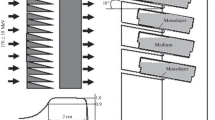
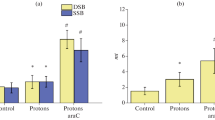
Abstract
The modifying effect of 1-β-D-arabinofuranosylcytosine (AraC) and hydroxyurea (HU) on the formation frequency of DNA double-strand breaks (DSB) in human U87 glioblastoma and murine B16 melanoma cells after accelerated proton and nitrogen ion (15N) exposure has been studied. The DNA comet assay and immunocytochemistry methods were used to determine the dependences of the frequency of DNA DSB formation and kinetics of their repair in tumor cells upon irradiation with particles of a wide linear energy transfer (LET) range under normal conditions and in the presence of modifiers. The combination of AraC and HU has the strongest modifying effect on the DSB yield. Under the influence of radiomodifiers, the dose change factor is ~2. Under normal conditions, DSB repair proceeds with exponential kinetics. In the presence of modifiers, a certain increase in the DNA DSB yield is observed after 6 h of post-irradiation incubation, followed by a decrease after 24 h of observation. The strongest modifying effect of AraC on the formation of radiation-induced foci (RIF) in glioblastoma cells is observed for proton exposure compared with nitrogen ions. In the presence of AraC, 24 hours after proton irradiation the RIF yield was 2.3 times higher than after exposure without it. For nitrogen ions with LET = 81 keV/μm and 180 keV/μm, the RIF yield in glioblastoma cells in the presence of AraC increases by 1.7 and 1.5 times 24 hours after irradiation, respectively, compared with the control samples. Possible mechanisms of tumor cell response to exposure to radiations with different LET in the presence of radiomodifiers are discussed.




Similar content being viewed by others
REFERENCES
A. V. Boreiko, V. N. Chausov, E. A. Krasavin, I. Ravnachka, and S. I. Stukova, “ The effect of DNA synthesis inhibitors on the induction and repair of DNA double-strand breaks in human lymphocytes under the action of radiation with different LET,” Phys. Part. Nucl. Lett. 8, 399—404 (2011).
V. N. Chausov, A. V. Boreyko, T. S. Bulanova, M. G. Zadneprianetc, E. V. Il’ina, L. Jezkova, E. A. Krasavin, R. A. Kozhina, E. A. Kuzmina, E. A. Kulikova, E. V. Smirnova, and S. I. Tiounchik, “Formation of direct and enzymatic DNA double-strand breaks under the influence of repair inhibitors under the action of radiation of different quality,” Pis’ma EChAYa 15, 573—588 (2018).
M. R. Miller and D. N. Chinault, “Evidence that DNA polymerases alpha and beta participate differentially in DNA repair synthesis induced by different agents,” J. Biol. Chem. 257, 46–49 (1982).
A. S. Prakasha Gowda, J. M. Polizzi, K. A. Eckert, and T. E. Spratt, “Incorporation of gemcitabine and cytarabine into DNA by DNA Polymerase β and Ligase III/XRCC1,” Biochemistry 49, 4833–4840 (2010).
M. Weinfeld, A. Rasouli-Nia, M. A. Chaudhry, and R. A. Britten, “Response of base excision repair enzymes to complex DNA lesions,” Radiat. Res. 156, 584–589 (2001).
L. Harrison and S. Malyarchuk, “Can DNA repair cause enhanced cell killing following treatment with ionizing radiation?,” Pathophysiol. Off. J. Int. Soc. Pathophysiol 8, 149–159 (2002).
B. Rydberg, “Radiation-induced heat-labile sites that convert into DNA double-strand breaks,” Radiat. Res. 153, 805–812 (2009).
C. E. Rube, A. Fricke, J. Wendorf, A. Stützel, M. Kühne, M. F. Ong, P. Lipp, and C. Rübe, “Accumulation of DNA double-strand breaks in normal tissues after fractionated irradiation,” Int. J. Radiat. Oncol. Biol. Phys. 76, 1206–1213 (2010).
C. E. Rübe, S. Grudzenski, M. Kühne, X. Dong, N. Rief, M. Löbrich, and C. Rübe, “DNA double-strand break repair of blood lymphocytes and normal tissues analysed in a preclinical mouse model: implications for radiosensitivity testing,” Clin. Cancer Res. 14, 6546–6555 (2008).
