Abstract
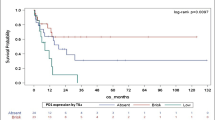
Melanoma is a highly aggressive tumor of melanocytes. The efficacy of different cytotoxic chemotherapy regimens does not exceed 20%. The search for markers for patient sensitivity to chemotherapy provides a rational basis for the development of cytotoxic chemotherapy. In this study, we evaluated six blood lymphocyte parameters (the efficacy of BER and MMR; the expression of MLH1, MSH2, FasR; and cell death) as prognostic markers for melanoma chemotherapy. We found that chemotherapy induced AP sites and ssDNA breaks in lymphocytes repaired through the BER pathway. However, ssDNA breaks were completely repaired after chemotherapy and, therefore, did not contribute to the toxic effect of chemotherapy. dsDNA breaks produced by a functioning MMR system appeared downstream of methylation adducts, which was confirmed by the linear correlation of these parameters in various patients. The number of dsDNA breaks, but not MMR, MLH1, and MSH2 expression, correlated to the efficacy of chemotherapy. A positive correlation was observed between lymphocyte death induced by chemotherapy and the patient’s clinical response, which may show that the nucleotide excision repair (NER) mechanism is implicated in the generation of double-strand breaks and the cytotoxic effect of chemotherapy.
Similar content being viewed by others
References
Al-Attar, A., Gossage, L., Fareed, K.R., Shehata, M., Mohammed, M., Zaitoun, A.M., Soomro, I., Lobo, D.N., Abbotts, R., Chan, S., and Madhusudan, S., Human Apurinic/Apyrimidinic Endonuclease (APE1) Is a Prognostic Factor in Ovarian, Gastro-Oesophageal and Pancreatico-Biliary Cancers, Br. J. Cancer, 2010, vol. 102, pp. 704–709.
Beranek, D.T., Distribution of Methyl and Ethyl Adducts Following Alkylation with Monofunctional Alkylating Agents, Mutat. Res., 1990, vol. 231, pp.11–30.
Friedman, H.S., Keir, S., Pegg, A.E., Houghton, P.J., Colvin, O.M., Moschel, R.C., Bigner, D.D., and Dolan, M.E., O6-Benzylguanine-Mediated Enhancement of Chemotherapy, Mol. Cancer Ther., 2002, vol. 1, pp.943–948.
Gogas, H.J., Kirkwood, J.M., and Sondak, W.K., Chemotherapy for Metastatic Melanoma, Cancer, 2007, vol. 109, pp. 455–464.
Hansen, R.J., Nagasubramanian, R., Delaney, S.M., Samson, L.D., and Dolan, M.E., Role of O6-Methylguanine-DNA Methyltransferase in Protecting from Alkylating Agent-Induced Toxicity and Mutations in Mice, Carcinogenesis, 2007, vol. 28, pp.1111–1116.
Hegde, M.L., Karza, T.K., and Mitra, S., Early Steps in the DNA Base Excision/Single-Strand Interruption Repair Pathway in Mammalian Cells, Cell Res., 2008, vol. 18, pp. 27–47.
Jenkins, G.J.S., Doak, S.H., Johnson, G.E., Quick, E., Waters, E.M., and Parry, J.M., Do Dose Response Thresholds Exist for Genotoxic Alkylating Agents?, Mutagenesis, 2005, vol. 20, pp.389–398.
Konca, K., Lankoff, A., Banasik, A., et al., Across-Platform Public Domain Pc Image-Analysis Program for Comet Assay, Mutat. Res., 2003, vol. 534, pp. 15–20.
Kreklau, E.L., Pollok, R.T., Bailey, B.J., Liu, N., Hartwell, J.R., Williams, D.A., and Erickson, L.C., Hematopoietic Expression of O6-Methylguanine DNA Methyltransferase-P140K Allows Intensive Treatment of Human Glioma Xenografts with Combination O6-Benzylguanine and 1,3-bis-(2-Chloroethyl)-1-Nitrosourea, Mol. Cancer Ther., 2003, vol. 2, pp. 1321–1329.
Liu, L., Nakatsuru, Y., and Gerson, S.L., Base Excision Repair As a Therapeutic Target in Colon Cancer, Clin. Cancer Res., 2002, vol. 8, pp. 2985–2991.
Ma, S., Egyhcozi, S., Ringborg, U., and Hansson, J., Immunohistochemical Analysis of DNA Mismatch Repair Protein and O6-Methylguanine-DNA Methyltransferase in Melanoma Metastases in Relation to Clinical Response to DTIC-Based Chemotherapy, Oncol. Rep., 2002, vol. 9, pp. 1015–1019.
Middleton, M.R., Lee, S.M., Arance, A., Wood, M., Thatcher, N., and Margison, G. P., O6-Methylguanine Formation, Repair Protein Depletion and Clinical Outcome with a 4 hr Schedule of Temozolomide in the Treatment of Advanced Melanoma: Results of a Phase II Study, Int. J. Cancer, 2000, vol. 88, pp. 469–473.
