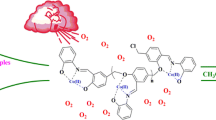
Abstract
In the present study, a dioxomolybdenum(VI) complex chelated with the ONO tridentate Schiff base ligand supported on chloromethylated polystyrene [MoO2L@CMP] was synthesized and characterized by various spectroscopic techniques like FT-IR, UV-Vis, DRS, SEM and EDAX. Then, its application was investigated in the oxidation of various alkyl and aryl sulfides using hydrogen peroxide in ethanol under reflux conditions. The supported catalyst could readily be recovered from the reaction mixture and reused several times with no considerable increase in its catalytic activity.








Similar content being viewed by others
REFERENCES
Maddah, B. and Tabrizi, A.S., Iran. J. Catal., 2016, vol. 6, p. 461.
Boruah, J.J., Das, S.P., Ankireddy, S.R., Gogoi, S.R., and Islam, N.S., Green Chem., 2013, vol. 15, p. 2944. https://doi.org/10.1039/C3GC40304A
Bezaatpour, A., Askarizadeh, E., Akbarpour, S., Amiri, M., and Babaei, B., Mol. Catal., 2017, vol. 436, p. 199. https://doi.org/10.1016/j.mcat.2017.04.021
Han, J.V.A., Klika, K.D., Drabowicz, J., and Wzorek, A., Chem. Soc. Rev., 2018, vol. 47, p. 1307. https://doi.org/10.1039/C6CS00703A
Salom-Roig, X. and Bauder, C., Synthesis, 2020, vol. 52, p. 964. https://doi.org/10.1055/s-0039-1690803
Gulbe, K. and Turks, M., J. Org. Chem., 2020, vol. 85, p. 5660. https://doi.org/10.1021/acs.joc.9b03403
Nfor, E.N., Husian, A., Majoumo-Mbe, F., Njah, I.N., Offiong, O.E., and Bourne, S.A., Polyhedron, 2013, vol. 63, p. 207. https://doi.org/10.1016/j.poly.2013.07.028
Pal, R., Kumar, V., Gupta, A.K., and Beniwal, V., Med. Chem. Res., 2014, vol. 23, p. 3327. https://doi.org/10.1007/s00044-014-0911-6
Ebrahimipour, S.Y., Khabazadeh, H., Castro, J., Sheikhshoaie, I., Crochet, A., and Fromm, K.M., Inorg. Chim. Acta, 2015, vol. 427, p. 52. https://doi.org/10.1016/j.ica.2014.11.023
Wu, M., Yang, D.D., Zheng, H.W., Liang, Q.F., Li, J.B., Kang, Y., Li, S., Jiao, C., Zheng, X.J., and Jin, L.P., Dalton Trans., 2021, vol. 50, p. 1507. https://doi.org/10.1039/D0DT04062B
Bakale, R.P., Naik, G.N., Mangannavar, C.V., Muchchandi, I.S., Shcherbakov, I.N., Frampton, C., and Gudasi, K.B., Eur. J. Med. Chem., 2014, vol. 73, p. 38. https://doi.org/10.1016/j.ejmech.2013.11.037
Dalia, S.A., Afsan, F., Hossain, M.S., Khan, M.N., Zakaria, C.M., Kudrat-E-Zahan, M., and Ali, M.M., Int. J. Chem. Stud., 2018, vol. 6, p. 2859.
Zhang, B., Luo, H., Xu, Q., Lin, L., and Zhang, B., Oncotarget, 2017, vol. 8, p. 13620. https://doi.org/10.18632/oncotarget.14620
Sahraei, A., Kargar, H., Hakimi, M., and Tahir, M.N., J. Mol. Struct., 2017, vol. 1149, p. 576. https://doi.org/10.1016/j.molstruc.2017.08.022
Kargar, H., Adabi Ardakani, A., Tahir, M.N., Ashfaq, M., and Munawar, K.S., J. Mol. Struct., 2021, vol. 1229, article ID 129842. https://doi.org/10.1016/j.molstruc.2020.129842
Matela, G., Anticancer Agents Med. Chem., 2020, vol. 20, p. 1908. https://doi.org/10.2174/1871520620666200507091207
Kaya, Y., Ercag, A., and Kaya, K., J. Coord. Chem., 2018, vol. 71, p. 3364. https://doi.org/10.1080/00958972.2018.1516872
Guo, Y., Xiao, L., Li, P., Zou, W., Zhang, W., and Hou, L., Mol. Catal., 2019, vol. 475, article ID 110498. https://doi.org/10.1016/j.mcat.2019.110498
Kargar, H., Fallah-Mehrjardi, M., Behjatmanesh-Ardakani, R., Munawar, K.S., Ashfaq, M., and Tahir, M.N., Transition Met. Chem., 2021, vol. 46, p. 437. https://doi.org/10.1007/s11243-021-00460-w
Tang, J., Yao, P., Huang, F., Luo, M., Wei, Y., and Bian, H., Tetrahedron: Asymmetry, 2017, vol. 28, p. 1700. https://doi.org/10.1016/j.tetasy.2017.10.021
Ardakani, M.H., Saeednia, S., and Sabet, M., Silicon, 2019, vol. 11, p. 1775. https://doi.org/10.1007/s12633-018-9992-z
