Abstract
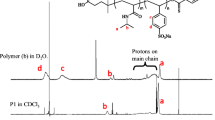
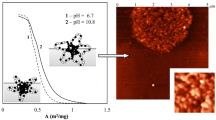
The self-organization of biocompatible amphiphilic block copolymers of D,L-lactide and ethylene oxide with various hydrophobic poly(D,L-lactide) block length in aqueous media has been investigated. It has been shown that the nature of nonselective organic solvent has a considerable effect on the size, size polydispersity, and morphology of micelles of a block copolymer with a long poly(D,L-lactide) block. To elucidate the dependence of properties of block copolymer micelles on the common organic solvent type (acetone, tetrahydrofuran, N,N-dimethylformamide, and acetonitrile) various parameters of the used solvents have been estimated, and correlation between the Flory–Huggins coefficient and the surface tension between the hydrophobic block and the nonselective organic solvent and the parameters of micellar structures has been found.




Similar content being viewed by others
REFERENCES
R. Nagarajan and K. Ganesh, J. Chem. Phys. 90, 5843 (1989).
Y. Mai and A. Eisenberg, Chem. Soc. Rev. 41, 5969 (2012);
B. Jin, Y. Chen, Y. Luo, and X. Li, Chinese J. Chem. 41, 93 (2020);
A. Choicair and A. Eisenberg, Eur. Phys. J. E 10, 37 (2003).
H. Phan, R. I. Minut, P. McCrorie, C. Vasey, R. R. Larder, E. Krumins, M. Marlow, R. Rahman, C. Alexander, V. Taresco, and A. K. Pearce, J. Polym. Sci., Part A: Polym. Chem. 57, 1801 (2019).
E. V. Razuvaeva, A. I. Kulebyakina, D. R. Streltsov, A. V. Bakirov, R. A. Kamyshinsky, N. M. Kuznetsov, S. N. Chvalun, and E. V. Shtykova, Langmuir 34, 15470 (2018).
Y. A. Kadina, E. V. Razuvaeva, D. R. Streltsov, N. G. Sedush, E. V. Shtykova, A. I. Kulebyakina, A. A. Puchkov, D. S. Volkov, A. A. Nazarov, and S. N. Chvalun, Molecules 26, 602 (2021).
A. M. Desyatskova, E. V. Kuznetsova, Y. A. Puchkova, E. V. Yastremsky, A. V. Bakirov, P. V. Dmitryakov, A. I. Buzin, and S. N. Chvalun, Mendeleev Commun. 33, 86 (2023).
N. G. Sedush, Y. A. Kadina, E. V. Razuvaeva, A. A. Puchkov, E. M. Shirokova, V. I. Gomzyak, K. T. Kalinin, A. I. Kulebyakina, and S. N. Chvalun, Nanobiotechnol. Rev. 16, 421 (2021).
I. Ali, F. Kareem, S. Rahim, S. Perveen, S. Ahmed, M. R. Shah, and M. I. Malik, React. Funct. Polym. 150, 104553 (2020).
A. A. Efimova, A. V. Sybachin, S. N. Chvalun, A. I. Kulebyakina, E. V. Kozlova, and A. A. Yaroslavov, Polym. Sci., Ser. B 57 (2), 140 (2015).
A. A. Efimova, S. N. Chvalun, A. I. Kulebyakina, E. V. Kozlova, and A. A. Yaroslavov, Polym. Sci., Ser. A 58 (2), 172 (2016).
F. Alexis, E. Pridgen, L. K. Molnar, and O. C. Farokhzad, Mol. Pharm. 5, 505 (2008).
Z. Lin, L. Xi, S. Chen, J. Tao, Y. Wang, X. Chen, P. Li, Z. Wang, and Y. Zheng, Acta Pharm. Sin. B 11, 1047 (2021).
C. I. C. Crucho and M. T. Barros, J. Mater. Sci. Eng. C 80, 771 (2017).
Y. Yu, L. Zhang, and A. Eisenberg, Macromolecules 31, 1144 (1998).
P. Bhargava, J. X. Zheng, P. Li, R. P. Quirk, F. W. Harris, and S. Z. D. Cheng, Macromolecules 39, 4880 (2006).
T. Riley, S. Stolnik, C. R. Heald, C. D. Xiong, M. C. Garnett, L. Illum, S. S. Davis, S. C. Purkiss, R. J. Barlow, and P. R. Gellert, Langmuir 17, 3168 (2001).
