Abstract
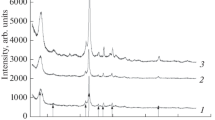
The stability of synthetic and natural zeolites in model biological media simulating the environment of the stomach (pH 1.8), blood plasma (pH 6.9), and intestines (pH 8) is studied. The effect of long-term exposure (up to 7 days) to biological media on the crystal structure of Beta, Rho, Y, and clinoptilolite zeolites is studied. The degree of degradation of the crystal structure of zeolites is controlled by X-ray phase analysis. Based on the results obtained, conclusions are drawn on the prospects for the use of synthetic and natural zeolites as drug carriers.

Similar content being viewed by others
REFERENCES
Bacakova, L., Vandrovcova, M., Kopova, I., and Jirka, I., Applications of zeolites in biotechnology and medicine—a review, Biomater. Sci., 2018, vol. 6, no. 5, pp. 974–989.
Vallet-Regí, M., Colilla, M., Izquierdo-Barba, I., and Manzano, M., Mesoporous silica nanoparticles for drug delivery: Current insights, Molecules, 2018, vol. 23, no. 1, p. 47.
Trewyn, B.G., Giri, S., Slowing, I.I., and Lin, V.S.Y., Mesoporous silica nanoparticle based controlled release, drug delivery, and biosensor systems, Chem. Commun., 2000, vol. 31, pp. 3236–3245.
Rimoli, M.G., Rabaioli, M.R., Melisi, D., Curcio, A., Mondello, S., and Mirabelli, R., Synthetic zeolites as a new tool for drug delivery, J. Biomed. Mater. Res. A, 2008, vol. 87, pp. 156–164.
Kontogiannidou, E., In vitro and ex vivo assessment of microporous faujasite zeolite (NaX-FAU) as a carrier for the oral delivery of danazol, J. Drug Deliv. Sci. Technol., 2019, vol. 51, pp. 177–184.
Golubeva, O.Yu., Brazovskaya, E.Y., Alikina, Y.A., D’yachenko, S., and Zhernovoi, A., Synthesis and study of nanocomposites based on beta zeolite and magnetite for targeted drug delivery, Glass Phys. Chem., 2019, vol. 45, no. 1, pp. 66–73.
Koubaissy, B., Toufaily, J., Yaseen, Z., Daou, T.J., Jradi, S., and Hamieh, T., Adsorption of uremic toxins over dealuminated zeolites, Adsorpt. Sci. Technol., 2017, vol. 35, no. 1, pp. 3–19.
Cheah, W.K., Ishikawa, K., Othman, R., and Yeoh, F.Y., Nanoporous biomaterials for uremic toxin adsorption in artificial kidney systems: A review, J. Biomed. Mater. Res., Part B, 2017, vol. 105, no. 5, p. 1232.
Ulyanova, N.Yu., Kurilenko, L.N., Shamova, O.V., Orlov, D.S., and Golubeva, O.Yu., Hemolitic activity and sorption ability of beta zeolite nanoparticles, Glass Phys. Chem., 2020, vol. 46, no. 2, pp. 155–161.
Souza, I.M.S., Garcia-Villén, F., Viseras, C., and Perger, S.B.C., Zeolites as ingredients of medicinal products, Pharmaceutics, 2023, vol. 15, no. 5, p. 1352.
Widder, K.J., Senyei, A.E., and Scarpelli, D.G., Magnetic microspheres: A model system for site specific drug delivery in vivo, Exp. Biol. Med., 1978, vol. 158, no. 2, pp. 141–146.
Shreya, A.B., Raut, S.Y., Managuli, R.S., Udupa, N., and Mutalik, S., Active targeting of drugs and bioactive molecules via oral administration by ligand-conjugated lipidic nanocarriers: Recent advances, AAPS PharmSciTech, 2018, vol. 20, no. 1, p. 15.
Homayun, B., Lin, X., and Choi, H.J., Challenges and recent progress in oral drug delivery systems for biopharmaceuticals, Pharmaceutics, 2019, vol. 11, no. 3, p. 129.
Rouge, N., Buri, P., and Doelker, E., Drug absorption sites in the gastrointestinal tract and dosage forms for site-specific delivery, Int. J. Pharmaceut., 1996, vol. 136, pp. 117–139.
Thakral, S., Thakral, N.K., and Majumdar, D.K., Eudragit: A technology evaluation, Expert Opin. Drug Deliv., 2013, vol. 10, no. 1, pp. 131–149.
Liu, L., Yao, W., Rao, Y., Lu, X., and Gao, J., PH-responsive carriers for oral drug delivery: Challenges and opportunities of current platforms, Drug Deliv., 2017, vol. 24, no. 1, pp. 569–581.
Tas, A.C., Synthesis of biomimetic Ca-hydroxyapatite powders at 37°C in synthetic body fluids, Biomaterials, 2000, vol. 21, no. 14, pp. 1429–1438.
Tang, T., Zhang, L., Dong, H., Fang, Z., Yu, Q., and Tang, T., RSC Adv., 2017, vol. 7, pp. 7711–7717.
Robson, H., Verified Synthesis of Zeolitic Materials, Amsterdam: Elsevier, 2016.
Golubeva, O.Yu. and Ul’yanova, N.Yu., Stabilization of silver nanoparticles and clusters in porous zeolite matrices with Rho, beta, and Paulingite structures, Glass Phys. Chem., 2015, vol. 41, no. 5, pp. 537–544.
Funding
This study was carried out as part of a state task of the Institute of Silicate Chemistry, Russian Academy of Sciences (subject no. 0081-2022-0001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interest.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ul’yanova, N.Y., Brazovskaya, E.Y. & Golubeva, O.Y. Study of the Stability of Zeolites in Model Biological Environments. Glass Phys Chem 49, 706–708 (2023). https://doi.org/10.1134/S1087659623600734
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659623600734