Abstract
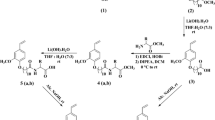
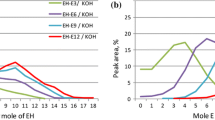
In this work, new series of modified nonionic polymerizable nonyl phenol ethoxylates (NPEs) were synthesized as organic solvent-soluble surfactants. The nonyl, phenyl and propenyl groups were acted as solvophilic segment. The modified polyoxyethylene, maleate chains were behaved as solvophobic segment. The new bifunctional surfmers were prepared by reacting polyoxyethylene 4-nonyl-2-propylenephenol nonionic reactive surfactant with maleic anhydride followed by esterification with poly(ethylene glycol). The chemical structure of the prepared surfactants was determined by 13C and 1H NMR analyses. The surface activities of these surfactants were determined in organic solvents including formamide, toluene and aqueous water solvent from surface tension measurements. The relationship between the surface activity and molecular structure of the organic solvent-soluble polymerizable NPE surfactants as well as the type and structure of organic solvents were discussed. The results showed that the prepared polymerizable NPE surfactants have been reduced the surface tension of both polar and non-polar organic solvents. Surface parameters such as surface excess concentration (Γmax), the area per molecule at interface (A min) and the effectiveness of surface tension reduction were determined from the adsorption isotherms of the prepared surfactants. The prepared polymerizable surfactant used to prepare crosslinked N-isopropylacrylamide (NIPAAm) and 2-acrylamido-2-methylpropane sulfonic acid (AMPS) copolymer microgel based on nonaqueous emulsion copolymerization.
Similar content being viewed by others
References
A. M. Warhurst, An Environmental Assessment of Alkylphenol Ethoxylates and Alkylphenols (Friends of the Earth Scotland, Edinburgh, 1995), pp. 75–105.
S. S. Talmage, Environmental and Human Safety of Major Surfactants. Alcohol Ethoxylates and Alkylphenol Ethoxylates (Lewis Publishers, Boca Raton, Florida, 1994), pp. 115–125.
C. G. Naylor, J. B. Williams, P. T. Varineau, and D. A. Webb, “Nonylphenol ethoxylates in an industrial river” in Proceedings of 4th World Surfactants Congress, Barcelona, Spain, 1996, pp. 378–391.
A. Soares, B. Guieysse, B. Jefferson, E. Cartmell, and J. N. Lester, Environ. Int. 34, 1033 (2008).
B. W. Green and D. P. J. Sheetz, Colloid Interface Sci. 32, 96 (1970).
B. W. Green and F. L. Saunders, J. Colloid Interface Sci. 33, 393 (1970).
S. L. Tsaur and R. B. J. Fitch, Colloid Interface Sci. 115, 450 (1987).
S. A. Chen and H. S. Chang, J. Polym. Sci., Part A: Polym. Chem. 23, 2615 (1985).
Y. Morozumi and Y. Saito, Fuels 24, 1854 (2010).
A. M. Fernandez, U. Held, A. Willing, and W. H. Breuer, Prog. Org. Coat. 53, 246 (2005).
L. N. Butler, C. M. Fellows, and R. G. Gilbert, Prog. Org. Coat. 53, 112 (2005).
H. Gutmann and A. S. Kertes, J. Colloid Interface Sci. 51, 406 (1975).
G.-G. Ying, Environ. Int. 32, 417 (2006).
E. Calvoa, R. Bravoa, A. Amigoa, and J. Gracia-Fadrique, Fluid Phase Equilib. 282, 14 (2009).
M. Li and K. Matyjaszewski, Macromolecules 36, 6028 (2003).
M. Dreja, W. Pyckhout-Hintzen, and B. Tieke, Macromolecules 31, 272 (1998).
W. R. Palani-Raj, M. Sasthav, and H. M. Cheung, Langmuir 8, 1931 (1992).
T. D. Li, C. H. Chew, S. C. Ng, L. M. Gan, W. K. Teo, J. Y. Gu, and G. Y. Zhang, J. Macromol. Sci. Pure Appl. Chem. A32, 969 (1995).
