Abstract
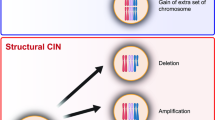
Genomic instability and heterogeneity are the key features of cancer cells that allow their survival under environmental stress. A rapid accumulation of a large number of mutations in a single event due to a massive rearrangement of fragmented chromosomes termed chromothripsis favors cancer progression and resistance to therapy. Complex chromosomal rearrangements that occur upon chromothripsis arise from a random ligation of multiple chromosome fragments by an error-prone NHEJ DNA repair. Here we studied the activation of DDR and NHEJ as markers of genomic instability in HindIIIG-1 cells obtained after depolyploidization of irradiated HindIIIG cells resistant to apoptosis. We also analyzed the potential involvement of chromothripsis-like mechanism and autophagy in the establishment of HindIIIG-1 cell line. Our results demonstrate that HindIIIG cells are characterized by high genomic instability, activation of DDR and NHEJ. Chromosome fragmentation and activation of NHEJ in irradiated HindIIIG and non-treated HindIIIG-1 cells suggest the implication of chromothripsis-like mechanism in the establishment of HindIIIG-1 cell line. We show that unlike HindIIIG cells, HindIIIG-1 cells acquired invasion ability and adhesion-independent cell growth that may indicate their metastatic potential. Degradation of damaged DNA, nuclei and micronuclei in HindIIIG-1 cells by autophagy suggests its role in cell death and survival and thereby in establishment of novel cell line.




Similar content being viewed by others
REFERENCES
Alexander, A., Cai, S.L., Kim, J., Nanez, A., Sahin, M., MacLean, K.H., Inoki, K., Guan, K.L., Shen, J., Person, M.D., Kusewitt, D., Mills, G.B., Kastan, M.B., and Walker, C.L., ATM signals to TSC2 in the cytoplasm to regulate mTORC1 in response to ROS, Proc. Natl. Acad. Sci. U. S. A., 2010, vol. 107, p. 4153. https://doi.org/10.1073/pnas.0913860107
Chellappan, S.P., Hiebert, S., Mudryj, M., Horo-witz, J.M., and Nevins, J.R., The E2F transcription factor is a cellular target for the RB protein, Cell, 1991, vol. 65, p. 1053. https://doi.org/10.1016/0092-8674(91)90557-F
Chitikova, Z.V., Gordeev, S.A., Bykova, T.V., Zubova, S.G., Pospelov, V.A., and Pospelova, T.V., Sustained activation of DNA damage response in irradiated apoptosis-resistant cells induces reversible senescence associated with mTOR downregulation and expression of stem cell markers, Cell Cycle, 2014, vol. 13, p. 1424. https://doi.org/10.4161/cc.28402
Cortés-Ciriano, I., Lee, J.J., Xi, R., Jain, D., Jung, Y.L., Yang, L., Gordenin, D., Klimczak, L.J., Zhang, C.-Z., Pellman, D., PCWAG Structural Variation Working Group, Park, P.J., and PCWAG Consortium, Comprehensive analysis of chromothripsis in 2,658 human cancers using whole-genome sequencing, Nat. Genet., 2020, vol. 52, p. 331. https://doi.org/10.1038/s41588-019-0576-7
Crasta, K., Ganem, N.J., Dagher, R., Lantermann, A.B., Ivanova, E.V., Pan, Y., Nezi, L., Protopopov, A., Chowdhury, D., and Pellman, D., DNA breaks and chromosome pulverization from errors in mitosis, Nature, 2012, vol. 482, p. 53. https://doi.org/10.1038/nature10802
Erenpreisa, J., Salmina, K., Huna, A., Kosmacek, E.A., Cragg, M.S., Ianzini, F., and Anisimov, A.P., Polyploid tumour cells elicit paradiploid progeny through depolyploidizing divisions and regulated autophagic degradation, Cell. Biol. Int., 2011, vol. 35, p. 687. https://doi.org/10.1042/CBI20100762
Erenpreisa, J., Huna, A., Salmina, K., Jackson, T.R., and Cragg, M.S., Macroautophagy-aided elimination of chromatin: sorting of waste, sorting of fate?, Autophagy, 2012, vol. 8, p. 1877. https://doi.org/10.4161/auto.21610
