Abstract
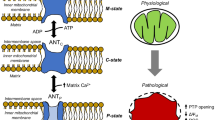
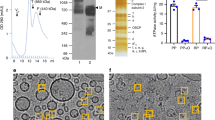
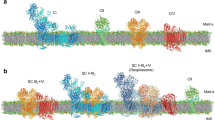
According to previous studies, ADP/ATP carrier (AAC) can possibly exist as a monomer or in a dimer state in the inner mitochondrial membrane; however, the question on its functional oligomeric state is still open. The aim of the present work is to establish the external factors that could control the functional oligomeric state of AAC (i.e., monomer or dimer). The study is based on the results of our previous work, which revealed that the volume regulation system of mitochondria (MVRS) affects the oxidative phosphorylation (OXPHOS) system: MVRS could transfer OXPHOS system functioning in a state of supercomplex. Consequently, one may expect that the volume regulation system could also control the functional state of AAC during phosphorylation. Here, on rat liver mitochondria we show that, depending on the incubation medium tonicity, AAC functions in two different ways: either as a monomer (in hypotonic and isotonic media) or as a dimer (in a hypertonic medium). Thus, the transition between the monomeric and dimeric forms of AAC is regulated by MVRS, as well as by functioning of OXPHOS. We conclude that the structural reorganization of AAC is associated with the entire OXPHOS reorganization into a supercomplex. It was also found that dimerization of AAC can occur not only due to the action of MVRS (in hypotonic media) but also under hypoxic conditions.
Similar content being viewed by others
Abbreviations
- MVRS:
-
mitochondrial volume regulation system
- AAC:
-
ADP/ATP carrier
- OXPHOS:
-
oxidative phosphorylation
- SAN:
-
small angle neutron scattering
- EM:
-
electron microscopy
- CATR:
-
carboxyatractyloside
- HEPES:
-
4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid)
- EDTA:
-
ethylenediaminetetraacetic acid
- BSA:
-
bovine serum albumin
References
Klingenberg M. 2008. The ADP and ATP transport in mitochondria and its carrier. Biochim. Biophys. Acta. 1778, 1978–2021.
Pebay-Peyroula E., Dahout-Gonzalez C., Kahn R., Trezeguet V., Lauquin G.J., Brandolin G. 2003. Structure of mitochondrial ADP/ATP carrier in complex with carboxyatractyloside. Nature. 426, 39–44.
Nury H., Manon F., Arnou B., le Maire M., Pebay-Peyroula E., Ebel C. 2008. Mitochondrial bovine ADP/ATP carrier in detergent is predominantly monomeric but also forms multimeric species. Biochemistry. 47, 12319–12331.
Pebay-Peyroula E., Brandolin G. 2004. Nucleotide exchange in mitochondria: Insight at a molecular level. Curr. Opin. Struct. Biol. 14, 420–425.
Clémençon B. 2012. Yeast mitochondrial interactosome model: Metabolon membrane proteins complex involved in the channeling of ADP/ATP. Int. J. Mol. Sci. 13(2), 1858–1885.
Costantini P., Belzacq A.S., Vieira H.L., Larochette N., de Pablo M.A., Zamzami N., Susin S.A., Brenner C., Kroemer G. 2000. Oxidation of a critical thiol residue of the adenine nucleotide translocator enforces Bcl-2-independent permeability transition pore opening and apoptosis. Oncogene. 19, 307–314.
Majima E., Ikawa K., Takeda M., Hashimoto M., Shinohara Y., Terada H. 1995. Translocation of loops regulates transport activity of mitochondrial ADP/ATP carrier deduced from formation of a specific intermolecular disulfide bridge catalyzed by copper-o-phenanthroline. J. Biol. Chem. 270, 29548–29554.
Hashimoto M., Majima E., Goto S., Shinohara Y., Terada H. 1999. Fluctuation of the first loop facing the matrix of the mitochondrial ADP/ATP carrier deduced from intermolecular cross-linking of Cys56 residues by bifunctional dimaleimides. Biochemistry. 38, 1050–1056.
Kunji E.R., Crichton P.G. 2010. Mitochondrial carriers function as monomers. Biochim.Biophys. Acta. 1797, 817–831.
Bamber L., Harding M., Monne M., Slotboom D.J., Kunji E.R. 2007. The yeast mitochondrial ADP/ATP carrier functions as a monomer in mitochondrial membranes. Proc. Natl. Acad. Sci. USA. 104, 10830–10834.
Crichton P.G., Harding M., Ruprecht J.J., Lee Y., Kunji E.R. 2013. Lipid, detergent, and Coomassie Blue G-250 affect the migration of small membrane proteins in blue native gels: Mitochondrial carriers migrate as monomers not dimers. J. Biol. Chem. 288, 22163–22173.
Bamber L., Slotboom D.J., Kunji E.R. 2007. Yeast mitochondrial ADP/ATP carriers are monomeric in detergents as demonstrated by differential affinity purification. J. Mol. Biol. 371, 388–395.
