrou, G., Bjornstedt, M., Kumar, S., and Holmgren, A., FEBS Lett., 1995, vol. 368, pp. 59–63.
Christensen, M.J., Nartey, E.T., Hada, A.L., Legg, R.L., and Barzee, B.R., Nutr. Cancer, 2007, vol. 58, pp. 197–204. https://doi.org/10.1080/01635580701328701
Liu, M., Hu, C., Xu, Q., Chen, L., Ma, K., Xu, N., and Zhu H., Biosci. Rep., 2015, vol. 35, pii: e00256. https://doi.org/10.1042/BSR20150092
Kensler, T.W., Wakabayashi, N., and Biswal, S., Annu. Rev. Pharmacol. Toxicol., 2007, vol. 47, pp. 89–116. https://doi.org/10.1146/annurev.pharmtox.46.120604.141046
Sakurai, T., Kanayama, M., Shibata, T., Itoh, K., Kobayashi, A., Yamamoto, M., and Uchida, K., Chem. Res. Toxicol., 2006, vol. 19, pp. 1196–1204. https://doi.org/10.1021/tx0601105
Youn, B.W., Fiala, E.S., and Sohn, O.S., Nutr. Cancer, 2001, vol. 40, pp. 28–33. https://doi.org/10.1207/S15327914NC401_7
Seo, Y.R., Kelley, M.R., and Smith, M.L., Proc. Natl. Acad. Sci. USA, 2002, vol. 99, pp. 14 548–14 553. https://doi.org/10.1073/pnas.212319799
Constantinescu-Aruxandei, D., Frincu, R.M., Capra, L., and Oancea, F., Nutrients, 2018, vol. 10, no. 10, pii: E1466. https://doi.org/10.3390/nu10101466
Fagan, S., Owens, R., Ward, P., Connolly, C., Doyle, S., and Murphy, R., Biol. Trace Elem. Res., 2015, vol. 166, pp. 245–259. https://doi.org/10.1007/s12011-015-0242-6
Gagandeep, K., Anu, K., and Sodhi, H.S., Food Biochem., 2018, vol. 42, e12467. https://doi.org/10.1111/jfbc.12467
Maseko, T., Callahan, D.L., Dunshea, F.R., Doronila, A., Kolev, S.D., and Ng, K., Food Chem., 2013, vol. 141, pp. 3681–3687. https://doi.org/10.1016/j.foodchem.2013.06.027
Xu, L., Gong, C., Li, G., Wei, J., Wang, T., Meng, W., Shi, M., and Wang, Y., Mol. Med. Rep., 2018, vol. 17, no. 5, pp. 6847–6851. https://doi.org/10.3892/mmr.2018.8641
Santofimia-Castano, P., Izquierdo-Alvarez, A., Plaza-Davila, M., Martinez-Ruiz, A., Fernandez-Bermejo, M., Mateos-Rodriguez, J.M., Salido, G.M., and Gonzalez, A., J. Cell Biochem., 2018, vol. 119, no. 1, pp. 1122–1133. https://doi.org/10.1002/jcb.26280
Wang, L., Yang, Z., Fu, J., Yin, H., Xiong, K., Tan, Q., Jin, H., Li, J., Wang, T., Tang, W., Yin, J., Cai, G., Liu, M., Kehr, S., Becker, K., and Zeng, H., Free Radic. Biol. Med., 2012, vol. 52, no. 5, pp. 898–908. https://doi.org/10.1016/j.freeradbiomed.2011.11.034
Diaz, M., Gonzalez, R., Plano, D., Palop, J.A., Sanmartin, C., and Encio, I., J. Cell Mol. Med., 2018, vol. 22, no. 1, 289–301. https://doi.org/10.1111/jcmm.13318
Rusetskaya, N.Yu., Strukturno-funktsionalnye zakonomernosti biologicheskogo deistviya khalkogenorganicheskikh soedinenii (Structure-Functional Features of Biological Effects of Organochalcogen Compounds). Doctoral dissertation, South Federal University, Rostov-on-Don, 2014.
