Abstract
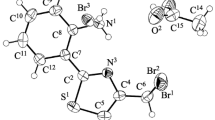
Iodination of 2-[allyl(methallyl)sulfanyl]-6-(trifluoromethyl)pyrimidin-4(3H)-ones was accompanied by cyclization to 2,3-dihydro[1,3]thiazolo[3,2-a]pyrimidin-4-ium triiodides. 3-(Iodomethyl)-3-methyl- 7-oxo-5-(trifluoromethyl)-2,3-dihydro[1,3]thiazolo[3,2-a]pyrimidin-4-ium triiodide was reduced with sodium iodide to 3,3-dimethyl-7-oxo-5-(trifluoromethyl)-2,3-dihydro[1,3]thiazolo[3,2-a]pyrimidin-4-ium iodide.
Similar content being viewed by others
References
Togninelli, A., Carmi, C., Petricci, E., Mugnaini, C., Massa, S., Corelli, F., and Botta, M., Tetrahedron Lett., 2006, vol. 47, p. 65.
Kamalakannan, P. and Venkappayya, D., J. Inorg. Biochem., 2002, vol. 90, p. 22.
Ondi, L., Lefebire, O., and Schlosser, M., Eur. J. Org. Chem., 2004, p. 3714.
Palumbo, A. and d’Ischia, M., Biochem. Biophys. Res. Commun., 2001, p. 793.
Noda, S., Takako, M., Takakura, S., Sakumato, S., Takatsuki, M., Yamasaki, K., Tateyama, S., and Yamaguchi, R., Arch. Toxicol., 2005, p. 414.
Frolova, T.V., Kim, D.G., and Slepukhin, P.A., Vestn. Yuzhn.-Ural. Gos. Univ., 2010, vol. 3, no. 11, p. 9.
Slivka, N.Yu., Gevaza, Yu.I., and Staninets, V.I., Chem. Heterocycl. Compd., 2004, vol. 40, p. 660.
Kim, D.G. and Shmygarev, V.I., Chem. Heterocycl. Compd., 1995, vol. 31, p. 183.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., Howard, J.A.K., and Puschmann, H., J. Appl. Crystallogr., 2009, vol. 42, p. 339.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.V. Frolova, D.G. Kim, P.A. Slepukhin, 2016, published in Zhurnal Organicheskoi Khimii, 2016, Vol. 52, No. 9, pp. 1356–1358.
Rights and permissions
About this article
Cite this article
Frolova, T.V., Kim, D.G. & Slepukhin, P.A. Iodocyclization of 2-[allyl(methallyl)sulfanyl]-6-(trifluoromethyl)pyrimidin-4(3H)-ones. Russ J Org Chem 52, 1344–1347 (2016). https://doi.org/10.1134/S1070428016090165
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428016090165