Nadin, S.B., Vargas-Roig, L.M., Drago, G., Ibarra, J., and Ciocca, D.R., DNA Damage and Repair in Peripheral Blood Lymphocytes from Healthy Individuals and Cancer Patients: A Pilot Study on the Implications in the Clinical Response to Chemotherapy, Cancer Lett., 2006, vol. 239, pp. 84–97.
Naumann, S.C., Roos, W.P., Jost, E., Belohlavek, C., Lennerz, V., Schmidt, C.W., Christmann, M., and Kaina, B., Temozolomide- and Fotemustine-Induced Apoptosis in Human Malignant Melanoma Cells: Response Related to MGMT, MMR, DSBs, and p53, British J. Cancer, 2009, vol. 100, pp. 322–333.
Ochs, K. and Kaina, B., Apoptosis Induced by DNA Damage O6-Methylguanine Is Bcl-2 and Caspase-9/3 Regulated and Fas/Caspase-8 Independent, Cancer Res., 2000, vol. 60, pp. 5815–5824.
Roos, W., Baumgartner, M., and Kaina, B., Apoptosis Triggered by DNA Damage O6-Methylguanine in Human Lymphocytes Requires DNA Replication and Is Mediated by p53 and Fas/CD95/Apo-1, Oncogene, 2004, vol. 23, pp. 359–367.
Roos, W.P., Batista, L.F.Z., Naumann, S.C., Wick, W., Weller, M., Menck, C.F.M., and Kaina, B., Apoptosis in Malignant Glioma Cells Triggered by the Temozolomide-Induced DNA Lesion O6-Methylguanine, Oncogene, 2007, vol. 26, pp. 186–197.
Sobol, R.W., Tawbi, H., Jukic, D.M., Mule, K., Mascari, R., and Kirkwood, J.M., Mismatch Repair (MMR) and Base Excision Repair (BER) Protein Expression Correlates with Clinical Response to Dacarbazine (DTIC)/Temozolomide (TMZ) Therapy of Patients with Metastatic Melanoma, J. Clin. Oncol., 2006, vol. 24, pp. 8015–8016.
Toft, N.J., Sansom, O.J., Brookes, R.A., Arends, M.J., Wood, M., Margison, G.P., Winton, D.J., and Clarke, A.R., In vivo Administration of O6-Benzylguanine Does not Influence Apoptosis or Mutation Frequency Following DNA Damage in the Murine Intestine, but Does Inhibit P450-Dependent Activation of Dacarbazine, Carcinogenesis, 2000, vol. 21, pp. 593–598.
Tronov, V.A., Konstantinov, E.M., and Kramarenko, I.I., Role of Excision Mechanisms of DNA Repair in Induction of Apoptosis, Biokhimiya, 2002, vol. 67, no. 7, pp. 882–889 [Biochemistry (Moscow), 2002, vol. 67, no. 7, pp. 730–736].
Tronov, V.A., Kramarenko, I.I., Smirnova, T.D., and Terekhov, S.M., Comparison of Genotoxic and Cytotoxic Effects of N′-methylnitrosourea on Cell Lines Differing in Activity of Mismatch Repair (MMR), Tsitologiya, 2006, vol. 48, no. 1, pp. 19–27.
Tronov, V.A., Loginova, M.Yu., and Kramarenko, I.I., Methylnitrosourea as Challenge Mutagen in Assessment of the DNA Mismatch Repair (MMR) Activity: Association with Some Types of Cancer, Genetika, 2008, vol. 44, no. 5, pp. 686–692 [Russ. J. Genet. (Engl. Transl.), 2008, vol. 44, no. 5, pp. 595–600].
Watson, A.J., Middleton, M.R., McGown, G., Thorncroft, M., Ranson, M., Hersey, P., McArthur, G., Davis, I.D., Thomson, D., Beith, J., Haydon, A., Kefford, R., Lorigan, P., Mortimer, P., Sabharwal, A., Hayward, O., and Margison, G.P., O6-Methylguanine-DNA Methyltransferase Depletion and DNA Damage in Patients with Melanoma Treated with Temozolomide Alone or with Lomeguatrib, British J. Cancer, 2009, vol. 100, pp. 1250–1256.
Wood, R.D., DNA Repair in Eukaryotes, Annu. Rev. Biochem., 1996, vol. 65, pp. 135–167.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.A. Tronov, D.N. Artamonov, M.E. Abramov, L.B. Gorbacheva, 2011, published in Tsitologiya, Vol. 53, No. 1, 2011, pp. 10–16.
Rights and permissions
About this article
Cite this article
Tronov, V.A., Artamonov, D.N., Abramov, M.E. et al. Cellular markers based on DNA repair (BER, MMR) expression of MLH1, MSH2, FasR and death of lymphocytes as predictive parameters for clinical response to chemotherapy of melanoma patients. Cell Tiss. Biol. 5, 29–36 (2011). https://doi.org/10.1134/S1990519X11010159
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X11010159