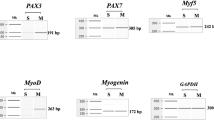
Abstract
Nuclear lamins form nuclear lamina located under the inner nuclear membrane. It was believed that the nuclear lamina plays predominantly a structural role. Recently, its involvement in regulatory processes have been described, e.g., chromatin organization and gene transcription. It is known that mutations in the LMNA gene lead to development of laminopathies, primarily affecting tissues of mesenchymal origin. Today, the mechanisms of the lamina regulation of cell differentiation are largely unknown. In the present work, we studied the effect of LMNA gene mutations on the process of muscle differentiation of primary satellite cells and in С2С12 cell line. The genome of satellite and С2С12 cells was modified by cell transduction via lentiviral constructs encoding LMNA G232E associated with the development of Emery–Dreyfus muscular dystrophy and LMNA R571S associated with the development of dilated cardiomyopathy. Cell morphology was assessed with immunofluorescence, and expression of myogenic genes was analyzed by qPCR. We showed that the analyzed mutations reduced the cell ability to differentiate (to fuse and to form myotubes). We proposed that these mutations enhanced expression of early and reduced expression of late markers of myogenesis. Thus, mutations in nuclear lamins can modify the process of muscle differentiation.
Similar content being viewed by others
Abbreviations
- LMNA :
-
nuclear lamin gene
- LMNA G232E :
-
lamin gene with G232E mutation
- LMNA R571S :
-
lamin gene with R571S mutation
References
Bentzinger, C.F., von Maltzahn, J, and Rudnicki, M.A., Extrinsic regulation of satellite cell specification, Stem Cell Res. Ther., 2010, vol. 1, p. 27.
Bentzinger, C.F., Wang, Y.X., and Rudnicki, M.A., Building muscle: molecular regulation of myogenesis, Cold Spring Harb. Perspect. Biol., 2012, vol. 4, pii a008342.
Bogdanova, M.A., Gudkova, A.Ya., Zabirnik, A.S., Ignatieva, E.V., Dmitrieva, R.I., Smolina, N.A., Kostareva, A.A., and Malashicheva, A.B., Nuclear lamins regulate osteogenic differentiation of mesenchymal stem cells, Cell Tissue Biol., 2014, vol. 8, no. 4, pp. 292–298.
Brosig, M., Ferralli, J., Gelman, L., Chiquet, M., and Chiquet-Ehrismann, R., Interfering with the connection between the nucleus and the cytoskeleton affects nuclear rotation, mechanotransduction and myogenesis, Int. J. Biochem. Cell Biol., 2010, vol. 42, pp. 1717–1728.
Burke, B. and Stewart, C.L., The nuclear lamins: flexibility in function, Nat. Rev. Mol. Cell Biol., 2013, vol. 14, pp. 13–24.
Cadot, B., Gache, V., Vasyutina, E., Falcone, S., Birchmeier, C., and Gomes, E.R., Nuclear movement during myotube formation is microtubule and dynein dependent and is regulated by Cdc42, Par6, and Par3, EMBO Rep., 2012, vol. 13, pp. 741–749.
Cohen, T.V., Gnocchi, V.F., Cohen, J.E., Phadke, A., Liu, H., Ellis, J.A., Foisner, R., Stewart, C.L., Zammit, P.S., and Partridge, T.A., Defective skeletal muscle growth in lamin A/C-deficient mice is rescued by loss of Lap2α, Hum. Mol. Genet., 2013, vol. 22, pp. 2852–2869.
Columbaro, M., Capanni, C., Mattioli, E., Novelli, G., Parnaik, V.K., Squarzoni, S., Maraldi, N.M., and Lattanzi, G., Rescue of heterochromatin organization in Hutchinson–Gilford progeria by drug treatment, Cell Mol. Life Sci., 2005, vol. 62, pp. 2669–2678.
Dubinska-Magiera, M., Zaremba-Czogalla, M., and Rzepecki, R., Muscle development, regeneration and laminopathies: how lamins or lamina-associated proteins can contribute to muscle development, regeneration and disease, Cell Mol. Life Sci., 2013, vol. 70, pp. 2713–2741.
Favreau, C., Higuet, D., Courvalin, J.C., and Buendia, B., Expression of a mutant lamin A that causes Emery–Dreifuss muscular dystrophy inhibits in vitro differentiation of C2C12 myoblasts, Mol. Cell Biol., 2004, vol. 24, pp. 1481–1492.
Frock, R.L., Kudlow, B.A., Evans, A.M., Jameson, S.A., Hauschka, S.D., and Kennedy, B.K., Lamin A/C and emerin are critical for skeletal muscle satellite cell differentiation, Genes Dev., 2006, vol. 20, pp. 486–500.
Furukawa, K., Ishida, K., Tsunoyama, T.A., Toda, S., Osoda, S., Horigome, T., Fisher, P.A., and Sugiyama, S., A-type and B-type lamins initiate layer assembly at distinct areas of the nuclear envelope in living cells, Exp. Cell Res., 2009, vol. 315, pp. 1181–1189.