Kargar, H., Moghimi, A., Fallah-Mehrjardi, M., Behjatmanesh-Ardakani, R., Amiri Rudbari, H., and Munawar, K.S., J. Sulfur Chem., 2022, vol. 43, p. 22. https://doi.org/10.1080/17415993.2021.1941020
Kargar, H., Kargar, K., Fallah-Mehrjardi, M., and Munawar, K.S., Monatsh. Chem., 2021, vol. 152, p. 593. https://doi.org/10.1007/s00706-021-02780-0
Mendel, R.F., Dalton Trans., 2005, p. 3404. https://doi.org/10.1039/B505527J
Quintal, S., da Silva, M.J.P., Martins, S.R.M., Sales, R., Felix, V., Drew, M.G.B., Meireles, M., Mourato, A.C., Nunes, C.D., Saraiva, M.S., Machuqueiro, M., and Calhorda, M.J., Dalton Trans., 2019, vol. 48, p. 8449. https://doi.org/10.1039/C9DT00469F
Sugimoto, H., Sato, M., Giles, L.J., Asano, K., Suzuki, T., Kirk, M.L., and Itoh, S., Dalton Trans., 2013, vol. 42, p. 15927. https://doi.org/10.1039/C3DT51485D
Ghosh, A.C., Samuel, P.P., and Schulzke, C., Dalton Trans., 2017, vol. 46, p. 7523. https://doi.org/10.1039/C7DT01470H
Kargar, H., Forootan, P., Fallah-Mehrjardi, M., Behjatmanesh-Ardakani, R., Amiri Rudbari, H., Munawar, K.S., Ashfaq, M., and Tahir, M.N., Inorg. Chim. Acta, 2021, vol. 523, article ID 120414. https://doi.org/10.1016/j.ica.2021.120414
Kargar, H. and Fallah-Mehrjardi, M., J. Iran. Chem. Soc., 2021, vol. 18, p. 3443. https://doi.org/10.1007/s13738-021-02282-0
Zaytseva, M.P., Muradova, A.G., Sharapaev, A.I., Yurtov, E.V., Grebennikov, I.S., and Savchenko, A.G., Russ. J. Inorg. Chem., 2018, vol. 63, p. 1684. https://doi.org/10.1134/S0036023618120239
Hassani, H., Ebrahim, S., and Feizi, N., Russ. J. Inorg. Chem., 2020, vol. 65, p. 940. https://doi.org/10.1134/S0036023620060054
Fakhri, H., Mahjoub, A., Nejat, R., and Maridiroosi, A., Inorg. Chem. Commun., 2021, vol. 123, article ID 108353. https://doi.org/10.1016/j.inoche.2020.108353
Yu, Y.Q. and Xu, D.Z., Tetrahedron, 2015, vol. 71, p. 2853. https://doi.org/10.1016/j.tet.2015.03.070
Mohammadi, E. and Movassagh, B., New. J. Chem., 2018, vol. 42, p. 11471. https://doi.org/10.1039/C8NJ01042K
Arashiba, K., Itabashi, T., Nakajima, K., and Nishibayashi, Y., Chem. Lett., 2019, vol. 48, p. 693. https://doi.org/10.1246/cl.190193
Kargar, H., Moghadam, M., Mirkhani, V., Tangestaninejad, S., Mohammadpoor-Baltork, I., and Rezaei, S., Transition Met. Chem., 2013, vol. 38, p. 1. https://doi.org/10.1007/s11243-012-9651-y
Kargar, H., Transition Met. Chem., 2014, vol. 39, p. 811. https://doi.org/10.1007/s11243-014-9863-4
Javadi, M.M., Moghadam, M., MohammadpoorBaltork, I., Tangestaninejad, S., Mirkhani, V., Kargar, H., and Tahir, M.N., Polyhedron, 2014, vol. 72, p. 19. https://doi.org/10.1016/j.poly.2014.01.004
Kargar, H., Bazrafshan, M., Fallah-Mehrjardi, M., Behjatmanesh-Ardakani, R., Amiri Rudbari, H., Munawar, K.S., Ashfaq, M., and Tahir, M.N., Polyhedron, 2021, vol. 202, article ID 115194. https://doi.org/10.1016/j.poly.2021.115194
Kargar, H., Kaka-Naeini, A., Fallah-Mehrjardi, M., Behjatmanesh-Ardakani, R., Amiri Rudbari, H., and Munawar, K.S., J. Coord. Chem., 2021, vol. 74, p. 1563. https://doi.org/10.1080/00958972.2021.1915488
Fu, Y., Huang, X., Zhong, S., Yi, W.J., and Li, L.J., Chem. Pap., 2019, vol. 73, p. 2183. https://doi.org/10.1007/s11696-019-00761-6
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interest.
Rights and permissions
About this article
Cite this article
Kargar, H., Anaridokht, F. & Fallah-Mehrjardi, M. Biomimetic Oxidation of Sulfides Catalyzed by Polystyrene-Bound Dioxomolybdenum Complex as an Efficient Recoverable Heterogeneous Catalyst. Russ J Org Chem 58, 549–556 (2022). https://doi.org/10.1134/S1070428022040121
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428022040121