Abstract
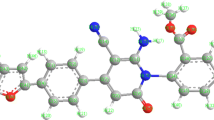
A series of new benzamidine derivatives have been synthesized in good yields by involving 1-(1H-benzimidazol-2-yl)-2-bromoethanone, 4-(1H-benzimidazol-2-yl)-1,3-thiazol-2-amine, and N-benzylidene-4-(1H-benzimidazol-2-yl)-1,3-thiazol-2-amine as reactive intermediates. The synthesized compounds were characterized by spectral data. Molecular docking study of N-(4-(1H-benzimidazol-2-yl)-1,3-thiazol-2-yl]-1-[(E)-phenyldiazenyl]-1-phenylmethanimine has been carried out as a representative case.




Similar content being viewed by others
REFERENCES
Pardeshi, V.A.S., Chundawat, N.S., Pathan, S.I., Sukhwal, P., Chundawat, T.P.S., and Singh, G.P., Synth Commun., 2021, vol. 51, no. 4, p. 485. https://doi.org/10.1080/00397911.2020.1841239
Sharma, S. and Rauf, A., Chin. Chem. Lett., 2009, vol. 20, no. 10, p. 1145. https://doi.org/10.1016/j.cclet.2009.04.032
Beltran-Hortelano, I., Alcolea, V., Font, M., and Perez-Silanes, S., Eur. J. Med. Chem., 2020, vol. 206, article ID 112692. https://doi.org/10.1016/j.ejmech.2020.112692
Joshi, B., Panda, S.K., Jouneghani, R.S., Liu, M., Parajuli, N., Leyssen, P., Neyts, J., and Luyten, W., Evidence-Based Complementary Altern. Med., 2020, article ID 1043471. https://doi.org/10.1155/2020/1043471
Smaili, A., Jebbari, S., Rifai, L.A., Faize, L., Koussa, T., Sir, H.A., Makroum, K., Belfaiza, M., El Kihel, A., Ahbala, M., Venisse, J.S., and Faize, M., Phytoparasitica, 2019, vol. 47, no. 5, p. 733. https://doi.org/10.1007/s12600-019-00764-9
Patel, V.M., Patel, N.B., Chan-Bacab, M.J., and Rivera, G., Synth. Commun., 2020, vol. 50, no. 6, p. 858. https://doi.org/10.1080/00397911.2020.1725057
Gut, J., Ang, K.K., Legac, J., Arkin, M.R., Rosenthal, P.J., and McKerrow, J.H., J. Microbiol. Methods, 2011, vol. 84, no. 3, p. 398. https://doi.org/10.1016/j.mimet.2010.12.026
North, T.W., Sequar, G., Townsend, L.B., Drach, J.C., and Barry, P.A., Antimicrob. Agents Chemother., 2004, vol. 48, no. 7, p. 290. https://doi.org/10.1128/AAC.48.7.2760-2765.2004
Desai, N.C., Shihory, N.R., Kotadiya, G.M., and Desai, P., Eur. J. Med. Chem., 2014, vol. 82, p. 480. https://doi.org/10.1016/j.ejmech.2014.06.004
Cheong, J.E., Zaffagni, M., Chung, I., Xu, Y., Wang, Y., Jernigan, F.E., Zetter, B.R., and Sun, L., Eur. J. Med. Chem., 2018, vol. 144, p. 372. https://doi.org/10.1016/j.ejmech.2017.11.037
Narasimhan, B., Sharma, D., and Kumar, P., Med. Chem. Res., 2012, vol. 21, no. 3, p. 269. https://doi.org/10.1007/s00044-010-9533-9
Nile, S.H., Kumar, B., and Park, S.W., Chem. Biol. Drug Des., 2013, vol. 82, no. 3, p. 290. https://doi.org/10.1111/cbdd.12141
Wang, X.J., Xi, M.Y., Fu, J.H., Zhang, F.R., Cheng, G.F., Yin, D.L., and You, Q.D., Chin. Chem. Lett., 2012, vol. 23, no. 6, p. 707. https://doi.org/10.1016/j.cclet.2012.04.020
Tahlan, S., Kumar, S., Kakkar, S., and Narasimhan, B., BMC Chem., 2019, vol. 13, article no. 66. https://doi.org/10.1186/s13065-019-0579-6
Rouf, A. and Tanyeli, C., Eur. J. Med. Chem., 2015, vol. 97, p. 911. https://doi.org/10.1016/j.ejmech.2014.10.058
El-Enany, W.A.M.A., Gomha, S.M., El-Ziaty, A.K., Hussein, W., Abdulla, M.M., Hassan, S.A., Sallam, H.A., and Ali, R.S., Synth. Commun., 2020, vol. 50, no. 1, p. 85. https://doi.org/10.1080/00397911.2019.1683207
Maghraby, M.T., Abou-Ghadir, O.M.F., AbdelMoty, S.G., Ali, A.Y., and Salem, O.I.A., Bioorg. Med. Chem., 2020, vol. 28, no. 7, article ID 115403. https://doi.org/10.1016/j.bmc.2020.115403
Mishra, C.B., Kumari, S., and Tiwari, M., Eur. J. Med. Chem., 2015, vol. 92, p. 1. https://doi.org/10.1016/j.ejmech.2014.12.031
Qin, Y.J., Wang, P.F., Makawana, J.A., Wang, Z.C., Wang, Z.N., Yan, G., Jiang, A.Q., and Zhu, H.L., Bioorg. Med. Chem. Lett., 2014, vol. 22, p. 5279. https://doi.org/10.1016/j.bmcl.2014.09.054
Madni, M., Hameed, S., Ahmed, M.N., Tahir, M.N., Al-Masoudi, N.A., and Pannecouque, C., Med. Chem. Res., 2017, vol. 26, no. 10, p. 2653. https://doi.org/10.1007/s00044-017-1963-1
Gupta, V. and Kant, V., Science International, 2013, vol. 1, no. 7, p. 253. https://doi.org/10.17311/sciintl.2013.253.260
Hosny, M.A., Zaki, Y.H., Mokbel, W.A., and Abdelhamid, A.O., BMC Chem., 2019, vol. 13, article no. 37. https://doi.org/10.1186/s13065-019-0559-x
Bondock, S., Khalifa, W., and Fadda, A.A., Eur. J. Med. Chem., 2007, vol. 42, no. 7, p. 948. https://doi.org/10.1016/j.ejmech.2006.12.025
Gjorgjieva, M., Tomasic, T., Barancokova, M., Katsamakas, S., Ilas, J., Tammela, P., Peterlin Masic, L., and Kikelj, D., J. Med. Chem., 2016, vol. 59, no. 19, p. 8941. https://doi.org/10.1021/acs.jmedchem.6b00864
ACKNOWLEDGMENTS
The authors thank Jawaharlal Nehru Technological University, Hyderabad, India, for providing necessary facilities to carry out this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interest.
Rights and permissions
About this article
Cite this article
Reddy, S.R., Reddy, G.G., Reddy, C.V.R. et al. Synthesis, Characterization, and Molecular Docking Study of N-[4-(1H-Benzimidazol-2-yl)-1,3-thiazol-2-yl]-1-[(E)-phenyldiazenyl)-1-phenylmethanimines. Russ J Org Chem 58, 858–864 (2022). https://doi.org/10.1134/S107042802206015X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042802206015X