Faustin B., Rossignol R., Rocher C., Bénard G., Malgat M., Letellier T. 2004. Mobilization of adenine nucleotide translocators as molecular bases of the biochemical threshold effect observed in mitochondrial diseases. J. Biol. Chem. 279, 20 411–20 421.
Krasinskaya I.P., Marshansky V.N., Dragunova S.F., Yaguzhinsky L.S. 1984. Relationships of respiratory chain and ATP-synthetase in energized mitochondria. FEBS Lett. 167, 176–180.
Claypool S.M., Oktay Y., Boontheung P., Loo J.A., Koehler C.M. 2008. Cardiolipin defines the interactome of the major ADP/ATP carrier protein of the mitochondrial inner membrane. J. Cell. Biol. 182, 937–950.
Orlov S.N. 1994. Ion transport across erythrocyte membrane: Mechanisms and volume-dependent regulation. Sov. Sci. Rev. F. Physiol. Gen. Biol. 8, 1–48.
Hoffmann E.K., Lambert I.H., Pedersen S.F. 2009. Physiology of cell volume regulation in vertebrates. Physiol. Rev. 89, 193–277.
Lang F. 2007. Mechanisms and significance of cell volume regulation. J. Am. Coll. Nutr. 26(5 Suppl.), 613S–623S.
Halestrap A.P. 1989. The regulation of the matrix volume of mammalian mitochondria in vivo and in vitro and its role in the control of mitochondrial metabolism. Biochim. Biophys.Acta. 973, 355–382.
Yaguzhinsky L.S., Yurkov V.I., Krasinskaya I.P. 2006. On the localized coupling of respiration and phosphorylation in mitochondria. Biochim. Biophys. Acta. 1757, 408–414.
Johnson D., Lardy H.A. 1967. Isolation of liver or kidney mitochondria. Meth. Enzymol. 10, 94–96.
Chance B., Williams G.R. 1956. The respiratory chain and oxidative phosphorylation. Adv. Enzymol. Relat. Subj. Biochem. 17, 65–134.
Kuklin A.I., Islamov A.K., Gordelii V.I. 2005. Twodetector system for small-angle neutron scattering instrument. Neutron News. 16, 16–18.
Murugova T.N., Gordelii V.I., Kuklin A.I., Kovalev Y.S., Iurkov V.I., Nurenberg A., Islamov A., Iaguzhinskii L.S. 2006. Detection of new two- membrane structures in native mitochondria by the method of small-angle scattering of neutrons. Biophysics. 51, 882–886.
Soloviev A.G., Solovieva T.M., Stadnik A.V., Islamov A.H., Kuklin A.I. 2003. The package for small-angle neutron scattering data treatment. JINR Commun. 10, 2003–2086.
Murugova T.N., Solodovnikova I.M., Yurkov V.I., Gordeliy V.I., Kuklin A.I., Ivankov O.I., Kovalev Yu.S., Popov V.I., Teplova V.V., Yaguzhinsky L.S. 2011. Potentials of small-angle neutron scattering for studies of the structure of “Live Mitochondria”. Neutron News. 22, 11–14.
Murugova T.N., Gordeliy V.I., Kuklin A.I., Solodovnikova I.M., Yaguzhinsky L.S. 2007. Study of three-dimensionally ordered structures of intact mitochondria by small-angle neutron scattering. Crystallography Repts. 52, 521–524.
Reynolds E.S. 1963. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell. Biol. 17, 208–212.
Klingenberg M., Grebe K., Scherer B. 1975. The binding of atractylate and carboxy-atractylate to mitochondria. Eur. J. Biochem. 52, 351–363.
Bauer M., Schubert A., Rocks O., Grimma S. 1999. Adenine nucleotide translocase-1, a component of the permeability transition pore, can dominantly induce apoptosis. J. Cell. Biol. 147, 1493–1502.
Pfeiffer H., Binder H., Klose G., Heremans K. 2003. Hydration pressure and phase transitions of phospholipids. I. Piezotropic approach. Biochim. Biophys. Acta. 1609, 144–147.
Pfeiffer H., Binder H., Klose G., Heremans K. 2003. Hydration pressure and phase transitions of phospholipids. II. Thermotropic approach. Biochim. Biophys. Acta. 1609, 148–152.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.S. Moiseeva, T.N. Murugova, I.M. Vangeli, I.M. Byvshev, S. Ravaud, R.A. Simonyan, V.I. Gordeliy, E. Pebay-Peyroula, L.S. Yaguzhinsky, 2017, published in Biologicheskie Membrany, 2017, Vol. 34, No. 6, pp. 155–163.
Rights and permissions
About this article
Cite this article
Moiseeva, V.S., Murugova, T.N., Vangeli, I.M. et al. On the mechanism and functional significance of the ADP/ATP carrier (AAC) dimerization. Biochem. Moscow Suppl. Ser. A 11, 321–329 (2017). https://doi.org/10.1134/S1990747817040079
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990747817040079