Abstract
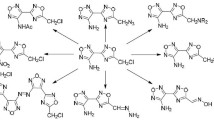
By condensation of 1-amino-2-hydroxynaphthalene with furoyl chloride in 1-methyl-2-pyrrolidone 2-(2-furyl)naphtho[1,2-d]oxazole was synthesized and brought into electrophylic substitution reactions: nitration, bromination, sulfonation, formylation, and acylation. The substituent commonly was introduced into the position 5 of the furan ring, but at the nitration and bromination electrophilic attack was directed both at the furan ring and the naphthalene fragment.
Similar content being viewed by others
References
Pechkin, A.A. and El’chaninov, M.M., Izv. Vuzov, Sev.-kav. Region, Estestv. nauki, 2000, vol. 28, p. 48.
Pozharskii, F.T. and Oleinikova, L.Ya., Khim. Geterotsikl. Soedin., 1972, p. 1555.
El’chaninov, M.M., Oleinikova, L.Ya., Simonov, A.M., Khim. Geterotsikl. Soedin., 1979, p. 1047.
Dorofeenko, G.N., Zh. Obshch. Khim., 1961, vol. 31, p. 994.
Pozharskii, A.F., Teoreticheskie osnovy khimii geterotsiklov (Theoretical Principles of Chemistry of Heterocycles), Moscow: Khimiya, 1985, p. 126.
Dunn, G.L., Actor, P., Di, and Pasquo, V.J., J. Med. Chem., 1966, vol. 9, p. 751.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Aleksandrov, A.S. Dedeneva, M.M. El’chaninov, 2011, published in Zhurnal Organicheskoi Khimii, 2011, Vol. 47, No. 5, pp. 707–709.
Rights and permissions
About this article
Cite this article
Aleksandrov, A.A., Dedeneva, A.S. & El’chaninov, M.M. Synthesis and some transformations of 2-(2-furyl)naphtho[1,2-d]oxazole. Russ J Org Chem 47, 710–713 (2011). https://doi.org/10.1134/S1070428011050095
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428011050095