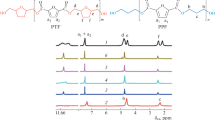
Abstract
Binary thermotropic fully aromatic copolyesters based on 4-hydroxybenzoic and 4'-hydroxybiphenyl-4-carboxylic acids and ternary copolyesters of 4-hydroxybenzoic, 4'-hydroxybiphenyl-4-carboxylic, and 3‑hydroxybenzoic acids containing 60–80% of 4-hydroxybenzoic acid units are synthesized by the melt polycondensation of corresponding acetyl derivatives. The melting temperature of the copolyesters is below 400°С, and the most promising among them melt at temperatures below 340°С which allows their melt processing by extrusion, molding, and injection molding. The tensile properties of some copolyesters are comparable with the corresponding mechanical properties of such commercially available analogs as Vectra A‑950 and Zenite HX-8000. All the synthesized copolyesters are stable in an inert atmosphere at least up to 450°С. The thermal stability of the copolyesters slightly decreases with an increase in the content of 3‑hydroxybenzoic acid units.




Similar content being viewed by others
REFERENCES
F. C. Bawden and N. W. Pirie, Proc. R. Soc. London. Ser. B 123 (832), 274 (1937).
A. Elliott and E. J. Ambrose, Discuss. Faraday Soc. 9, 246 (1950).
S. L. Kwolek, P. W. Morgan, J. R. Schaefgen, and L. W. Gulrich, Macromolecules 10 (6), 1390 (1977)
S. L. Kwolek, U.S. Patent No. 3 671 542 (1972).
Lyu Xiaolin, Xiao Anqi, Shi Dong, Li Yujie, Shen Zhihao, Chen Er-Qiang, Zheng Shijun, Fan Xing-He, and Zhou Qi-Feng, Polymer 202, 122740 (2020).
V. G. Kulichikhin, A. Ya. Malkin, and S. P. Papkov, Vysokomol. Soedin., Ser. A 26 (3), 451 (1984).
T. R. Deberdeev, A. I. Akhmetshina, L. K. Karimova, E. K. Ignat’eva, R. Ya. Deberdeev, and A. A. Berlin, Polym. Sci., Ser. C 62 (2), 145 (2020).
H. S. Han and P. K. Bhowmik, Prog. Polym. Sci. 22 (7), 1431 (1997).
J. I. Jin and C. S. Kang, Prog. Polym. Sci. 22 (5), 937 (1997).
V. G. Kulichikhin and N. A. Pláte, Vysokomol. Soedin., Ser. A 33 (1), 3 (1991).
W. J. Jackson, Mol. Cryst. Liq. Cryst., Sect. B: Nonlinear Opt. 169 (1), 23 (1989).
J. Economy, Mol. Cryst. Liq. Cryst., Sect. B: Nonlinear Opt. 169 (1), 1 (1989).
S. G. Cottis, J. Economy, and L. C. Wohrer, U.S. Patent No. 3 975 487 (1976).
W. Volksen, Jr. Lyerla, J. Economy, and B. Dawson, J. Polym. Sci., Polym. Chem. Ed. 21 (8), 2249 (1983).
A. Konishi, M. Hamaguchi, and H. Umetsu, U.S. Patent No. 0145380 (2016).
W. J. Jackson and H. F. Kuhfuss, J. Polym. Sci., Polym. Chem. Ed. 34 (15), 3031 (1996).
W. J. Jackson and H. F. Kuhfuss, U.S. Patent No. 3 804 805 (1974).
G. W. Calundann, U.S. Patent, No. 4 161 470 (1979).
N. A. Pláte, V. G. Kulichikhin, and R. V. Talroze, Pure Appl. Chem. 63 (7), 925 (1991).
B. A. Yul’chibaev, Z. B. Artykova, E. U. Urinov, N. R. Ashurov, B. Kh, Strelets, and V. G. Kulichikhin, Vysokomol. Soedin., Ser. B 34 (5), 23 (1992).
B. A. Yul’chibaev, A. N. Pefilov, E. U. Urinov, N. R. Ashurov, B. Kh. Strelets, and V. G. Kulichikhin, Vysokomol. Soedin., Ser. A 34 (11), 69 (1992).
B. A. Yul’chibaev, A. N. Pefilov, B. Kh. Strelets, and V. G. Kulichikhin, Vysokomol. Soedin., Ser. B 37 (1), 166 (1995).
Russell Gilkey and John R. Caldwell, J. Appl. Polym. Sci. 2 (5), 198 (1959).
Katherine V. Heifferon, Glenn A. Spiering, Samantha J. Talley, Maruti Hegde, Robert B. Moore, S. Richard Turner, and Timothy E. Long, Polym. Chem. 10 (31), 4287 (2019).
Pavel A. Mikhaylov, Kirill V. Zuev, Marina P. Filatova, Boris Kh. Strelets, and Valery G. Kulichikhin, Polymers 13 (11), 1720 (2021).
P. A. Mikhailov, A. G. Kalita, K. V. Zuev, D. V. Kramarev, O. B. Ushakova, and V. G. Kulichikhin, Polym. Sci., Ser. B 63 (6), 745 (2021).
Hans R. Kricheldorf and G. Schwarz, Polymer 25 (4), 520 (1984).
D. K. Yang and W. R. Krigbaum, J. Polym. Sci., Polym. Phys. Ed. 27 (4), 819 (1989).
Bryan B. Sauer, Richard Beckerbauer, and Lixiao Wang, J. Polym. Sci., Polym. Phys. Ed. 31 (12), 1861 (1993).
Z. Yerlikaya, S. Aksoy, and E. Bayramli, J. Polym. Sci., Polym. Chem. Ed. 39 (19), 3263 (2001).
Z. Yerlikaya, S. Aksoy, and E. Bayramli, J. Appl. Polym. Sci. 90 (12), 3260 (2003).
Gert Schwarz and Hans R. Kricheldorf, Makromol. Chem., Rapid Commun. 9 (10), 717 (1988).
Chen Qian and Donald G. Baird, Polymer 102, 63 (2016).
Funding
This work was performed within the framework of State Assignment for Topchiev Institute of Petrochemical Synthesis, Russian Academy of Sciences.
This work was supported by the Russian Science Foundation (project no. 17-79-30108).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by T. Soboleva
Rights and permissions
About this article
Cite this article
Mikhaylov, P.A., Kalita, A.G. & Kulichikhin, V.G. Synthesis of New Thermotropic Fully Aromatic Copolyesters from Hydroxybenzoic and Hydroxybiphenylcarboxylic Acids. Polym. Sci. Ser. B 64, 393–401 (2022). https://doi.org/10.1134/S1560090422700282
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090422700282