Rusetskaya, N.Yu., Sovr. Probl. Nauki i Obraz., 2014, vol. 2. URL: www.science-education.ru/116-12266.
Rusetskaya, N.Yu., Borodulin, V.B., Sarattsev, A.V., and Borodulin, Ya.V., Fund.Issl., 2013, vol. 4, pp. 125–129. URL: http://www.fundamental-research.ru/ru/ article/view?id=31111.
Rusetskaya, N.Yu., Borodulin, V.B., Goroshinskaya, I.A., Martyanova, V.A., Borodulin, Ya.V., and Dimidov, A.P., Izvestiya Vuzov. S.-K. region.Estestvennye Nauki, 2013, vol. 2, pp. 52–56.
Borodulin, V.B., Rusetskaya, N.Yu., and Sarattsev, A.V., Antioksidantnaya aktivnost’ sera-, selen- i tellurorganicheskikh soedinenii (Antioxidant Activity of Sulfur-, Selenium-, and Telluroorganic Compounds) Saratov: Saratov Medical University, 2015.
Rusetskaya, N.Yu., Sovr. probl. nauki i obraz., 2014, vol. 3, URL: http://www.science-education.ru/ru/ article/view?id=12752.
Rodionova, T.N., Antipov, V.A., and Lazarev, V.G., Farmakologiya selenorganicheskogo preparata DAFS-25 i ego ispolzovanie v zhivotnovodstve i veterinarii (Pharmacology of the Organoselenium Compound DAPS-25 and Its Application in Veterinary), Saratov: ITs Nauka, 2010.
Rusetskaya, N.Y. and Borodulin, V.B., Biochemistry (Moscow)Supplement Series B: Biomedical Chemistry, 2015, vol. 9, pp. 45–57.
Drevko, Ya.B., Sitnikova, T.S., Burov, A.M., Drevko, B.I., and Shchegolev, S.Yu., Biotekhnologiya, 2015, vol. 31, no. 6, pp. 65–71.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All authors declare that they have no conflict of interest that requires disclosure in this article.
Additional information
Translated by A. Medvedev
Abbreviations used: AP-1—Activator protein-1; ARE—Antioxidant-responsive element; ASK1—Apoptosis signal regulated kinase 1; BAA—biologically active additives; COX—Cyclooxygenase; Cul3—Cullin-3-containing ubiquitin ligase complex E3; DAPS-25—Diacetophenonylidene; DAMP—Damage-associated molecular pattern, DD—Death domen; GCL—Glutamate-cysteine ligase; GPX—Glutathione peroxidase; GR—Glutathione reductase; GST—Glutathione S-transferase; GRX1—Glutaredoxin 1; GLRXR—Glutaredoxin reductase; HMOX—Heme oxygenase 1; IL—Interleukin; Keap1—Kelch-like ECH-associated protein-1; LOX—Lipoxygenase; LPS—Lipopolysaccharides; MAPK—Mitogen activated protein kinase (MAP kinase); MCh ETC—Mitochondrial electron transport chain; MYD88—Myeloid differentiation factor; NF-κB—Nuclear factor-κB, NOS—NO-synthase, NOX—NADP-oxidase; NQO1—NAD(P)H quinone oxidoreductase 1; NP— Nanoparticles; Nrf2—Nuclear factor erythroid-2-related factor 2; NRX—Nucleoredoxin; PAMP—Pathogen-associated molecular pattern; ROS—reactive oxygen species; SOD—Superoxide dismutase; TLR—Toll-like receptor; TNF-α—Tumor necrosis factor-α; TNFR—Tumor necrosis factor receptor; Trnau1ap—tRNA-selenocysteine 1-associated protein; TRX—Thioredoxin, Thioredoxin; TRXR—Thioredoxin reductase.
Rights and permissions
About this article
Cite this article
Rusetskaya, N.Y., Fedotov, I.V., Koftina, V.A. et al. Selenium Compounds in Redox Regulation of Inflammation and Apoptosis. Biochem. Moscow Suppl. Ser. B 13, 277–292 (2019). https://doi.org/10.1134/S1990750819040085
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990750819040085