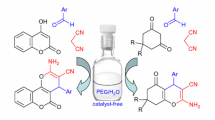
Abstract
Clean, practical, and efficient electrochemical synthesis of pharmaceutically relevant 4H-pyran derivatives by one-pot three-component combination of an aryl aldehyde, malononitrile, and a dicarbonyl compoundis developed. The synthesis is performed in ethanol with lithium perchlorate as a supporting electrolyte in an undivided cell on a platinum electrode under constant potential electrolysis conditions.
Similar content being viewed by others
References
Nefzi, A., Ostresh, J.M., and Houghten, R.A., Chem. Rev., 1997, vol. 97, p. 449.
Evans, B.E., Rittle, K.E., Bock, M.G., Di Pardo, R.M., Freidinger, R.M., Whitter, W.L., Lundell, G.F., Veber, D.F., Anderson, P.S., Chang, R.S.L., Lotti, V.J., Cerino, D.J., Chen, T.B., Kling, P.J., Kunkel, K.A., Springer, J.P., and Hirshfield, J., J. Med. Chem., 1988, vol. 31, p. 2235. doi https://doi.org/10.1021/jm00120a002
Poupaert, J. and Carato, P., Curr Med Chem., 2005, vol. 12, p. 877.
Armstrong, R.W., Combs, A.P., Tempest, P.A., Brown, S.D., and Keating, T.A., Acc. Chem. Res., 1996, vol. 29, p. 123.
Terret, N.K., Gardner, M., Gordon, D.W., and Kobylecki, R.J., Tetrahedron, 1995, vol. 51, p. 8135.
Wender, P.A., Handy, S.T., and Wright, D.L., Chem. Ind., 1997, vol. 765, p. 767.
Trost, B.M., Angew. Chem. Int. Ed., 1995, vol. 34, p. 259.
Mironov, M.A., QSAR Comb. Sci., 2006, vol. 25, p. 423.
Martin, N., Pascual, C., Seoane, C., and Soto, J.L., Heterocycles, 1987, vol. 26, p. 2811.
Adbel-Fattah, A.H., Hesien, A.M., Metwally, S.A., and Elnagdi, M.H., Liebigs Ann Chem., 1989, p. 585.
Quintela, J.M., Peinador, C., and Moreira, M.J., Tetrahedron, 1995, vol. 51, p. 5901.
Srivastava, S., Batra, S., and Bhaduri, A.P., Indian J Chem Sect B, 1996, vol. 35B, p. 602.
Akiyoshi, A., Takashi, S., Naohisa, O., Yasuo, S., Motoniro, S., Junji, N., and Masayosh, K., PCT Int. Appl., JP2009179589 (A), 2009.
Kitamura, R.O.S.P., Romoff, M.C.M., Young, M.J., and Kato, J.H.G., Phytochem. 2006, vol. 67, p. 2398.
Tangmouo, J.G., Meli, A.L., Komguem, J., Kuete, V., Ngounou, F.N., Lontsi, D., Beng, V.P., Choudhary, M.I., and Sondengam, B.L., Tetrahedron Lett., 2006, vol. 47, p. 3067. doi https://doi.org/10.1016/j.tetlet.2006.03.006
Cocco, M.T., Congiu, C., and Onnis, V., Bioorg. Med. Chem., 2003, vol. 11, p. 495.
Ballini, R., Bosica, G., Conforti, M.L., Maggi, R., Mazzacani, A., Righi, P., and Sartori, G., Tetrahedron, 2001, vol. 57, p. 1395.
Pratap, U.R., Jawale, D.V., Netankar, P.D., and Mane, R.A., Tetrahedron Lett., 2011, vol. 52, p. 5817.
Li, Y., Chen, H., Shi, C., Shi, D., and Ji, S., J. Comb. Chem., 2010, vol. 12, p. 231.
Devi, I. and Bhuyan, P.J., Tetrahedron Lett., 2004, vol. 45, p. 8625.
Akbarzadeh, T., Rafinejad, A., Mollaghasem, J.M., and Safari, M., Arch. Pharm. Chem. Life Sci., 2012, vol. 345, p. 386.
Mahmoodi, A., Aliabadi, A., Emami, S., and Safavi, M., Chem. Life Sci., 2010, vol. 343, p. 41.
Paul, S., Bhattacharyya, P., and Das, A.R., Tetrahedron Lett., 2011, vol. 52, p. 4636.