I. A. B. Pijpers, F. Meng, J. C. M. van Hest, and L. K. E. A. Abdelmohsen, Polym. Chem. 11, 275 (2020).
R. Simonutti, D. Bertani, R. Marotta, S. Ferrario, D. Manzone, M. Mauri, M. Gregori, A. Orlando, and M. Masserini, Polymer 218, 123511 (2021).
I. Astafieva, X. F. Zhong, and A. Eisenberg, Macromolecules 26, 7339 (1993).
F. Ahmed and D. E. Discher, J. Controlled Release 96, 37 (2004).
E. Tomaszewska, K. Soliwoda, K. Kadziola, B. Tkacz-Szczesna, G. Celichowski, M. Cichomski, W. Szmaja, and J. J. Grobelny, Nanomater. 2013, 1 (2013).
A. M. de Oliveira, E. Jӓger, A. Jӓger, P. Stepánek, and E. C. Giacomeli, Colloids Surf., A 436, 1092 (2013).
E. V. Kuznetsova, N. M. Kuznetsov, K. T. Kalinin, P. V. Lebedev-Stepanov, A. A. Novikov, and S. N. Chvalun, Colloid J. 84, 704 (2022).
A. D. Glova, S. G. Falkovich, D. I. Dmitrienko, A. V. Lyulin, S. V. Larin, V. M. Nazarychev, M. Karttunen, and S. V. Lyulin, Macromolecules 51, 552 (2018).
J. X. Zheng, H. Xiong, W. Y. Chen, K. Lee, R. M. Van Horn, R. P. Quirk, B. Lotz, E. L. Thomas, A.-C. Shi, and S. Z. D. Cheng, Macromolecules 39, 641 (2006).
R. Nagarajan, J. Colloid Interface Sci. 449, 416 (2015).
G. L. Baker, E. B. Vogel, and M. R. Smith, Polym. Rev. 48, 64 (2008).
C. Witschi and E. Doelker, Eur. J. Pharmacol. Biopharm. 43, 215 (1997).
Properties of Solvents Used in Organic Chemistry http://murov.info/orgsolvents.htm (accessed on April 20, 2023).
R. Subrahmanyam, P. Gurikov, P. Dieringer, M. Sun, and I. Smirnova, Gels 1, 291 (2015).
G. Ovejero, P. Pérez, M. D. Romero, I. Guzmán, and E. Diéz, Eur. Polym. J. 43, 1444 (2007).
A. D. Glova, S. D. Melnikova, A. A. Mercurieva, S. V. Larin, and S. V. Lyulin, Polymers 11, 2056 (2019).
L. J. Garfield and S. E. Petrie, J. Phys. Chem. 68, 1750 (1964).
T. S. Chow, Macromolecules 13, 362 (1980).
Hansen Solubility Parameters. A User’s Handbook, 2nd ed. (CRC Press Taylor and Francis, Boca Raton, 2007).
S. Venkatram, C. Kim, A. Chandrasekaran, and R. Ramprasad, J. Chem. Inf. Model. 59, 4188 (2019).
ACKNOWLEDGMENTS
We are grateful to the Resource Centers “Optics” and “Nanoprobe” at the National Research Center “Kurchatov Institute” for the opportunity to perform DLS and TEM measurements.
Funding
This work was supported by the Russian Science Foundation (project no. 21-73-00071).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by T. Soboleva
Supplementary Information
Rights and permissions
About this article
Cite this article
Kuznetsova, E.V., Shirokova, E.M., Puchkova, Y.A. et al. Study of the Effect of Nonselective Organic Solvent Nature on the Self-Organization of Amphiphilic Block Copolymers of D,L-Lactide and Ethylene Oxide in Aqueous Solution. Polym. Sci. Ser. C 65, 128–142 (2023). https://doi.org/10.1134/S1811238223700352
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1811238223700352