Abstract
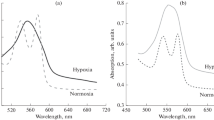
Despite the active research that has taken place on the functional properties of erythrocytes under pathological conditions, this issue is quite relevant. One of the causes of fetal and newborn distress is hypoxia. The consequences of the negative impact of oxygen deficiency on the embryo and fetus can manifest themselves both in utero and after birth, leading to various diseases. The aim of this work was to study the effect of acidosis, as a marker of perinatal hypoxia, on the membrane of erythrocytes in newborns of the early neonatal period. The use of an atomic-force microscope allowed us to obtain cell images and profiles to assess the morphological and structural features of erythrocytes during hypoxia in children in the early neonatal period. It was established that perinatal hypoxia causes changes in the morphology and structures of erythrocyte membranes. The early neonatal period is characterized by changes in morphological forms and the instability of erythrocyte membranes.







Similar content being viewed by others
REFERENCES
Bialevich, E.I, Kostin, D.G, and Slobozhanina, E.I., Caspase-3 activity in human erythrocytes under oxidative stress, Izv. Nats. Akad. Nauk Belarusi, Ser. Biol. Nauk, 2015, no. 2, p. 34.
Binnig, G., Quate, C.F., and Gerber, C., Atomic force microscope, Phys. Rev. Lett., 1986, vol. 56, p. 93.
Chaika, N.A., Danilova, L.A., and Litvinenko, L.A., Preeclampsia and neonatal health, Med.: Teor. Prakt., 2019, no. 4, p. 593.
Chumakova, S.P., Urazova, O.I., Zima, A.P., and Novitskii, V.V., Features of the physiology of erythrocytes. Hemolysis and eryptosis, Gematol. Transfuziol., 2018, vol. 63, p. 343.
Demchenkov, E.L, Nagdalian, A.A., Budkevich, R.O., Oboturova, N.P., and Okolelova, A.I., Usage of atomic force microscopy for detection of the damaging effect of CdCl2 on red blood cells membrane, Ecotoxicol. Environ. Saf., 2020, vol. 208, p. 111683. https://doi.org/10.1016/j.ecoenv.2020.111683
Dodd, J.M., Grivell, R.M., O’Brien, C.M., Dowswell, T., and Deussen, A.R., Prenatal administration of progestogens for preventing spontaneous preterm birth in women with a multiple pregnancy, Cochrane Database Syst. Rev., 2017, vol. 2017, no. 10, p. D012024. https://doi.org/10.1002/14651858.CD012024.pub2
Geekiyanage, N.M., Balanant, M.A., Sauret, E., Saha, S., Flower, R., Lim, C.T., and Gu, Y.T., A coarse-grained red blood cell membrane model to study stomatocyte-discocyte-echinocyte morphologies, PLoS One, 2019, vol. 14, p. e0215447. https://doi.org/10.1371/journal.pone.0215447
Jaferzadeh, K., Sim, M., Kim, N., and Moon, I., Quantitative analysis of three-dimensional morphology and membrane dynamics of red blood cells during temperature elevation, Sci. Rep., 2019, vol. 9, p. 1.
Kamruzzahan, A.S.M., Kienberger, F., Stroh, C.M., Berg, J., Huss, R., Ebner, A., Zhu, R., Rankl, C., Gruber, H.J., and Hinterdorfer, P., Imaging morphological details and pathological differences of red blood cells using tapping-mode AFM, Biol. Chem., 2004, vol. 385, p. 955.
Khadartsev, A.A., Naumova, E.M., Valentinov, B.G., and Grachev, R.V., Erythrocytes and oxidative stress (literature review), Zh. Nov. Med. Tekhnol., 2022, vol. 29, p. 93.
Kim, Y., Park, J., and Kim, M., Diagnostic approaches for inherited hemolytic anemia in the genetic era, Blood Res., 2017, vol. 52, p. 84.
Kodippili, G.C., Spector, J., and Sullivan, C., Imaging of the diffusion of single band 3 molecules on normal and mutant erythrocytes, Blood, 2009, vol. 113, p. 6237.
Kononenko, V.L., Red blood cell flicker. 1. Theoretical models and methods of registration, Biol. Membr., 2009, vol. 26, no. 5, p. 352.
Kozlova, E.K., Chernysh, A.M, Moroz, V.V., and Kuzovlev, A.N., Analysis of nanostructure of red blood cells membranes by space Fourier transform of AFM images, Micron, 2013, vol. 44, p. 218. https://doi.org/10.1016/j.micron.2012.06.012
Kozlova, E., Chernysh, A., Sergunova, V., Gudkova, O., Manchenko, E., and Kozlov, A., Atomic force microscopy study of red blood cell membrane nanostructure during oxidation–reduction processes, J. Mol. Recogn., 2018, vol. 31, no. 10, p. 2724. https://doi.org/10.1002/jmr.2724
Kozlova, E., Chernysh, A., Sergunova, V., Manchenko, E., Moroz, V., and Kozlov, A., Conformational distortions of the red blood cell spectrin matrix nanostructure in response to temperature changes in vitro, Scanning, 2019, vol. 2019, p. 8218912. https://doi.org/10.1155/2019/8218912
Lewis, S.M., Bain, B., and Bates, E., Practical and Laboratory Hematology, Churchill Livingstone, 2006.
