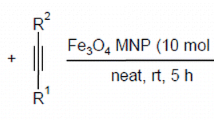Abstract
Pyrazoles and hydrazones, as two significant kinds of potentially bioactive compounds, were produced with good to excellent yields by condensation of β-dicarbonyl compounds with hydrazines in aqueous media in the presence of Fe3O4@CeO2 nanocomposite as an efficient heterogeneous nanocatalyst. The magnetic nanocatalyst can readily be separated using an external magnet and reused at least six times without significant loss in activity. The products were characterized by IR and 1H and13C NMR spectra.







Similar content being viewed by others
REFERENCES
Bahri-Laleh, N., Nekoomanesh-Haghighi, M., and Amin Mirmohammadi, S., J. Organomet. Chem., 2012, vol. 719, p. 74. https://doi.org/10.1016/j.jorganchem.2012.08.017
Rahmatiyan, S., Bahri-Laleh, N., Hanifpour, A., and Nekoomanesh-Haghighi, M., Polym. Int., 2019, vol. 68, p. 94. https://doi.org/10.1002/pi.5700
Climent, M.J., Corma, A., and Iborra, S., RSC Adv., 2012, vol. 2, p. 16. https://doi.org/10.1039/C1RA00807B
Gad-Allah, T.A., Fujimura, K., Kato, S., Satokawa, S., and Kojima, T., J. Hazard. Mater., 2008, vol. 154, p. 572. https://doi.org/10.1016/j.jhazmat.2007.10.068
Casas, L., Roig, A., Rodrı́guez, E., Molins, E., Tejada, J., and Sort, J., J. Non-Cryst. Solids, 2001, vol. 285, p. 37. https://doi.org/10.1016/S0022-3093(01)00429-X
Maleki, A., Hajizadeh, Z., and Abbasi, H., Carbon Lett., 2018, vol. 27, p. 42. https//doi.org/10.5714/CL.2018.27.042
Rostamnia, S., Alamgholiloo, H., and Jafari, M., Appl. Organomet. Chem., 2018, vol. 32, p. 4370. https://doi.org/10.1002/aoc.4370
Maggio, B., Daidone, G., Raffa, D., Plescia, S., Mantione, L., Cutuli, V.M.C., Mangano, N.G., and Caruso, A., Eur. J. Med. Chem., 2001, vol. 36, p. 737. https://doi.org/10.1016/S0223-5234(01)01259-4
Choi, W.K., El-Gamal, M.I., Choi, H.S., and Baek, D., Eur. J. Med. Chem., 2011, vol. 46, p. 5754. https://doi.org/10.1016/j.ejmech.2011.08.013
Bernardino, A.M.R., Gomez, A.O., Charret, K.S., Freitas, A.C.C., Machado, G.M.C., CantoCavalheiro, M.M., Leon, L.L., and Amaral, V.F., Eur. J. Med. Chem., 2006, vol. 41, p. 80. https://doi.org/10.1016/j.ejmech.2005.10.007
Prakash, O., Kumar, R., and Sehrawat, R., Eur. J. Med. Chem., 2009, vol. 44, p. 1763. https://doi.org/10.1016/j.ejmech.2008.03.028
Jayanna, N.D., Vagdevi, H.M., Dharshan, J.C., Raghavendra, R., and Telkar, S.B., Med. Chem. Res., 2013, vol. 22, p. 5814. https://doi.org/10.1007/s00044-013-0565-9
Cottineau, B., Toto, P., Marot, C., Pipaud, A., and Chenault, J.,Bioorg. Med. Chem. Lett., 2002, vol. 12, p. 2105. https://doi.org/10.1016/S0960-894X(02)00380-3
Pereira, A.L., Barreiro, E.J.L., Freitas, A.C.C., Correa, C.J.C., and Gomes, L.N.L.F., J. Liq. Chromatogr., 1991, vol. 14, p. 1161. https://doi.org/10.1080/01483919108049310
Martin, S.F., Pure Appl. Chem., 2009, vol. 81, p. 195. https://doi.org/10.1351/PAC-CON-08-07-03
Meyer, C.D., Joiner, C.S., and Stoddart, J.F., Chem. Soc. Rev., 2007, vol. 36, p. 1705. https://doi.org/10.1039/B513441M
Gawronski, J., Wascinska, N., and Gajewy, J., Chem. Rev., 2008, vol. 108, p. 5227. https://doi.org/10.1021/cr800421c
Tehrani, K.A. and De Kimpe, N., Science of Synthesis, Padwa, A., Ed., Stuttgart: Thieme, 2004, vol. 27, p. 245.