Feng, Z., Zhang, H., Levine, A.J., Jin, S., The coordinate regulation of the p53 and mTOR pathways in cells, Proc. Natl. Acad. Sci. USA, 2005, vol. 102, pp. 8204−8209. https://doi.org/10.1073/pnas.0502857102 González, F., Georgieva, D., Vanoli, F., Shi, Z.D., Stadtfeld, M., Ludwig, T., Jasin, M., Huangfu, D., Homologous recombination DNA repair genes play a critical role in reprogramming to a pluripotent state, Cell Rep, 2013, vol. 3, pp. 651−660. https://doi.org/10.1016/j.celrep.2013.02.005
Guan, J.-L., Simon, A.K., Prescott, M., Menendez, J.A., Liu, F., Wang, C., Wolvetang, E., Vazquez-Martin, A., and Zhang, J., Autophagy in stem cells, Autophagy, 2013, vol. 9, p. 830. https://doi.org/10.4161/auto.24132
Hatch, E.M. and Hetzer, M.W., Linking micronuclei to chromosome fragmentation, Cell, 2015, vol. 161, p. 1502. https://doi.org/10.1016/j.cell.2015.06.005
Hiebert, S.W., Chellappan, S.P., Horowitz, J.M., and Nevins, J.R., The interaction of RB with E2F coincides with an inhibition of the transcriptional activity of E2F, Genes Dev., 1992, vol. 6, p. 177. https://doi.org/10.1101/gad.6.2.177
Howell, W. and Black, D.A., Controller silver staining of nucleolus organizer regions with protective colloidal developer: a one-step method, Experientia, 1980, vol. 36, p. 1014. https://doi.org/10.1007/BF01953855
Jones, M.J.K. and Jallepalli, P.V., Chromothripsis: chromosomes in crisis, Dev. Cell, 2012, vol. 23, p. 908. https://doi.org/10.1016/j.devcel.2012.10.010
Kneissig, M., Keuper, K., de Pagter, M.S., van Roos-malen, M.J., Martin, J., Otto, H, Passerini, V., Campos Sparr, A., Renkens, I., Kropveld, F., Vasudevan, A., Sheltzer, J.M., Kloosterman, W.P., and Storchova, Z., Micronuclei-based model system reveals functional consequences of chromothripsis in human cells, eLife, 2019, vol. 8, e50292. https://doi.org/10.7554/eLife.50292
Levine, B. and Yuan, J., Autophagy in cell death: an innocent convict?, J. Clin. Invest., 2005, vol. 115, p. 2679. https://doi.org/10.1172/JCI26390
Mao, Z., Bozzella, M., Seluanov, A., and Gorbunova, V., DNA repair by nonhomologous end joining and homologous recombination during cell cycle in human cells, Cell Cycle, 2008, vol. 7, p. 2902. https://doi.org/10.4161/cc.7.18.6679
Mathew, R., Kongara, S., Beaudoin, B., Karp, C.M., Bray, K., Degenhardt, K., Chen, G., Jin, S., and White, E., Autophagy suppresses tumor progression by limiting chromosomal instability, Genes Dev., 2007, vol. 21, p. 1367. http://www.genesdev.org/cgi/doi/ 10.1101/gad.1545107
Mehta, A. and Haber, J.E., Sources of DNA double-strand breaks and models of recombinational DNA repair, Cold Spring Harb. Perspect. Biol., 2014, vol. 6, p. a016428. https://doi.org/10.1101/cshperspect.a016428
Menendez, J.A., Vellon, L., Oliveras-Ferraros, C., Cufí, S, and Vazquez-Martin, A., mTOR-regulated senescence and autophagy during reprogramming of somatic cells to pluripotency: a roadmap from energy metabolism to stem cell renewal and aging, Cell Cycle, 2011, vol. 10, p. 3658. https://doi.org/10.4161/cc.10.21.18128
Mijaljica, D. and Devenish, R.J., Nucleophagy at a glance, J. Cell Sci., 2013, vol. 126, p. 4325. https://doi.org/10.1242/jcs.133090
Mizushima, N. and Levine, B., Autophagy in mammalian development and differentiation, Nat. Cell Biol., 2010, vol. 12, p. 823. https://doi.org/10.1038/ncb0910-823
Negrini, S., Gorgoulis, V.G., and Halazonetis, T.D., Genomic instability—an evolving hallmark of cancer, Nat. Rev. Mol. Cell Biol., 2010, vol. 11, p. 220. https://doi.org/10.1038/nrm2858
Ozkinay, C. and Mitelman, F., A simple trypsin-Giemsa technique producing simultaneous G- and C-banding in human chromosomes, Hereditas, 1979, vol. 90, p. 1. https://doi.org/10.1111/j.1601-5223.1979.tb01287.x
Panier, S. and Boulton, S.J., Double-strand break repair: 53BP1 comes into focus, Nat. Rev. Mol. Cell Biol., 2014, vol. 15, p. 7. https://doi.org/10.1038/nrm3719