Lim, H., Wortis, W.G.M., and Mukhopadhyay, R., Red blood cell shapes and shape transformations. Newtonian mechanics of a composite membrane. Sections 2.5–2.8, in Soft Matter, Hoboken: Wiley-VCH Verlag GmbH & Co., KGaA, 2009, p. 83. https://doi.org/10.1002/9783527623372.ch2a
Melchenko, E.A., Application of atomic-power microscopy at research of biophysical properties of red blood cells membranes, Nauka. Innovatsii. Tekhnol., 2015, no. 3, p. 131.
Moroz, V.V., Golubev, A.M., Afanasyev, A.V., Kuzovlev, A.N., Sergunova, V.A., Gudkova, O.E., and Chernysh, A.M., Structure and function of the erythrocyte in normal and critical conditions, Obshch. Reanimatol., 2012, vol. 8, p. 52.
Mushkambarov, N.N. and Kuznetsov, S.L., Molekulyarnaya biologiya. Uchebnoe posobie dlya studentov med. vuzov (Molecular Biology: Study Guide for Medical Students), Moscow: Med. Inform. Agentstvo, 2007.
Niece, K.L., Boyd, N.K., and Akers, K.S., In vitro study of the variable effects of proton pump inhibitors on voriconazole, Antimicrob. Agents Chemother., 2015, vol. 59, p. 5548. https://doi.org/10.1128/AAC.00884-15
Novitskii, V.V., Ryazantseva, N.V., Stepovaya, E.A., Bystriskii, L.D., and Tkachenko, S.B., Klinicheskii patomorfoz eritrotsita: atlas (Clinical Erythrocyte Pathomorphosis: Atlas), Tomsk, 2003.
Perepelitsa, S.A., Sergunova, V.A., and Gudkova, O.E., The red blood cell membrane of preterm infants in the early neonatal period, Obshch. Reanimatol., 2014a, vol. 10, p. 46.
Perepelitsa, S.A., Sergunova, V.A., Gudkova, O.E., and Alekseyeva, S.V., Features of erythrocyte membranes of premature neonates in multiple pregnancies, Obshch. Reanimatol., 2014b, vol. 10, p. 12.
Perepelitsa, S.A., Sergunova, V.A., and Gudkova, O.E., The effect of perinatal hypoxia on red blood cell morphology in newborns, Obshch. Reanimatol., 2017, vol. 13, p. 14.
Perrone, S., Tataranno, M.L., Stazzoni, G., Del Vecchio, A., and Buonocore, G., Oxidative injury in neonatal erythrocytes, J. Matern. Fetal Neonatal. Med., 2012, vol. 25, p. 104. O’Sullivan, M.P., Looney, A.M., Moloney, G.M., Finder, M., Hallberg, B., Clarke, G., Boylan, G.B., and Murray, D.M., Validation of altered umbilical cord blood microRNA expression in neonatal hypoxic-ischemic encephalopathy, JAMA Neurol., 2019, vol. 76, p. 333. https://doi.org/10.1001/jamaneurol.2018.4182
Ryazantseva, N.V. and Novitskii, V.V., Typical disorders in molecular organization of erythrocyte membrane in patient with somatic and mental pathology, Usp. Fiziol. Nauk, 2004, vol. 35, no. 1, p. 53.
Revin, V.V., Gromova, N.V., Revina, E.S., Prosnik-ova, K.V, Revina, N.V., Bochkareva, S.S., Stepush-kina, O.G., Grunyushkin, I.P., Tairova, M.R., and Incina, V.I., Effects of polyphenol compounds and nitrogen oxide donors on lipid oxidation, membrane-skeletal proteins, and erythrocyte structure under hypoxia, BioMed. Res. Int., 2019, p. 6758017. https://doi.org/10.1155/2019/6758017
Rudenko, S.V., Erythrocyte morphological states, phases, transitions and trajectories, Biochim. Biophys. Acta, Biomembranes, 2010, vol. 1798, p. 1767.
Sergunova, V.A., Kozlova, E.K., Myagkova, E.A., and Chernysh, A.M., In vitro measurement of the elastic properties of the native red blood cell membrane, Obshch. Reanimatol., 2015, vol. 11, p. 39.