Bhattacharyya, P., Prodhan, K., Paul, S., and Das, A.R., Tetrahedron. Lett., 2012, vol. 53, p. 4687.
Daqing, Shi., Wu, Nan., and Qiya, Zhuang., Chin. J. Chem., 2009, vol. 27, p. 167.
Tabatabaeian, K., Heidari, H., Mamaghani, M., and Mahmoodi, N.O., Appl. Organomet. Chem., 2012, vol. 26, p. 56.
Saeedi, M., Tetrahedron, 2010, vol. 66, p. 5345.
Guihuang, X., Liu, J., Den, J., Wang, T., Chen, W., and Zeng, B., Tetrahedron, 2011, vol. 67, p. 6202.
Banerjee, S., Horn, A., Khatri, H., and Sereda, G., Tetrahedron Lett., 2011, vol. 52, p. 1878.
Anastas, P. and Eghbali, N., Chem. Soc. Rev., 2010, vol. 39, p. 301.
Ramachary, D.B. and Kishor, M., J. Org. Chem., 2007, vol. 72, p. 5056.
Ganem, B., Acc. Chem. Res., 2009, vol. 42, p. 463.
Balamurugan, K., Perumal, S., and Menendez, J.C., Tetrahedron, 2011, vol. 67, p. 3201.
Muschelknautz, C., Frank, W., and Muller, T., J. Org. Lett., 2011, vol. 13, p. 2556.
D’Souza, D., Rominger, M., Muller, F., Angew, T.J., Chem. Int. Ed., 2005, vol. 44, p. 153.
Hernandez, J.G., Eur. J. Org. Chem., 2010, vol. 75, 7107.
Li, S., Wang, J.X., Wen, X., and Ma, X., Tetrahedron, 2011, vol. 67, p. 849.
Singh, S., Sharma, L.K., Saraswat, A., Siddiqui, I.R., Kehri, H.K., Singh, R.K.P., RSC Adv., 2013, vol. 3, p. 4237.
Siddiqui, I.R., Srivastava, A., Shamim, S., Srivastava, A., Shireen, Waseem, M.A., Singh, R.K.P., Synlett, 2013, p. 2586.
Upadhyay, A., Sharma, L.K., Singh, V.K., and Singh, R.K.P., Tetrahedron Lett., 2016.
Singh, V.K., Sharma, L.K., Singh, R.K.P., Tetrahedron Lett., 2016, vol. 57, p. 407.
Sharma, L.K., Singh, S., Singh, VK., Singh, R.K.P., J. Indian Chem. Soc., 2016, vol. 93, p. 827.
Sharma, L.K. Saraswat, A., Singh, S., Singh, R.K.P., Proceedings of NASI, 2015, vol. 85, p. 29.
Moshtaghi, Z.A., Eskandari, I., and Moghani, D., Chem. Sci. Trans., 2012, vol. 1, p. 91.
Ranu, B.C. and Banerjee, S., Indian J. Chem. Soc., 2008, vol. 47, p. 1108.
Gurumurthi, S., Sundari, V., and Valliappan, R., Eur. J.Chem., 2009, vol. 6, p. 466.
Yu, L.Q., Liu, F., You, Q.D., Org. prep. Proced. Int., 2009, vol. 41, p. 77.
Hasaninejad, A., Shekouhy M., Golzar N., Zare A., and Doroodmand M.M., Appl. Catal. A, 2011, vol. 402, p. 11.
Piao M.Z., Imafuku K., Tetrahedron Lett., 1997, vol. 38, p. 5301.
Stoyanov, E.V., Ivanov, I.C., Heber D.S., Molecules, 2000, vol. 5, p. 19.
Kumar, D., Reddy, V.B., Sharad, U., Dube, and Kapur, S., Eur. J. Med. Chem, 2009, vol. 44, p. 3805.
Peng Y., Song G., and Huang F., Monatsherfte fur Chemie, 2005, vol. 136, p. 727.
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Organicheskoi Khimii, 2019, Vol. 55, No. 5, pp. 815.
Rights and permissions
About this article
Cite this article
Malviya, J., Kala, S., Sharma, L.K. et al. Efficient Three-Component One-Pot Synthesis of 4H-Pyrans. Russ J Org Chem 55, 686–693 (2019). https://doi.org/10.1134/S1070428019050178
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428019050178