L. Jakl, E. Marková, L. Koláriková, and I. Belyaev, “Biodosimetry of low dose ionizing radiation using DNA repair foci in human lymphocytes,” Genes 11, 58 (2020).
S. Nair, M. Engelbrecht, X. Miles, R. Ndimba, R. Fisher, P. Plessis, J. Bolcaen, J. Nieto-Camero, E. de Kock, and C. Vandevoorde, “The impact of dose rate on DNA double-strand break formation and repair in human lymphocytes exposed to fast neutron irradiation,” Int. J. Mol. Sci. 20, 5350 (2019).
F. Hanton, P. Chaudhary, D. Doria, D. Gwynne, C. Maiorino, C. Scullion, H. Ahmed, T. Marshall, K. Naughton, L. Romagnani, S. Kar, G. Schettino, P. McKenna, S. Botchway, D. R. Symes, P. P. Rajeev, K. M. Prise, and M. Borghesi, “DNA DSB repair dynamics following irradiation with laser-driven protons at ultra-high dose rates,” Sci. Rep. 9, 1–10 (2019).
E. A. Krasavin, A. V. Boreyko, M. G. Zadneprianetc, E. V. Il’ina, R. A. Kozhina, E. A. Kuzmina, E. A. Kulikova, E. V. Smirnova, G. N. Timoshenko, S. I. Tiunchik, and V. N. Chausov, “Effect of DNA synthesis inhibitors on the biological efficiency of the proton beam in the modified Bragg peak,” Pis’ma EChAYa 2, 181–190 (2019).
K. Końca, A. Lankoff, A. Banasik, H. Lisowska, T. Kuszewski, S. Góźdź, Z. Koza, and A. Wojcik, “A cross-platform public domain PC image-analysis program for the comet assay,” Mutat. Res. 534, 15–20 (2003).
M. G. Zadneprianetc, A. V. Boreyko, T. S. Bulanova, L. Jezkova, E. A. Krasavin, E. A. Kulikova, E. V. Smirnova, M. Falk, and I. Falkova, “Patterns of formation and elimination of γH2AX/53VP1 foci under the action of γ-rays and accelerated heavy ions,” Radiats. Biol. Radioekol. 58, 146—156 (2018).
P. Matula, M. Maška, O. Danĕk, P. Matula, M. Kozubek, “Acquiarium: Free software for the acquisition and analysis of 3d images of cells in fluorescence microscopy,” in Proceedings of 2009 IEEE International Symposium on Biomedical Imaging from Nano to Macro, ISBI 2009 (2009), pp. 1138–1141.
E. Bobkova, D. Depes, J. H. Lee, L. Jezkova, I. Falkova, E. Pagacova, O. Kopecna, M. Zadneprianetc, A. Bacikova, E. Kulikova, E. Smirnova, T. Bulanova, A. Boreyko, E. Krasavin, F. Wenz, F. Bestvater, G. Hildenbrand, M. Hausmann, and M. Falk, “Recruitment of 53BP1 proteins for DNA repair and persistence of repair clusters differ for cell types as detected by single molecule localization microscopy,” Int. J. Mol. Sci. 19, 3713 (2018).
S. C. Short, C. Martindale, S. Bourne, G. Brand, M. Woodcock, and P. Johnston,” DNA repair after irradiation in glioma cells and normal human astrocytes,” Neur. Oncol. 9, 404–411 (2007).
V. Michalik, “Model of DNA damage induced by radiations of various qualities,” Int. J. Radiat. Biol. 62, 9–20 (1992).
H. Nikjoo, P. O’Neill, W. E. Wilson, and D. T. Goodhead, “Computational approach for determining the spectrum of DNA damage induced by ionizing radiation,” Radiat. Res. 156, 577–583 (2001).
A. Urushibara, N. Shikazono, P. O’Neill, K. Fujii, S. Wada, and A. Yokoya, “LET dependence of the yield of single-, double-strand breaks and base lesions in fully hydrated plasmid DNA films by 4 He 2+ ion irradiation,” Int. J. Radiat. Biol. 84, 23–33 (2008).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by E. Glushachenkova
Rights and permissions
About this article
Cite this article
Boreyko, A.V., Zadneprianetc, M.G., Chausov, V.N. et al. Combined Action of DNA Synthesis Inhibitors and Accelerated Protons on Malignant Tumor Cells. Phys. Part. Nuclei Lett. 20, 683–689 (2023). https://doi.org/10.1134/S1547477123040131
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1547477123040131