Håkelien, A.M., Delbarre, E., Gaustad, K.G., Buendia, B., and Collas, P., Expression of the myodystrophic R453W mutation of lamin A in C2C12 myoblasts causes promoterspecific and global epigenetic defects, Exp. Cell Res., 2008, vol. 314, pp. 1869–1880.
Lammerding, J., Schulze, P.C., Takahashi, T., Kozlov, S., Sullivan, T., Kamm, R.D., Stewart, C.L., and Lee, R.T., Lamin A/C deficiency causes defective nuclear mechanics and mechanotransduction, J. Clin. Invest., 2004, vol. 113, pp. 370–378.
de Las Heras, J.I., Meinke, P., Batrakou, D.G., Srsen, V., Zuleger, N., Kerr, A.R., and Schirmer, E.C., Tissue specificity in the nuclear envelope supports its functional complexity, Nucleus, 2013, vol. 4, pp. 460–477.
Malashicheva, A., Kanzler, B., Tolkunova, E., Trono, D., and Tomilin, A., Lentivirus as a tool for lineage-specific gene manipulations, Genesis, 2007, vol. 45, pp. 456–459.
Malashicheva, A.B., Kanzler, B., Tolkunova, E., Trono, D., and Tomilin, A., Lentivirus as a tool for lineagespecific gene manipulations, Tsitologiia, 2008, vol. 50, no. 4, pp. 370–375.
Malashicheva, A.B., Zabirnik, A.S., Smolina, N.A., Dmitrieva, R.I., and Kostareva, A.A., Lamin A/C mutations alter differentiation potential of mesenchymal stem cells, Cell Tissue Biol., 2013, vol. 7, no. 4, pp. 325–328.
Markiewicz, E., Tilgner, K., Barker, N., van, de, Wetering, M., Clevers, H., Dorobek, M., Hausmanowa-Petrusewicz, I., Ramaekers, F.C., Broers, J.L., Blankesteijn, W.M., Salpingidou, G., Wilson, R.G., Ellis, J.A., and Hutchison, C.J., The inner nuclear membrane protein emerin regulates beta-catenin activity by restricting its accumulation in the nucleus, EMBO J., 2006, vol. 25, pp. 3275–3285.
Millay, D.P., O’Rourke, J.R., Sutherland, L.B., Bezprozvannaya, S., Shelton, J.M., Bassel-Duby, R., and Olson, E.N., Myomaker is a membrane activator of myoblast fusion and muscle formation, Nature, 2013, vol. 499, pp. 301–305.
Millay, D.P., Sutherland, L.B., Bassel-Duby, R., and Olson, E.N., Myomaker is essential for muscle regeneration, Genes Dev., 2014, vol. 28, pp. 1641–1646.
Morgan, J.E. and Partridge, T.A., Muscle satellite cells, Int. J. Biochem. Cell Biol., 2003, vol. 35, pp. 1151–1156.
Park, Y.E., Hayashi, Y.K., Goto, K., Komaki, H., Hayashi, Y., Inuzuka, T., Noguchi, S., Nonaka, I., and Nishino, I., Nuclear changes in skeletal muscle extend to satellite cells in autosomal dominant Emery–Dreifuss muscular dystrophy/limb-girdle muscular dystrophy 1B, Neuromuscul. Disord., 2009, vol. 19, pp. 29–36.
Pekovic, V., Harborth, J., Broers, J.L., Ramaekers, F.C., van Engelen, B., Lammens, M., von Zglinicki, T., Foisner, R., Hutchison, C., and Markiewicz, E., Nucleoplasmic LAP2alpha-lamin A complexes are required to maintain a proliferative state in human fibroblasts, J. Cell Biol., 2007, vol. 176, pp. 163–172.
Smolina, N.A., Davidova, A.Ya., Schukina, I.A., Karpushev, A.V., Malashicheva, A.B., Dmitrieva, R.I., and Kostareva, A.A., Comparative assessment of different approaches for obtaining terminally differentiated cell lines, Cell Tissue Biol., 2014, vol. 8, no. 4, pp. 321–329.
Sylvius, N., Bonne, G., Straatman, K., Reddy, T., Gant, T.W., and Shackleton, S., MicroRNA expression profiling in patients with lamin A/C-associated muscular dystrophy, FASEB J., 2011, vol. 25, pp. 3966–3978.
Yin, H., Price, F., and Rudnicki, M.A., Satellite cells and the muscle stem cell niche, Physiol. Rev., 2013, vol. 93, pp. 23–67.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © K.I. Perepelina, N.A. Smolina, A.S. Zabirnik, R.I. Dmitrieva, A.B. Malashicheva, A.A. Kostareva, 2017, published in Tsitologiya, 2017, Vol. 59, No. 2, pp. 117–124.
Rights and permissions
About this article
Cite this article
Perepelina, K.I., Smolina, N.A., Zabirnik, A.S. et al. The role of LMNA mutations in myogenic differentiation of C2C12 and primary satellite cells. Cell Tiss. Biol. 11, 213–219 (2017). https://doi.org/10.1134/S1990519X17030087
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X17030087