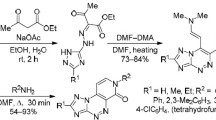
Abstract
2-Chloropyridine-3,4-diamine reacted with hetarenecarboxylic acids (pyridine-2-, pyridine-3-, and pyridine-4-carboxylic acids and 6-oxo-1,6-dihydropyridazine-3-carboxylic acid) in polyphosphoric acid at 160–170°C to give the corresponding 2-hetarylimidazo[4,5-c]pyridin-4-ones. Nitration of the latter with a mixture of concentrated nitric and sulfuric acids led to the formation of 2-hetaryl-7-nitroimidazo[4,5-c]pyridin-4-ones which were converted into 2-hetaryl-7-methylimidazo[4,5-d]pyridazin-4-ones by the action of hydrazine hydrate at 140–150°C.
Similar content being viewed by others
References
Gerhardt, G.A., Aldous, D.L., and Castle, R.N., J. Heterocycl. Chem., 1965, vol. 2, p. 247.
Austel, V., Heider, J., Hauel, N., Reiffen, M., Nickl, J., Meel, J., and Dideren, W., FRG Patent Appl. no. 3347290, 1985; Ref. Zh., Khim., 1986, no. 13O79P.
Heider, J., Hauel, N., Austel, V., Meel, J., and Dideren, W., FRG Patent Appl. no. 3445299, 1986; Ref. Zh., Khim., 1987, no. 3O147P.
Itai, T. and Suzuki, S., Chem. Pharm. Bull., 1960, vol. 8, p. 999.
Yanai, M., Kinoshita, T., Takeda, O., Sadaki, H., and Watanabe, H., Chem. Pharm. Bull., 1970, vol. 18, p. 1685.
Kenji, K., Hiromu, N., Yusho, O., Keizo, T., and Hirohisa, O., J. Heterocycl. Chem., 1984, vol. 21, p. 1249.
Gres’ko, S.V., Smolyar, N.N., and Yutilov, Yu.M., Russ. J. Org. Chem., 2001, vol. 37, p. 1026.
Yutilov, Yu.M. and Svertilova, I.A., Khim. Geterotsikl. Soedin., 1982, p. 705; Yutilov, Yu.M. and Smolyar, N.N., Khim. Geterotsikl. Soedin., 1984, p. 132; Yutilov, Yu.M. and Smolyar, N.N., Zh. Org. Khim., 1986, vol. 22, p. 1793; Yutilov, Yu.M., Smolyar, N.N., and Gres’ko, S.V., Russ. J. Org. Chem., 1995, vol. 31, p. 273; Yutilov, Yu.M. and Svertilova, I.A., Russ. J. Org. Chem., 1999, vol. 35, p. 583; Gres’ko, S.V., Smolyar, N.N., and Yutilov, Yu.M., Ukr. Khim. Zh., 2003, vol. 69, p. 110; Yutilov, Yu.M., Smolyar, N.N., Eres’ko, A.B., and Gres’ko, S.V., Russ. J. Org. Chem., 2004, vol. 40, p. 1015.
Yutilov, Yu.M. and Svertilova, I.A., Russ. J. Org. Chem., 1999, vol. 35, p. 451.
Sing, B., Heterocycles, 1984, vol. 22, p. 1801.
Yutilov, Yu.M. and Svertilova, I.A., USSR Inventor’s Certificate no. 1187438, 1985; Byull. Izobret., 1987, no. 29.
Bremer, O., Justus Liebigs Ann. Chem., 1939, vol. 539, p. 276; Koenigs, E., Milds, M., and Gurlt, H., Chem. Ber., 1924, vol. 57, p. 1179; Stetsenko, L.V. and Miroshnichenko, N.S., Ukr. Khim. Zh., 1973, vol. 49, p. 703; Svertilova, I.A., Smolyar, N.N., and Yutilov, Yu.M., Ukr. Khim. Zh., 1986, vol. 62, p. 64.
Gabriel, S., Chem. Ber., 1909, vol. 42, p. 655.
Author information
Authors and Affiliations
Additional information
Original Russian Text © N.N. Smolyar, D.A. Lomov, Yu.M. Yutilov, 2007, published in Zhurnal Organicheskoi Khimii, 2007, Vol. 43, No. 6, pp. 898–901.
Rights and permissions
About this article
Cite this article
Smolyar, N.N., Lomov, D.A. & Yutilov, Y.M. Synthesis of 2-hetarylimidazo[4,5-d]pyridazine derivatives. Russ J Org Chem 43, 897–900 (2007). https://doi.org/10.1134/S1070428007060164
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1070428007060164