Abstract
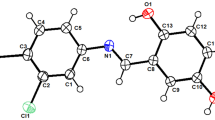
Previously unknown N-substituted 2-(benzylimino)-4-styryl-2,5-dihydrofuran-3-carboxamides were synthesized by reaction of 2-imino-2,5-dihydrofuran-3-carboxamides with benzylamine in glacial acetic acid. It was found that a part of benzylamine reacts as a nucleophile at the imino group and that the other part undergoes oxidation followed by dehydration to generate benzylimine which acts as a synthetic equivalent of benzaldehyde toward the 4-methyl group. The synthesized compounds were characterized by IR and NMR spectra and elemental analyses. 2-(Benzylimino)-N,5,5-trimethyl-4-(2-phenylethenyl)-2,5-dihydrofuran-3-carboxamide was also synthesized independently by reaction of previously reported 2-imino-4-(2-phenylethenyl)-N,5,5-trimethyl-2,5-dihydrofuran-3-carboxamide with benzylamine in glacial acetic acid.




Similar content being viewed by others
REFERENCES
Avetisyan, A.A., Karapetyan, L.V., and Tadevosyan, M.D., Russ. J. Org. Chem., 2009, vol. 45, p. 1031. https://doi.org/10.1134/S1070428009070082
Avetisyan, A. and Karapetyan, L., Heterocycl. Commun., 2012, vol. 18, p. 263. https://doi.org/10.1515/hc-2012-0113
Tokmajyan, G. and Karapetyan, L., J. Braz. Chem. Soc., 2016, vol. 27, p. 967.
Tokmajyan, G. and Karapetyan, L., J. Heterocycl. Chem., 2017, vol. 54, p. 1630. https://doi.org/10.1002/jhet.2713
Karapetyan, L.V. and Tokmajyan, G.G., Russ. J. Org. Chem., 2020, vol. 56, p. 1484. https://doi.org/10.1134/S1070428020080217
Karapetyan, L.V. and Tokmajyan, G.G., Russ. J. Org. Chem., 2020, vol. 56, p. 1844. https://doi.org/10.1134/S1070428020080217
Karapetyan, L.V., Tokmajyan, G.G., and Paronikyan, R.V., Russ. J. Org. Chem., 2021, vol. 57, p. 131. https://doi.org/10.1134/S1070428021010206
Karapetyan, L.V. and Tokmajyan, G.G., Russ. J. Org. Chem., 2021, vol. 57, p. 661. https://doi.org/10.1134/S1070428021040230
Avetsyan, A.A., Akhnazaryan, A.A., and Melikyan, G.S., Arm. Khim. Zh., 1988, vol. 41, p. 756.
Perlessy, A., Avetisian, A.A., Aknazarian, A.A., and Melikian, G.S., Collect. Czech. Chem. Commun., 1989, vol. 54, p. 1666. https://doi.org/10.1135/cccc19891666
Avetsyan, A.A., Kagramanyan, A.A., and Melikyan, G.S., Arm. Khim. Zh., 1989, vol. 42, p. 633.
Avetisyan, A.A., Alvandzhyan, A.G., and Avetisyan, K.S., Russ. J. Org. Chem., 2011, vol. 47, p. 265. https://doi.org/10.1134/S1070428011020175
Avetisyan, A.A., Alvandzhyan, A.G., and Avetisyan, K.S., Russ. J. Org. Chem., 2011, vol. 47, p. 433. https://doi.org/10.1134/S1070428011030183
Belikov, M.Yu., Fedoseev, S.V., Ievlev, M.Yu., and Ershov, O.V., Russ. J. Org. Chem., 2019, vol. 55, p. 1623. https://doi.org/10.1134/S1070428019100270
Karapetyan, L.V. and Tokmajyan, G.G., Russ. J. Org. Chem., 2019, vol. 55, p. 727. https://doi.org/10.1134/S1070428019050257
Rukovodstvo po provedeniyu issledovanii doklinicheskikh lekarstvennykh sredstv (A Guide to Preclinical Trials of Drugs). Red. A.N. Mironov. M.: Meditsina, 2012.
Mashkovskii, M.D., Lekarstvennye sredstva (Medicines), Moscow: Novaya Volna, 2020, 16th ed.
Avetissyan, A. and Karapetyan, L., Synth. Commun., 2009, vol. 39, p. 7. https://doi.org/10.1080/00397910701739022
ACKNOWLEDGMENTS
The authors thank the staff of the Molecular Structure Research Center (Yerevan, Armenia) for performing spectral studies and elemental analysis of the synthesized compounds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interest.
Additional information
Translated from Zhurnal Organicheskoi Khimii, 2021, Vol. 57, No. 12, pp. 1759–1765 https://doi.org/10.31857/S0514749221120119.
Rights and permissions
About this article
Cite this article
Karapetyan, L.V., Tokmajyan, G.G. & Stepanyan, H.M. Synthesis and Antibacterial Activity of N-Substituted 2-(Benzylimino)-4-styryl-2,5-dihydrofuran-3-carboxamides. Russ J Org Chem 57, 1974–1978 (2021). https://doi.org/10.1134/S1070428021120113
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428021120113