Dias, E.L., Brookhart, M., and White, P.S., J. Am. Chem. Soc., 2001, vol. 123, p. 2442. https://doi.org/10.1021/ja003608g
Dias, E.L., Brookhart, M., and White, P.S., Chem. Commun., 2001, p. 423. https://doi.org/10.1039/B007815H
Zakerinasab, B., Nasseri, M.A., Hassani, H., and Samieadel, M.M.,Res. Chem. Intermed., 2016, vol. 42, p. 3169. https://doi.org/10.1007/s11164-015-2204-1
Hassani, H., Nasseri, M.A., Zakerinasab, B., and Rafiee, F.,Appl. Organomet. Chem., 2016, vol. 30, no. 6, p. 408. https://doi.org/10.1002/aoc.3447
Hassani, H., Zakerinasab, B., Nasseri, M.A., and Shavakandi, M.,RSC Adv., 2016, vol. 6, p. 17560. https://doi.org/10.1039/C5RA24252E
Hassani, H., Zakerinasab, B., and Hossien Poor, H., Appl. Organomet. Chem., 2017, vol. 32, p. 3945. https://doi.org/10.1002/aoc.3945
Hassani, H., Zakerinasab, B., and Nozarie, A., Asian J. Green Chem., 2018, vol. 2, p. 59. https://doi.org/10.22631/ajgc.2017.101572.1032
Wei, Y., Han, B., Hu, X., Lin, Y., Wang, X., and Deng, X.,Procedia Eng., 2012, vol. 27, p. 632. https://doi.org/10.1016/j.proeng.2011.12.498
Fan, X.-W., Lei, T., Zhou, C., Meng, Q.-Y., Chen, B., Tung, C.-H., and Wu, L-Z., J. Org. Chem., 2016, vol. 81, p. 7127. https://doi.org/10.1021/acs.joc.6b00992
Zakerinasab, B., Nasseri, M.A., Hassani, H., and Samieadel, M.M.,Res. Chem. Intermed., 2016, vol. 42, p. 3169. https://doi.org/10.1007/s11164-015-2204-1
Hu, J., Chen, S., Sun, Y., Yang, J., and Rao, Y., Org. Lett., 2012, vol. 14, p. 5030. https://doi.org/10.1021/ol3022353
Nakamichi, N., Kawashita, Y., and Hayashi, M., Synthesis, 2004, vol. 2004, no. 7, p. 1015. https://doi.org/10.1055/s-2004-822343
Akbari, A., Org. Chem. Res., 2017, vol. 3, no. 2, p. 145. https://doi.org/10.22036/org.chem.2017.46823.1047
Xiong, W., Chen, J.X., Liu, M.C., Ding, J.C., Wu, H.Y., and Su, W.K., J. Braz. Chem. Soc., 2009, vol. 20, p. 367. https://doi.org/10.1590/S0103-50532009000200023
Shetty, M.R. and Samant, S.D., Synth. Commun., 2012, vol. 42, p. 1411. https://doi.org/10.1080/00397911.2010.540365
Funding
This study was performed under financial support by the Payame Noor University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interests.
Rights and permissions
About this article
Cite this article
Hassani, H., Jahani, Z. Synthesis of 1,3,5-Trisubstituted Pyrazoles and Hydrazones Using Fe3O4@CeO2 Nanocomposite as an Efficient Heterogeneous Nanocatalyst. Russ J Org Chem 56, 485–490 (2020). https://doi.org/10.1134/S1070428020030185
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428020030185