L. M. Gan, T. D. Li. C. H. Chew, W. K. Teo, and L. H. Gan, Langmuir 11, 3316 (1995).
A. Hammouda and M.-P. Pileni, Langmuir 11, 3656 (1995).
M. Dreja and B. Tieke, Macromol. Rapid Commun. 17, 825 (1996).
K. Landfester, N. Bechthold, F. Tiarks, and M. Antonietti, Macromolecules 32, 5222 (1999).
X. J. Xu, H. L. Goh, K. S. Siow, and L. M. Gan, Langmuir 17, 6077 (2001).
A. Guyot and K. Tauer, Adv. Polym. Sci. 111, 43 (1994).
F. Candau and R. Zana, in Polymeric Materials Encyclopedia, Ed. by J. C. Salamone (CRC Press, Boca Ration, 1996), p. 4287.
K. M. Lusvardi, K. V. Schubert, and E. W. Kaler, Ber Bunsen-Ges. Phys. Chem. 100, 373 (1996).
K. Landfester, M. Willert, and M. Antonietti, Macromolecules 33, 2370 (2000).
L.-I. Atanase and G. Riess, Polym. Int. 60, 1563 (2011).
T. Tadros, Adv. Colloid Interface Sci. 147–148, 281 (2009).
M. R. Thompson, C. Tzoganakis, and G. L. Rempel, J. Polym. Sci., Part A: Polym. Chem. 36, 2371 (1998).
A. M. Al-Sabagh and A. M. Atta, J. Chem. Technol. Biotechnol. 74, 1075 (1999).
V. Annadanam, S. Sainath, M. Isokawa, M. Suzuki, S. Ishii, S. Matsuura, N. Nagai, and T. Fujita, Macromolecules 42, 4356 (2009).
A. Tomlinson, T. N. Danks, and D. M. Heyes, Langmuir 13, 5881 (1997).
T. E. Sandovala, L. J. Espinozab, I. A. Guerrac, A. F. Olead, and M. P. Garate, Colloids Surf. A 398, 17 (2012).
M. K. Bernett and W. A. Zisman, J. Phys. Chem. 66, 1207 (1962).
A. M. Atta, Polym. Int. 56, 984 (2007).
A. M. Atta, M. E. Abdel-Rauf, N. E. Maysour, A. M. Abdul-Rahiem, and A. A. Abdel-Azim, J. Polym. Res. 13, 39 (2006).
A.M. Atta, H. I. Al-Shafy, and Z. M. Mohamed, Polym. Adv. Technol. 22, 1879 (2011).
M. J. Rosen, Surfactants and Interfacial Phenomena (John Wiley, New York, 1985), pp. 15–55.
J. A. Garate, M. A. Valenzuela, M. P. Garate, and A. F. Olea, J. Surf. Deterg. 12, 231 (2009).
X. J. Xu, H. L. Goh, K. S. Siow, and L. M. Gan, Langmuir 17, 6077 (2001).
B. P. Binks, P. D. I. Fletcher, and S. N. Kotsev, Langmuir 13, 6669 (1997).
B. P. Binks, P. D. I. Fletcher, and W. F. C. Sager, J. Mol. Liq. 72, 177 (1997).
P. K. Sansanwal, J. Sci. Ind. Res. 65, 57 (2006).
C. Batig and H. Akbas, J. Chem. Thermodyn. 43, 1349 (2011).
S.-C. Chen, S.-W. Kuo, C.-S. Liao, and F.-C. Chang, Macromolecules 41, 8865 (2008).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Atta, A.M., Dyab, A.K.F., Al-Lohedan, H.A. et al. Novel reactive polymerizable nonyl phenol ethoxylate surfactants as emulsifier in non-aqueous emulsion polymerization. Polym. Sci. Ser. B 56, 770–787 (2014). https://doi.org/10.1134/S1560090414060025
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090414060025