Abstract
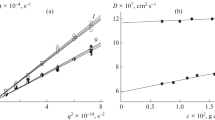
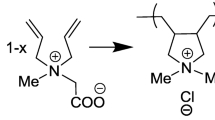
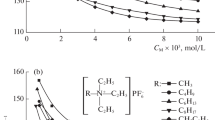
The specific and equivalent conductivity of the diluted aqueous solutions of diallyammonium polyelectrolytes, initial monomers (N,N-diallylammonium trifluoroacetate, N,N-diallyl-N-methylammonium trifluoroacetate, N,N-diallyl-N,N-dimethylammonium chloride), and also potassium trifluoroacetate and trifluoroacetic acid solutions are studied. The limiting ionic mobility of diallylammonium cations and trifluoroacetate anion are found. The regularities of concentration changes in the equivalent conductance of polyelectolytes solutions are established. The degree of dissociation of diallyammonium polymers is shown to depend both on the counterion nature and on the amine structure.
Similar content being viewed by others
References
L. M. Timofeeva, Yu. A. Vasilieva, N. A. Klescheva, and D. A. Topchiev, Russ. Chem. Bull 48 (5), 856 (1999).
L. M. Timofeeva, Yu. A. Vasilieva, N. A. Klescheva, G. L. Gromova, and D. A. Topchiev, Int. J. Quantum Chem. 88 (5), 531 (2002).
L. M. Timofeeva, Yu. A. Vasilieva, N. A. Klescheva, G. L. Gromova, G. I. Timofeeva, A. I. Rebrov, and D. A. Topchiev, Macromol. Chem. Phys. 203 (16), 2296 (2002).
L. M. Timofeeva, N. A. Klescheva, Yu. A. Vasilieva, G. L. Gromova, G. I. Timofeeva, and M. P. Filatova, Polym. Sci., Ser. A 47 (6), 551 (2005).
L. M. Timofeeva, N. A. Klescheva, A. F. Moroz, and L. V. Didenko, Biomacromolecules 10 (11), 2976 (2009).
L. M. Timofeeva, N. A. Klescheva, M. O. Shleeva, M. P. Filatova, Yu. A. Simonova, Yu. A. Ermakov, and A. S. Kaprelyants, Appl. Microbiol. Biotechnol. 99 (6), 2557 (2015).
D. C. Crick, L. E. Quadri, and P. J. Brennan, in Handbook of Tuberculosis: Molecular Biology and Biochemistry, Ed. by S. H. E. Kaufmann and R. Rubin (Wiley-VCH, Weinheim, 2008), p. 1.
S. S. Chavadi, K. C. Onwueme, U. R. Edupuganti, J. Jerome, D. Chatterjee, C. E. Soll, and L. E. Quadri, Microbiology 158, 1379 (2012).
D. A. Enarson, in Development of New Antituberculosis Drugs, Ed. by W. W. Yew (Nova Sci. Publ., New York, 2006), p. 1.
R. M. Fuoss and U. P. Strauss, J. Polym. Sci. 3 (2), 246 (1948).
H. Eisenberg, J. Polym. Sci. 30 (121), 47 (1958).
G. S. Manning, J. Phys. Chem. 85, 1506 (1981).
C. Wandrey, Langmuir 15, 4069 (1999).
C. Wandrey, D. Hunkeler, U. Wendler, and W. Jaeger, Macromolecules 33, 7136 (2000).
D. A. Topchiev, G. T. Bikasheva, A. I. Martynenko, N. N. Kaptsov, L. A. Gudkova, and V. A. Kabanov, Vysokomol. Soedin., Ser. B 22 (4), 269 (1980).
L. I. Antropov, Theoretical Electrochemistry (Univ. Press of the Pacific, Honolulu, HI, 2001).
M. G. Harrisasn and J. B. Milne, Can. J. Chem. 49 (11), 1888 (1971).
A. K. Lyashchenko and A. S. Lileev, J. Chem. Eng. Data 55, 2008 (2010).
A. S. Lileev and A. K. Lyashchenko, J. Mol. Liq. 91, 21 (2009).
R. Buchner, C. Holz, J. Stauber, and J. Barthel, Phys. Chem. Chem. Phys. 4, 2169 (2002).
A. Lileev, D. Loginova, A. Lyashchenko, L. Timofeeva, and N. Kleshcheva, J. Mol. Liq. 131–132 (1), 101 (2007).
L. M. Timofeeva, N. A. Kleshcheva, D. V. Loginova, A. S. Lileev, and A. K. Lyashchenko, Polym. Sci., Ser. A 50 (3), 273 (2008).
F. Bordi, C. Cametti, and T. Gili, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys. 66, 021803 (2002).
A. Y. Grosberg and A. R. Khokhlov, in Statistical Physics of Macromolecules (AIP Press, NewYork, 1994).
A. V. Dobrynin and M. Rubinstein, Macromolecules 34, 1964 (2001).
A. Deshkovski, S. Obukhov, and M. Rubinstein, Phys. Rev. Lett. 86 (2001).
T. Odijk, Macromolecules 12, 688 (1979).
Q. Liao, A. V. Dobrynin, and M. Rubinstein, Macromolecules 36, 3399 (2003).
S. Miertus, E. Scrocco, and J. Tomasi, Chem. Phys. 55 (1), 117 (1981).
G. E. Chudinov, D. V. Napolov, and M. V. Basilevsky, Chem. Phys. 160, 41 (1992).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.M. Timofeeva, I.V. Balakaeva, A.S. Lileev, Yu.A. Simonova, A.K. Lyashchenko, 2017, published in Vysokomolekulyarnye Soedineniya, Seriya C, 2017, Vol. 59, No. 1, pp. 149–156.
Rights and permissions
About this article
Cite this article
Timofeeva, L.M., Balakaeva, I.V., Lileev, A.S. et al. General trends in concentration and temperature behavior of equivalent conductance of N,N-diallylammonium polymers aqueous solutions: Effect of counterion nature and amine structure. Polym. Sci. Ser. C 59, 141–148 (2017). https://doi.org/10.1134/S1811238217010131
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1811238217010131