Abstract
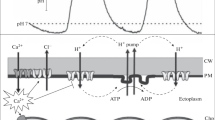
A microelectrode study with Chara corallina cells has shown that post-excitation changes of membrane potential and plasmalemma resistance, induced by the action potential (AP) generation, differ substantially for cell areas producing zones of high and low external pH. In cell regions producing alkaline zones, the AP generation was followed by post-excitation hyperpolarization by about 50 mV, concomitant with four- to eightfold increase in plasmalemma resistance and a considerable drop of pericellular pH. In the acidic areas the post-excitation hyperpolarization was weak or absent, and the membrane resistance showed no significant increase within 1–2 min after AP. The membrane excitation in the acidic zones was accompanied by a noticeable pH increase near the cell surface, indicative of the inhibition of plasma membrane H+ pump. The results suggest that the high local conductance of the plasmalemma is closely related to alkaline zone formation and the depolarized state of illuminated cell under resting conditions. Excitation-induced changes of membrane potential and pH in the cell vicinity were fully reversible, with the recovery period of ∼15 min at a photon flux density of ∼100 μE/(m2 s). At shorter intervals between excitatory stimuli, differential membrane properties of nonuniform regions turned smoothed and could be overlooked. It is concluded that the origin of alkaline zones in illuminated Chara cells cannot be ascribed to hypothetical operation of H+/HCO −3 symport or OH−/HCO −3 antiport.
Similar content being viewed by others
Abbreviations
- AP:
-
action potential
- MP:
-
membrane potential
- pHo :
-
pH of the outer medium near the cell surface
- R m :
-
plasma membrane resistance
References
Tazawa, M., Cell Physiological Aspects of the Plasma Membrane Electrogenic H+ Pump, J. Plant Res., 2003, vol. 116, pp. 419–442.
Plieth, C., Tabrizi, H., and Hansen, U.-P., Relationship between Banding and Photosynthetic Activity in Chara corallina as Studied by the Spatially Different Induction Curves of Chlorophyll Fluorescence Observed by an Image Analysis System, Physiol. Plant., 1994, vol. 91, pp. 205–211.
Bulychev, A.A., Zykov, S.V., Rubin, A.B., and Müller, S.C., Transitions from Alkaline Spots to Regular Bands during pH Pattern Formation at the Plasmalemma of Chara Cells, Eur. Biophys. J., 2003, vol. 32, pp. 144–153.
Lucas, W.J., The Influence of Light Intensity on the Activation and Operation of the Hydroxyl Efflux System of Chara corallina, J. Exp. Bot., 1975, vol. 26, pp. 347–360.
Lucas, W.J., Keifer, D.W., and Sanders, D., Bicarbonate Transport in Chara corallina: Evidence for Cotransport of HCO −3 with H+, J. Membrane Biol., 1983, vol. 73, pp. 263–274.
Lucas, W.J. and Nuccitelli, R., HCO −3 and OH− Transport across the Plasmalemma of Chara: Spatial Resolution Obtained using Extracellular Vibrating Probe, Planta, 1980, vol. 150, pp. 120–131.
Fisahn, J.M., Hansen, U.-P., and Lucas, W.J., Reaction Kinetic Model of a Proposed Plasma Membrane Two-Cycle H+-Transport system of Chara corallina, Proc. Natl. Acad. Sci. USA, 1992, vol. 89, pp. 3261–3265.
Beilby, M.J. and Bisson, M.A., Chara Plasmalemma at High pH: Voltage Dependence of the Conductance at Rest and during Excitation, J. Membrane Biol., 1992, vol. 125, pp. 25–39.
Kitasato, H., Membrane Potential Genesis in Nitella Cells, Mitochondria, and Thylakoids, J. Plant Res., 2003, vol. 116, pp. 401–418.
Beilby, M.J., Mimura, I.T., and Shimmen, T., The Proton Pump, High pH Channels, and Excitation: Voltage Clamp Studies of Intact and Perfused Cells of Nitellopsis obtusa, Protoplasma, 1993, vol. 175, pp. 144–152.
Shimmen, T. and Wakabayashi, A., Involvement of Membrane Potential in Alkaline Band Formation by Internodal Cells of Chara corallina, Plant Cell Physiol., 2008, vol. 49, pp. 1614–1620.
Smith, J.R. and Walker, N.A., Membrane Conductance of Chara Measured in the Acid and Basic Zones, J. Membrane Biol., 1983, vol. 73, pp. 193–202.
Bisson, M.A. and Walker, N.A., The Chara Plasmalemma at High pH. Electrical Measurements Show Specific Passive Uniport of H+ or OH−, J. Membrane Biol., 1980, vol. 56, pp. 1–7.
Yao, X. and Bisson, M.A., Passive Proton Conductance is the Major Reason for Membrane Depolarization and Conductance Increase in Chara buckellii in High-Salt Conditions, Plant Physiol., 1993, vol. 103, pp. 197–203.