Pelletier, J., George Thomas, G., and Volarević, S., Ribosome biogenesis in cancer: new players and therapeutic avenues, Nat. Rev. Cancer, 2018, vol. 18, p. 51. https://doi.org/10.1038/nrc.2017.104
Polager, S., Ofir, M., and Ginsberg, D., E2F1 regulates autophagy and the transcription of autophagy genes, Oncogene, 2008, vol. 27, p. 4860. https://doi.org/10.1038/onc.2008.117
Pospelova, T.V., Chitikova, Z.V., and Pospelov, V.A., An integrated approach for monitoring cell senescence, Meth-ods Mol. Biol., 2013, vol. 965, p. 383. https://doi.org/10.1007/978-1-62703-239-1_26
Rappold, I., Iwabuchi, K., Date, T., and Chen, J., Tumor suppressor p53 binding protein 1 (53BP1) is involved in DNA damage-signaling pathways, J. Cell Biol., 2001, vol. 153, p. 613. https://doi.org/10.1083/jcb.153.3.613
Rello-Varona, S., Lissa, D., Shen, S., Niso-Santano, M., Senovilla, L., Mariño, G., Vitale, I., Jemaá, M., Harper, F., Pierron, G., Castedo, M., and Kroemer, G., Autophagic removal of micronuclei, Cell Cycle, 2012, vol. 11, p. 170. https://doi.org/10.4161/cc.11.1.18564
Rogakou, E.P., Pilch, D.R., Orr, A.H., Ivanova, V.S., and Bonner, W.M., DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139, J. Biol. Chem., 1998, vol. 273, p. 5858. https://doi.org/10.1074/jbc.273.10.5858
Stephens, P.J., Greenman, C.D., Fu, B., Yang, F., Bignell, G.R., Mudie, L.J., Pleasance, E.D., Lau, K.W., Beare, D., Stebbings, L.A., McLaren, S., Lin, M.-L., McBride, D.J., Varela, I., Nik-Zainal, S., Leroy, C., et al., Massive genomic rearrangement acquired in a single catastrophic event during cancer development, Cell, 2011, vol. 144, p. 24. https://doi.org/10.1016/j.cell.2010.11.055
Thompson, S.L., Duane, A., and Compton, D.A., Chromosome missegregation in human cells arises through specific types of kinetochore–microtubule attachment errors, Proc. Natl. Acad. Sci. U. S. A., vol. 108, p. 17974. https://doi.org/10.1073/pnas.1109720108
Tripathi, D.N., Chowdhury, R., Trudel, L.J., Tee, A.R., Slack, R.S., Walker, C.L., and Wogan, G.N., Reactive nitrogen species regulate autophagy through ATM-AMPK-TSC2-mediated suppression of mTORC1, Proc. Natl. Acad. Sci. U. S. A., 2013, vol. 110, E2950.https://doi.org/10.1073/pnas.1307736110
Vanzo, R., Bartkova, J., Merchut-Maya, J.M., Hall, A., Bouchal, J., Dyrskjøt, L., Frankel, L.B., Gorgoulis, V., Maya-Mendoza, A., Jäättelä, M., and Bartek, J., Autophagy role(s) in response to oncogenes and DNA replication stress, Cell Death Differ., 2020, vol. 27, p. 1134. https://doi.org/10.1038/s41418-019-0403-9
Yartseva, N.M., Chitikova, Z.V., Bykova, T.V., Zubova, S.G., Kochetkova, E.U, Pospelov, V.A., and Pospelova, T.V., Alterations of karyotype in irradiated apoptosis-resistant HindIIIG cells after prolonged cultivation, Tsitologiia, 2020, vol. 62, no. 12, p. 880.
Zhang, C.-Z., Leibowitz, M.L., and Pellman, D., Chromothripsis and beyond: rapid genome evolution from complex chromosomal rearrangements, Genes Dev., 2013, vol. 27, p. 2513. https://doi.org/10.1101/gad.229559.113
Zhang, C.-Z., Spektor, A., Cornils, H., Francis, J.M., Jackson, E.K., Liu, S., Meyerson, M., and Pellman, D., Chromothripsis from DNA damage in micronuclei, Nature, 2015, vol. 522, pp. 179–184. https://doi.org/10.1038/nature14493
Funding
The work was supported by budgetary funding of Institute of Cytology, Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interests.
The authors did not perform experiments with animals or humans.
Rights and permissions
About this article
Cite this article
Chitikova, Z.V., Yartseva, N.M., Bykova, T.V. et al. Establishment of HindIIIG-1 Cell Line, Obtained after Irradiation of HindIIIG Cells Resistant to Apoptosis, Characterizes by Genomic Instability, Altered DNA Repair Mechanisms and Activation of Autophagy. Cell Tiss. Biol. 15, 370–380 (2021). https://doi.org/10.1134/S1990519X2104009X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X2104009X