Abstract
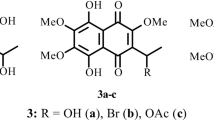
A 4-step preparative conversion of the available sea urchins pigment spinochrome E into the holothurian Polycheira rufescens pigment namakochrome was developed. Spinochrome E was converted into hexaacetate, and one β-acetate group in the quinone core of the latter was selectively removed. The resulting pentaacetate was treated with diazomethane, and all protective acetate groups were removed by treatment with methanolic HCl. The total yield of namakochrome over 4 steps was ~46–51%.



Similar content being viewed by others
REFERENCES
Thomson, R.H., Naturally Occurring Quinones, 4th Edn., London–New York: Blackie Academic & Professional, 1997.
Lebedev, A.V., Ivanova, M.V., and Levitsky, D.O., Life Sci., 2005, vol. 76, p. 863. https://doi.org/10.1016/j.lfs.2004.10.007
Lebedev, A.V., Ivanova, M.V., and Levitsky, D.O., Hemoglobin, 2008, vol. 32, p. 165. https://doi.org/10.1080/03630260701700017
Mishchenko, N.P., Fedoreev, S.A., and Bagirova, V.L., Pharm. Chem. J., 2003, vol. 37, p. 48. https://doi.org/10.1023/A:1023659331010
Jeong, S.H., Kim, H.K., Song, I.S., Noh, S.J., Marquez, J., Ko, K.S., Rhee, B.D., Kim, N., Mishchenko, N.P., Fedoreyev, S.A., Stonik, V.A., and Han, J., Mar. Drugs, 2014, vol. 12, p. 4602. https://doi.org/10.3390/md12084602
Seo, D.Y., McGregor, R.A., Noh, S.J., Choi, S.J., Mishchenko, N.P., Fedoreyev, S.A., Stonik, V.A., and Han, J., Mar. Drugs, 2015, vol. 13, p. 5722. https://doi.org/10.3390/md13095722
Lee, S.R., Pronto, J.R.D., Sarankhuu, B.-E., Ko, K.S., Rhee, B.D., Kim, N., Mishchenko, N.P., Fedoreyev, S.A., Stonik, V.A., and Han, J., Mar. Drugs, 2014, vol. 12, p. 3560. https://doi.org/10.3390/md12063560
Soliman, A.M., Mohamed, A.S., and Marie, A.S., Austin J. Endocrinol. Diabetes, 2016, vol. 3, p. 1045.
Yoon, C.S., Kim, H.K., Mishchenko, N.P., Vasileva, E.A., Fedoreyev, S.A., Stonik, V.A., and Han, J., Mar. Drugs, 2018, vol. 17, p. 2 https://doi.org/10.3390/md17010002
Mukai, T., Bull. Chem. Soc. Jpn., 1960, vol. 33, p. 453. https://doi.org/10.1246/bcsj.33.453
Mukai, T., Bull. Chem. Soc. Jpn., 1960, vol. 33, p. 1234. https://doi.org/10.1246/bcsj.33.1234
Artyukov, A.A., Kupera, E.V., Rutskova, T.A., Makhan’kov, V.V., Glazunov, V.P., Kozlovskaya, E.P., and Kol’tsova, E.A., RF Patent no. 2411939, 2009; Byull. Izobret., 2011, no. 5.
Sabutski, Yu.E., Semenova, M.N., Yurchenko, E.A., Polonik, N.S., Denisenko, V.A., Dmitrenok, P.S., Semenov, V.V., and Polonik, S.G., Nat. Prod. Commun., 2017, vol. 12, p. 1081. https://doi.org/10.1177/1934578X1701200721
Moore, R.E., Singh, H., Chang, C.W.J., and Scheuer, P.J., Tetrahedron, 1967, vol. 23, p. 3271.
Smith, I. and Thomson, R.H., J. Chem. Soc. (C), 1961, p. 1008. https://doi.org/10.1039/JR9610001008
Minkin, V.I., Olekhnovich, L.P., and Zhdanov, Yu.A., Zh. Vsesoyuz. Khim. Obshch., 1977, vol. 22, p. 273.
Utkina, N.K. and Pokhilo, N.D., Nat. Prod. Commun., 2012, vol. 7, p. 901. https://doi.org/10.1177/1934578X1200700725
ACKNOWLEDGMENTS
The authors are grateful to Cand. Sci. (Phys.-Math.) V.P. Glazunov for recording the IR spectra.
Funding
The work was financially supported by the Russian Science Foundation (project no. 19-14-00047).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Polonik, S.G., Denisenko, V.A., Popov, R.S. et al. Preparative Monomethylation of 2,3,5,6,7,8-Hexahydroxynaphthalene-1,4-dione (Spinochrome E). The First Direct Synthesis of Namakochrome: the Natural Pigment of the Holothurian Polycheira rufescens. Russ J Org Chem 56, 207–212 (2020). https://doi.org/10.1134/S1070428020020050
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428020020050