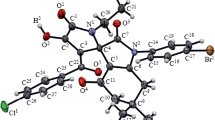
Abstract
1,3-Dipolar cycloaddition of diphenyldiazomethane to substituted 1H-pyrrole-2,3-diones followed by the loss of a nitrogen molecule leads to the formation of spiro[pyrrole-3,2'-oxiran]-2-ones (4-oxo-2,2,6-triphenyl-1-oxa-5-azaspiro[2.4]hept-6-enes), which are of interest for medical chemistry. The reaction proceeds under mild conditions with a good yield in the absence of catalysts and additives and does not require column chromatography to isolate the products.


Similar content being viewed by others
REFERENCES
Mehta, G. and Sengupta, S., Tetrahedron, 2017, vol. 73, p. 6223. https://doi.org/10.1016/j.tet.2017.09.030
Close, A., Mauli, R., and Sigg, H.P., Helv. Chim. Acta, 1966, vol. 49, p. 204. https://doi.org/10.1002/hlca.660490126
Sakamura, S., Niki, H., Obata, Y., Sakai, R., and Matsumoto, T., Agric. Biol. Chem., 1969, vol. 33, p. 698. https://doi.org/10.1080/00021369.1969.10859365
Gehrt, A., Erkel, G., Anke, T., and Sterner, O., J. Antibiot., 1998, vol. 51, p. 455. https://doi.org/10.7164/antibiotics.51.455
Koizumi, F., Matsuda, Y., and Nakanishi, S., J. Antibiot., 2003, vol. 56, p. 464. https://doi.org/10.7164/antibiotics.56.464
Koizumi, F., Ishiguro, H., Ando, K., Kondo, H., Yoshida, M., Matsuda, Y., and Nakanishi, S., J. Antibiot., 2003, vol. 56, p. 603. https://doi.org/10.7164/antibiotics.56.603
Dandia, A., Singh, R., Saha, M., and Shivpuri, A., Pharmazie, 2002, vol. 57, p. 602.
Dandia, A., Singh, R., and Bhaskaran, S., Ultrason. Sonochem., 2011, vol. 18, p. 1113. https://doi.org/10.1016/j.ultsonch.2010.12.010
Schönberg, A. and Junghans, K., Chem. Ber., 1963, vol. 96, p. 3328. https://doi.org/10.1002/cber.19630961234
Eistert, В., Muller, G.W., and Arackal, T.J., Liebigs Ann.Chem., 1976, vol. 6, p. 1023. https://doi.org/10.1002/jlac.197619760607
El-Nabi, H.A.A., Tetrahedron Lett., 1997, vol. 53, p. 1813. https://doi.org/10.1016/S0040-4020(96)01107-6
Okaev, E.V. and Zvonok, A.M., Chem. Heterocycl. Comp., 1995, vol. 31, p. 249. https://doi.org/10.1007/BF01373539
Smith, L.I. and Howard, K.L., Org. Synth., 1944, vol. 24, p. 53. https://doi.org/10.15227/orgsyn.024.0053
Ziegler, E., Kollenz, G., and Igel, H., Monatsh. Chem., 1972, vol. 103, p. 450. https://doi.org/10.1007/BF00904947
Silaichev P.S., Dmitriev M.V., Aliev Z.G., and Maslivets A.N. Russ. J. Org. Chem. 2010, 46, 1173–1177. https://doi.org/10.1134/S1070428010080105
Funding
The work was financially supported by the Russian Science Foundation (project no. 19-13-00290).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Moroz, A.A., Zhulanov, V.E., Dmitriev, M.V. et al. Synthesis of Oxirane Derivatives of 1H-Pyrrole-2,3-diones. Russ J Org Chem 56, 193–196 (2020). https://doi.org/10.1134/S1070428020020025
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428020020025