Bulychev, A.A., Kamzolkina, N.A., Luengviriya, J., Rubin, A.B., and Müller, S.C., Effect of a Single Excitation Stimulus on Photosynthetic Activity and Light-Dependent pH Banding in Chara Cells, J. Membrane Biol., 2004, vol. 202, pp. 11–19.
Eremin, A., Bulychev, A., Krupenina, N., Mair, T., Hauser, M., Stannarius, R., Müller, S., and Rubin, A. Excitation-Induced Dynamics of External pH Pattern in Chara corallina Cells and Its Dependence on External Calcium Concentration, Photochem. Photobiol. Sci., 2007, vol. 6, pp. 103–109.
Krupenina, N.A. and Bulychev, A.A., Action Potential in a Plant Cell Lowers the Light Requirement for Non-photochemical Energy-Dependent Quenching of Chlorophyll Fluorescence, Biochim. Biophys. Acta, 2007, vol. 1767, pp. 781–788.
Serikawa, K.A., Porterfield, D.M., Smith, P.J.S., and Mandoli, D.F., Calcification and Measurement of Net Proton and Oxygen Flux Reveal Subcellular Domains in Acetabularia acetabulum, Planta, 2000, vol. 211, pp. 474–483.
Kiyosawa, K., Ca2+ and Phosphate Releases from Calcified Chara Cell Walls in Concentrated KCl Solution, J. Exp. Bot., 2001, vol. 52, pp. 223–229.
Berestovsky, G.N. and Kataev, A.A., Voltage-Gated Calcium and Ca2+-Activated Chloride Channels and Ca2+ Transients: Voltage-Clamp Studies of Perfused and Intact Cells of Chara, Eur. Biophys. J., 2005, vol. 34, pp. 973–986.
Lunevsky, V.S., Zherelova, O.M., Vostrikov, I.Y., and Berestovsky, G.N., Excitation of Characeae Cell Membranes as a Result of Activation of Calcium and Chloride Channels, J. Membrane Biol., 1983, vol. 72, pp. 43–58.
Smith, J.R., Effect of a Spatially Inhomogeneous Membrane upon the Measured Electrical Properties of Chara, J. Membrane Biol., 1983, vol. 73, pp. 185–192.
Baudenbacher, F., Fong, L.E., Thiel, G., Wacke, M., Jazbinsek, V., Holzer, J.R., Stampfl, A., and Trontelj, Z., Intracellular Axial Current in Chara corallina Reflects the Altered Kinetics of Ions in Cytoplasm under the Influence of Light, Biophys. J., 2005, vol. 88, pp. 690–697.
Smith, J.R. and Beilby, M.J., Inhibition of Electrogenic Transport Associated with the Action Potential in Chara, J. Membrane Biol., 1983, vol. 71, pp. 131–140.
Shimmen, T., Unique After-hyperpolarization Accompanying Action Potential in Chara globularis, J. Plant Res., 1994, vol. 107, pp. 371–375.
Kitasato, H., The Influence of H+ on the Membrane Potential and Ion Fluxes of Nitella, J. Gen. Physiol., 1968, vol. 52, pp. 60–87.
Bulychev, A.A. and Krupenina, N.A., Facilitated Permeation of Methyl Viologen into Chloroplasts In Situ during Electric Pulse Generation in Excitable Plant Cell Membranes, Biol. Membrany (Rus.), 2008, vol. 25, pp. 343–351 [Transl. version in Biochemistry (Moscow) Suppl. Series A, 2008, vol. 2, pp. 387–394].
Zimmermann, M.R., Maischak, H., Mithöfer, A., Boland, W., and Felle, H., System Potentials, a Novel Electrical Long-Distance Apoplastic Signal in Plants, Induced by Wounding, Plant Physiol., 2009, vol. 149, pp. 1593–1600.
Bulychev, A.A. and Krupenina, N.A., Effects of Plasma Membrane Excitation on Spatially Distributed H+ Fluxes, Photosynthetic Electron Transport and Non-photochemical Quenching in the Plant Cell, Bioelectrochemistry Research Developments, Bernstein, E.M., Ed., New York: Nova Science Publishers, 2008, pp. 159–188.
Lucas, W.J., Photosynthetic Fixation of 14Carbon by Internodal Cells of Chara corallina, J. Exp. Bot., 1975, vol. 26, pp. 331–346.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Bulychev, N.A. Krupenina, 2010, published in Biologicheskie Membrany, 2010, Vol. 27, No. 2, pp. 209–217.
The article was translated by the authors.
Rights and permissions
About this article
Cite this article
Bulychev, A.A., Krupenina, N.A. Inactivation of plasmalemma conductance in alkaline zones of Chara corallina after generation of action potential. Biochem. Moscow Suppl. Ser. A 4, 232–239 (2010). https://doi.org/10.1134/S1990747810020169
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990747810020169