Sergunova, V., Leesment, S., Kozlov, A., Inozemtsev, V., Platitsina, P., Lyapunova, S., Onufrievich, A., Polyakov, V., and Sherstyukova, E., Investigation of red blood cells by atomic force microscopy, Sensors (Basel), 2022, vol. 22, p. 2055. https://doi.org/10.3390/s22052055
Shankaran, S., Therapeutic hypothermia for neonatal encephalopathy, Curr. Opin. Pediatr., 2015, vol. 2, p. 152.
Sherstyukova, E.A., Inozemtsev, V.A., Kozlov, A.P., Gudkova, O.E., and Sergunova, V.A., Atomic force microscopy in the assessment of erythrocyte membrane mechanical properties with exposure to various physicochemical agents, Al’manakh Klin. Med., 2021, vol. 49, p. 427.
Starodubtseva, M.N., Voropayev, E.V., Petrenyov, D.R., Mitsura, V.M., and Yegorenkov, N.I., AFM diagnostics of red blood cell pathology based on the physical and mechanical image of the cell membrane, Probl. Zdor. Ekol., 2015, vol. 44, no. 2, p. 99.
Starodubtseva, M.N., Karachrysafi, S., Shkliarava, N.M., Chelnokova, I.A., Kavvadas, D., Papadopoulou, K., Samara, P., Papaliagkas, A., Sioga, A., Komnenou, A., Karampatakis, V., and Papamitsou, T., The Effects of intravitreal administration of antifungal drugs on the structure and mechanical properties peripheral blood erythrocyte surface in rabbits, Int. J. Mol. Sci., 2022, vol. 23, p. 10464. https://doi.org/10.3390/ijms231810464
Steiner, L.A. and Gallagher, P.G., Erythrocyte disorders in the perinatal period, Semin. Perinatol., 2007, vol. 31, p. 254.
Tachev, K.D., Danov, K.D., and Kralchevsky, P.A., On the mechanism of stomatocyte–echinocyte transformations of red blood cells: experiment and theoretical model, Colloids Surf. B: Biointerfaces, 2004, vol. 34, p. 123. https://doi.org/10.1016/j.colsurfb.2003.12.011
Troshkina, N.A., Tsirkin, V.I., and Dvoryanskiy, S.A., Erythrocyte: membrane structure and function, Vyatsk. Med. Vestn., 2007, vol. 3, no. 2, p. 32.
Vaschenko, V.I. and Vil’yaninov, V.N., Eryptosis (quasi-apoptosis) of the human red blood cells. Its role in medicinal therapy, Obz. Klin. Farmakol. Lek. Ter., 2019, vol. 17, p. 5. https://doi.org/10.17816/RCF1735-38
Volodin, N.N., Neonatologiya. Natsional’noe rukovodstvo (Neonatology: National Guidelines. Short Edition), Moscow: GEOTAR-Media, 2019.
Zhang, Y., Zhang, W., Wang, S., Wang, C., Xie, J., Chen, X., Xu, Y., and Mao, P., Detection of erythrocytes in patients with multiple myeloma using atomic force microscopy, Scanning, 2012, vol. 34, p. 295.
Zhong, Q., Inniss, D., Kjoller, K., and Elings, V., Fractured polymer/silica fiber surface studied by tapping mode atomic force microscopy, Surf. Sci. Lett., vol. 290, p. 688.
Funding
This work was financially supported by state order no. FZWM-2020-0010 of the I. Kant Baltic Federal University and state order no. FGWS-2021-0003 of the Federal Research and Clinical Center of Intensive Care Medicine and Rehabilitology (Moscow).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflicts of interest.Statement of compliance with standards of research involving humans as subjects. The study was approved by the Ethics Committee of the Federal Research and Clinical Center of Intensive Care Medicine and Rehabilitology (protocol no. 2/20 dated June 10, 2020) and the Independent Ethics Committee of the Clinical Research Center of I. Kant Baltic Federal University (extract from the minutes of the session of IEC no. 14 dated October 27, 2020) and performed at Maternity Hospital of Kaliningrad Oblast no. 1. Blood sampling was carried out by the staff of the neonatal intensive-care unit as part of a planned clinical examination. No additional blood sampling was performed. Informed consent of the legal representatives of the child to conduct medical manipulations and research is in the hospital neonatal record. All stages of the study comply with the legislation of the Russian Federation, as well as international ethical standards and regulatory documents of research organizations.
Additional information
Translated by D. Novikova
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations: AFM—atomic-force microscope; EDTA—ethylenediaminetetraacetic acid; BE—base excess; НСО3–—bicarbonate ion; pCO2—carbon-dioxide partial pressure in blood.
Rights and permissions
About this article
Cite this article
Denisenko, O.D., Perepelitsa, S.A., Sergunova, V.A. et al. Morphological Changes in Erythrocytes of Newborns after Perinatal Hypoxia. Cell Tiss. Biol. 17, 682–691 (2023). https://doi.org/10.1134/S1990519X23060068
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X23060068