Abstract
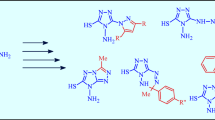
The reaction of dihydroimidazole-2-thiol with N-(3-chloro-2-oxopropyl)phthalimide gave 2-[3-(4,5-dihydroimidazol-2-ylsulfanyl)-2-oxopropyl]-1,3-dioxo-1H-isoindole which underwent intramolecular heterocyclization to dihydroimidazothiazole system by the action of a dehydrating agent. Treatment of 3-(1,3-dioxo-1H-isoindol-2-ylmethyl)-5,6-dihydroimidazo[2,1-b]thiazole with concentrated hydrochloric acid led to the formation of dihydroimidazo[2,1-b]thiazol-3-ylmethanamine. Water-soluble quaternary 3-(1,3-dioxo-1H-isoindol-2-ylmethyl)-5,6-dihydroimidazo[2,1-b]thiazolium salts were obtained by alkylation of 3-(1,3-dioxo-1H-isoindol-2-ylmethyl)-5,6-dihydroimidazo[2,1-b]thiazole with alkyl halides.
Similar content being viewed by others
References
Dianov, V.M., Zeleev, M.Kh., and Spirikhin, L.V., Russ. J. Org. Chem., 2005, vol. 41, p. 153.
Dianov, V.M., Zarudii, F.A., and Strokin, Yu.V., Khim.-Farm. Zh., 1998, no. 3, p. 27.
Iradyan, M.A., Aroyan, R.A., Engoyan, A.P., Pogosyan, A.V., Stepanyan, G.M., Arsenyan, F.G., and Garibdzhanyan, V.T., Khim.-Farm. Zh., 1991, no. 7, p. 38.
Sharpe, C.J., Shadbolt, R.S., Ashford, A., and Ross, J.W., J. Med. Chem., 1971, vol. 14, p. 977.
Author information
Authors and Affiliations
Additional information
Original Russian Text © V.M. Dianov, 2006, published in Zhurnal Organicheskoi Khimii, 2006, Vol. 42, No. 7, pp. 1046–1048.
Rights and permissions
About this article
Cite this article
Dianov, V.M. Synthesis of quaternary 3-(1,3-dioxo-1H-isoindol-2-ylmethyl)-5,6-dihydroimidazo[2,1-b]thiazolium salts. Russ J Org Chem 42, 1028–1030 (2006). https://doi.org/10.1134/S1070428006070165
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1070428006070165