Abstract
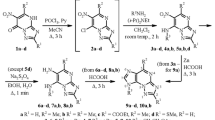
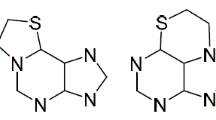
The alkylation of purine-6-thione with prenyl bromide and 3-butenyl bromide in the presence of alkali gave 6-[prenyl(but-3-enyl)sulfanyl]purines which reacted with halogens to afford fused [1,3]thiazino[2,3-i]purinium systems.
Similar content being viewed by others
References
Volonte, C. and Greene, L.A., Neurochem., 1992, vol.58, p.700. doi https://doi.org/10.1111/j.1471-4159.1992.tb09774.x
Kochergin, P.M., Aleksandrova, E.V., Tolvinskaya, L.S., Zhukova, I.B., Pukhal’skaya, V.G., Telegin, L.Yu., Pevnitskii, L.A., and Korsunskii, V.S., Pharm. Chem. J., 2000, vol.34, p.579. doi https://doi.org/10.1023/A:1010383903327
Yadav, V., Chu, C.K., and Rais, R.H., J. Med. Chem., 2004, vol.47, p.1987. doi https://doi.org/10.1021/jm030537y
Biagi, G., Costantini, A., Costantino, L., Giorgi, I., Livi, O., Pecorari, P., Rinaldi, M., and Scartoni, V., J. Med. Chem., 1996, vol.39, p.2529. doi https://doi.org/10.1021/jm950876u
Aleksandrova, E.V., Kochergin, P.M., and Valashek, I.E., Pharm. Chem. J., 2001, vol.35, p.172. doi https://doi.org/10.1023/a:1010418231536
Press, J.B., Hajos, Z.G., and Sawyers, R.A., Tetrahedron Lett., 1990, vol.31, p.1373. doi https://doi.org/10.1016/S0040-4039(00)88809-7
Press, J.B., McNally, J.J., Hajos, Z.G., and Sawyers, R.A., J. Org. Chem., 1992, vol.57, p.6335. doi https://doi.org/10.1021/jo00049a052
Biagi, G., Costantini, A., Costantino, L., Giorgi, I., Livi, O., Pecorari, P., Rinaldi, M., and Scartoni, V., J. Med. Chem., 1996, vol.39, p.2529. doi https://doi.org/10.1021/jm950876u
Rinaldi, M., Pecorari, P., Costantino, L., Provvisionato, A., Malagoli, M., and Cermelli, C., Farmaco, 1992, vol.47, p.1315.
Skoog, F., Hamzi, H.Q., Szweykowska, A.M., Leonard, N.J., Carraway, K.L., Fujii, T., Helgeson, J.P., and Loeppky, R.N., Phytochem., 1967, vol.6, p.1169. doi https://doi.org/10.1016/s0031-9422(00)86080-x
Kim, D.G. and Galina, Yu.R., Chem. Heterocycl. Compd., 2004, vol.40, p.1339. doi https://doi.org/10.1007/s10593-005-0076-9
Vas’kevich, R.I., Vas’kevich, A.I., Rusanov, E.B., Staninets, V.I., and Vovk, M.V., Russ. J. Org. Chem., 2013, vol.49, p.123. doi https://doi.org/10.1134/S1070428013010211
Frolova, T.V., Bahteeva, E.I., and Kim, D.G., Butlerov Comm., 2011, vol.26, p.70.
Slivka, N.Yu., Gevaza, Yu.I., and Staninets, V.I., Chem. Heterocycl. Compd., 2004, vol.40, p.660. doi https://doi.org/10.1023/B:COHC.0000037323.22839.9f
Il’inykh, E.S. and Kim, D.G., Chem. Heterocycl. Compd., 2011, vol.47, p.636. doi https://doi.org/10.1007/s10593-011-0809-x
Kim, D.G., Sudolova, N.M., Slepuhin, P.A., and Charushin, V.N., Chem. Heterocycl. Compd., 2011, vol.46, p.1420. doi https://doi.org/10.1007/s10593-011-0686-3
Tarasova, N.M., Candidate Sci. (Chem.) Dissertation, Chelyabinsk, 2017.
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © K.Yu. Petrova, D.G. Kim, 2019, published in Zhurnal Organicheskoi Khimii, 2019, Vol. 55, No. 2, pp. 186–191.
Rights and permissions
About this article
Cite this article
Petrova, K.Y., Kim, D.G. Synthesis of [1,3]Thiazino[2,3-i]purinium Systems by Halocyclization of 6-(Prenylsulfanyl)- and 6-(But-3-enylsulfanyl)purines. Russ J Org Chem 55, 142–146 (2019). https://doi.org/10.1134/S1070428019020039
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428019020039