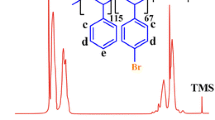
Abstract
The capacity of the coupling reactions of polystyrene bearing an ω-terminal bromine group in the presence of various nitrones and nitroso compounds has been disclosed. As exemplified by the coupling using С-phenyl-N-tert-butylnitrone, acetonitrile and tetrahydrofuran have been found the best solvents of the probed series (toluene, ethyl acetate, benzene, acetonitrile, and tetrahydrofuran). The said solvents have revealed the strongest coordination ability towards the catalytical system based on copper(I) bromide and have afforded high coupling degree (above 90%) preserving low polydispersity of the synthesized samples. It has been shown that the nitroso compounds involved in the coupling with styrene favor the formation of terminal nitroxyl fragments. In contrast to nitroso compounds, the nitrones have afforded symmetrical high-molecular alkoxyamines bearing a nitroxyl fragment in the middle of the polymer chain.






Similar content being viewed by others
REFERENCES
Reversible Deactivation Radical Polymerization: Mechanisms and Synthetic Methodologies, Ed. by K. Matyjaszewski, H. Gao, B. S. Sumerlin, and N. V. Tsarevsky (Am. Chem. Soc., Washington, 2018).
Nitroxide Mediated Polymerization: From Fundamentals to Applications in Materials Science, Ed. by D. Gigmes (The Royal Society of Chemistry, UK, 2015).
G. A. Moad, J. Polym. Sci., Part A: Polym. Chem. 57 (3), 216 (2019).
S. Coiai, E. Passaglia, and F. Cicogna, Polym. Int. 68 (1), 27 (2019).
T. G. Ribelli, F. Lorandi, M. Fantin, and K. Matyjaszewski, Macromol. Rapid Commun. 40 (1) Art. 1800616 (2018).
X. Pan, M. Fantin, F. Yuan, and K. Matyjaszewski, Chem. Soc. Rev. 47 (14), 5457 (2018).
D. F. Grishin and I. D. Grishin, Russ. Chem. Rev. 90 (2), 231 (2021).
A. Anastasaki, J. Willenbacher, C. Fleischmann, W. R. Gutekunst, and C. J. Hawker, Polym. Chem. 8 (4), 689 (2017).
G. Wang and J. Huang, Polym. Chem. 5 (2), 277 (2014).
Z. N. Zhang, G. W. Wang, and J. L. Huang, J. Polym. Sci., Part A: Polym. Chem. 49 (13), 2811 (2011).
E. H. H. Wong, C. Boyer, M. H. Stenzel, C. Barner-Kowollik, and T. Junkers, Chem. Commun. 46 (11), 1959 (2010).
E. H. H. Wong, M. H. Stenzel, T. Junkers, and C. Barner-Kowollik, Macromolecules 43 (8), 3785 (2010).
E. H. H. Wong, O. Altintas, M. H. Stenzel, C. Barner-Kowollik, and T. Junkers, Chem. Commun. 47 (19), 5491 (2011).
L. Barner, A. S. Quick, A. P. Vogt, V. Winkler, T. Junkers, and C. Barner-Kowollik, Polym. Chem. 3 (8), 2266 (2012).
C. Detrembleur, A. Debuigne, O. Altintas, M. Conradi, E. H. H. Wong, C. Jerome, C. Barner-Kowollik, and T. Junkers, Polym. Chem. 3 (1), 135 (2012).
A. F. Voter, E. S. Tillman, P. M. Findeis, and S. C. Radzinski, ACS Macro Lett. 1 (8), 1066 (2012).
S. C. Blackburn, K. D. Myers, and E. S. Tillman, Polymer 68, 284 (2015).
M. M. Arce, C. W. Pan, M. M. Thursby, J. P. Wu, E. M. Carnicom, and E. S. Tillman, Macromolecules 49 (20), 7804 (2016).
J. P. Wu, C. W. Pan, K. E. Heiler, M. E. Ching, and E. S. Tillman, Polymer 127, 66 (2017).
M. Du, C. Deng, X. Wu, H. Liu, and H. Liu, Macromol. Chem. Phys. 218 (15), Art. 1700069 (2017).
E. V. Kolyakina, F. H. Shoipova, A. B. Alyeva, and D. F. Grishin, Russ. Chem. Bull. 70 (9), 1736 (2021).
V. Sciannamea, R. Jérôme, and C. Detrembleur, Chem. Rev. 108 (3), 1104 (2008).
E. V. Kolyakina and D. F. Grishin, Usp. Khim. 78 (6), 579 (2009).
D. F. Grishin, L. L. Semenycheva, and E. V. Kolyakina, Dokl. Akad. Nauk 362 (5), 634 (1998).
E. V. Kolyakina, L. L. Semenycheva, and D. F. Grishin, Polym. Sci., Ser. A 43 (12), 1223 (2001).
M. V. Pavlovskaya, E. V. Kolyakina, V. V. Polyanskova, L. L. Semenycheva, and D. F. Grishin, Zh. Prikl. Khim. 75, 1905 (2002).
M. Yu. Zaremski, A. P. Orlova, E. S. Garina, A. V. Olenin, M. B. Lachinov, and V. B. Golubev, Polym. Sci., Ser. A 45 (6), 502 (2003).
M. Yu. Zaremski, A. L. Reznichenko, Yu. V. Grinevich, E. S. Garina, M. B. Lachinov, and V. B. Golubev, Polym. Sci., Ser. A 47 (6), 908 (2005).
V. Sciannamea, C. Guerrero-Sanchez, U. S. Schubert, J.-M. Catala, R. Jerome, and C. Detrembleur, Polymer 46 (23), 9632 (2005).
V. Sciannamea, J. M. Catala, R. Jerome, and C. Detrembleur, J. Polym. Sci., Part A: Polym. Chem. 45 (7), 1219 (2007).
D. F. Grishin, E. V. Kolyakina, M. V. Pavlovskaya, M. A. Lazarev, and A. A. Shchepalov, in Controlled Living Radical Polymerization: Progress in RAFT, DT, NMP and OMRP. ACS Symp. Ser., Ed. by K. Matyjiaszewski (Am. Chem. Soc., Washington, 2009), Vol. 1024, p. 95.
E. V. Kolyakina, A. B. Alyeva, E. V. Sazonova, A. A. Shchepalov, and D. F. Grishin, Izv. Akad. Nauk, Ser. Khim., No. 8, 1585 (2019).
E. V. Kolyakina, A. B. Alyeva, E. V. Sazonova, E. A. Zakharychev, and D. F. Grishin, Polym. Sci., Ser. B 62, 328 (2020).
E. V. Kolyakina, A. B. Alyeva, E. A. Zakharychev, and D. F. Grishin, Izv. Akad. Nauk, Ser. Khim. 70 (10), 1997 (2021).
E. H. H. Wong, T. Junkers, and C. Barner-Kowollik, J. Polym. Sci., Part A: Polym. Chem. 46 (21), 7273 (2008).
T. Junkers, E. H. H. Wong, M. H. Stenzel, and C. Barner-Kowollik, Macromolecules 42 (14), 5027 (2009).
A. Weissberger, E. Proskauer, J. Riddick, and E. Toops, Organic Solvents (Interscience Publ., New York, 1955).
R. R. Holmes, R. P. Bayer, and A. Simple, J. Am. Chem. Soc. 82 (13), 3454 (1960).
L. J. Smith and F. L. Taylor, J. Am. Chem. Soc. 57 (8), 2460 (1935).
J. C. Stowell, Org. Chem. 36, 3055 (1971).
W. D. Emmons, J. Am. Chem. Soc. 79 (21), 5739 (1957).
J. Hamer and A. Macaluso, Chem. Rev. 64 (4), 473 (1964).
U. Paulsen and B. Lindeke, Acta. Pharm. Suec. 15 (4), 264 (1978).
I. A. Kirilyuk, I. A. Grigor’ev, and L. B. Volodarskii, Izv. Akad. AN SSSR, Ser. Khim., No. 9, 2113 (1991).
R. Neumann, F. de La Vega, and A. Bar-On, Org. Chem. 60 (5), 1315 (1995).
Nitrile Oxides, Nitrones, and Nitronates in Organic Synthesis: Novel Strategies in Synthesis, Ed. by H. Feuer, 2nd ed. (Wiley, USA, 2008).
C. Reichardt, Solvents and Solvent Effects in Organic Chemistry (Wiley-VCH: Weinheim, 2003).
Handbook of Solvents, Ed. by G. Wypych (William Andrew Publ., ChemTec Publ., Toronto, New York, 2001).
R. Schmid, V. N. Sapunov, Non-formal Kinetics. In Search for Chemical Reaction Pathways (Verlag Chemie, Weinheim, 1982).
S. Youn, D. Y. Kim, W. J. Cho, J. M. L. Madridejos, H. M. Lee, M. Kolaski, J. Lee, C. Baig, S. K. Shin, M. Filatov, and K. S. Kim, J. Phys. Chem. A 120 (46), 9305 (2016).
C. Sambiagio, S. P. Marsden, A. J. Blacker, and P. C. McGowan, Chem. Soc. Rev. 43 (10), 3525 (2014).
V. E. Zubarev, The Method of Spin Traps: Application in Chemistry, Biology, and Medicine (Mosk. Gos. Univ., Moscow, 1984) [in Russian].
ACKNOWLEDGMENTS
Authors are grateful to I.A. Kirilyuk and I.F. Zhurko (Vorozhtsov Novosibirsk Institute of Organic Chemistry, Siberian Branch, RAS) for the provided nitrones (BMPO, DIO, and DIOO).
Funding
This study was financially supported by the Russian Foundation for Basic Research (project code 20-03-00150).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by E. Karpushkin
Rights and permissions
About this article
Cite this article
Kolyakina, E.V., Shoipova, F.K. & Grishin, D.F. Nitrones and Nitroso Compounds in the Coupling Reactions for the Synthesis of Macromolecular Structures Based on Polystyrene. Polym. Sci. Ser. B 64, 359–372 (2022). https://doi.org/10.1134/S1560090422700257
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090422700257